Gene Expression Changes in the Human Diaphragm After Cardiothoracic Surgery
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Functions of the Mineralocorticoid Receptor in the Hippocampus By
Functions of the Mineralocorticoid Receptor in the Hippocampus by Aaron M. Rozeboom A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Cellular and Molecular Biology) in The University of Michigan 2008 Doctoral Committee: Professor Audrey F. Seasholtz, Chair Professor Elizabeth A. Young Professor Ronald Jay Koenig Associate Professor Gary D. Hammer Assistant Professor Jorge A. Iniguez-Lluhi Acknowledgements There are more people than I can possibly name here that I need to thank who have helped me throughout the process of writing this thesis. The first and foremost person on this list is my mentor, Audrey Seasholtz. Between working in her laboratory as a research assistant and continuing my training as a graduate student, I spent 9 years in Audrey’s laboratory and it would be no exaggeration to say that almost everything I have learned regarding scientific research has come from her. Audrey’s boundless enthusiasm, great patience, and eager desire to teach students has made my time in her laboratory a richly rewarding experience. I cannot speak of Audrey’s laboratory without also including all the past and present members, many of whom were/are not just lab-mates but also good friends. I also need to thank all the members of my committee, an amazing group of people whose scientific prowess combined with their open-mindedness allowed me to explore a wide variety of interests while maintaining intense scientific rigor. Outside of Audrey’s laboratory, there have been many people in Ann Arbor without whom I would most assuredly have gone crazy. -

The Switch from Fermentation to Respiration in Saccharomyces Cerevisiae Is Regulated by the Ert1 Transcriptional Activator/Repressor
INVESTIGATION The Switch from Fermentation to Respiration in Saccharomyces cerevisiae Is Regulated by the Ert1 Transcriptional Activator/Repressor Najla Gasmi,* Pierre-Etienne Jacques,† Natalia Klimova,† Xiao Guo,§ Alessandra Ricciardi,§ François Robert,†,** and Bernard Turcotte*,‡,§,1 ‡Department of Medicine, *Department of Biochemistry, and §Department of Microbiology and Immunology, McGill University Health Centre, McGill University, Montreal, QC, Canada H3A 1A1, †Institut de recherches cliniques de Montréal, Montréal, QC, Canada H2W 1R7, and **Département de Médecine, Faculté de Médecine, Université de Montréal, QC, Canada H3C 3J7 ABSTRACT In the yeast Saccharomyces cerevisiae, fermentation is the major pathway for energy production, even under aerobic conditions. However, when glucose becomes scarce, ethanol produced during fermentation is used as a carbon source, requiring a shift to respiration. This adaptation results in massive reprogramming of gene expression. Increased expression of genes for gluconeogenesis and the glyoxylate cycle is observed upon a shift to ethanol and, conversely, expression of some fermentation genes is reduced. The zinc cluster proteins Cat8, Sip4, and Rds2, as well as Adr1, have been shown to mediate this reprogramming of gene expression. In this study, we have characterized the gene YBR239C encoding a putative zinc cluster protein and it was named ERT1 (ethanol regulated transcription factor 1). ChIP-chip analysis showed that Ert1 binds to a limited number of targets in the presence of glucose. The strongest enrichment was observed at the promoter of PCK1 encoding an important gluconeogenic enzyme. With ethanol as the carbon source, enrichment was observed with many additional genes involved in gluconeogenesis and mitochondrial function. Use of lacZ reporters and quantitative RT-PCR analyses demonstrated that Ert1 regulates expression of its target genes in a manner that is highly redundant with other regulators of gluconeogenesis. -

Association Analyses of Known Genetic Variants with Gene
ASSOCIATION ANALYSES OF KNOWN GENETIC VARIANTS WITH GENE EXPRESSION IN BRAIN by Viktoriya Strumba A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Bioinformatics) in The University of Michigan 2009 Doctoral Committee: Professor Margit Burmeister, Chair Professor Huda Akil Professor Brian D. Athey Assistant Professor Zhaohui S. Qin Research Statistician Thomas Blackwell To Sam and Valentina Dmitriy and Elizabeth ii ACKNOWLEDGEMENTS I would like to thank my advisor Professor Margit Burmeister, who tirelessly guided me though seemingly impassable corridors of graduate work. Throughout my thesis writing period she provided sound advice, encouragement and inspiration. Leading by example, her enthusiasm and dedication have been instrumental in my path to becoming a better scientist. I also would like to thank my co-advisor Tom Blackwell. His careful prodding always kept me on my toes and looking for answers, which taught me the depth of careful statistical analysis. His diligence and dedication have been irreplaceable in most difficult of projects. I also would like to thank my other committee members: Huda Akil, Brian Athey and Steve Qin as well as David States. You did not make it easy for me, but I thank you for believing and not giving up. Huda’s eloquence in every subject matter she explained have been particularly inspiring, while both Huda’s and Brian’s valuable advice made the completion of this dissertation possible. I would also like to thank all the members of the Burmeister lab, both past and present: Sandra Villafuerte, Kristine Ito, Cindy Schoen, Karen Majczenko, Ellen Schmidt, Randi Burns, Gang Su, Nan Xiang and Ana Progovac. -
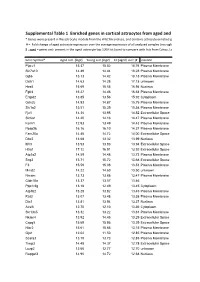
Supplemental Table 1 Enriched Genes in Cortical Astrocytes from Aged
Supplemental Table 1 Enriched genes in cortical astrocytes from aged and young-adult mice * Genes were present in the astrocyte module from the WGCNA analysis, and contains astrocyte enriched genes compared to microglia and oligodendrocytes # = Fold change of aged astrocyte expression over the average expression of all analyzed samples (microglia, astrocytes: young, old, with and without myelin contamination) $ ; aged = genes only present in the aged astrocyte top 1000 list (used to compare with lists from Cahoy, Lovatt, Doyle; see Fig. 4B), all = genes present in all astrocyte top 1000 lists Gene Symbol* Aged astr. (log2) Young astr.(log2) FC (aged/ aver.)# Location Ptprz1 15.37 15.02 18.76 Plasma Membrane Slc7a10 14.49 14.44 18.28 Plasma Membrane Gjb6 15.13 14.42 18.18 Plasma Membrane Dclk1 14.63 14.28 17.18 unknown Hes5 15.69 15.55 16.94 Nucleus Fgfr3 15.27 14.46 16.54 Plasma Membrane Entpd2 13.85 13.56 15.92 Cytoplasm Grin2c 14.93 14.87 15.75 Plasma Membrane Slc1a2 15.51 15.39 15.58 Plasma Membrane Fjx1 14.36 13.98 14.52 Extracellular Space Slc6a1 14.20 14.16 14.47 Plasma Membrane Kcnk1 12.93 13.49 14.43 Plasma Membrane Ppap2b 16.16 16.10 14.37 Plasma Membrane Fam20a 14.48 14.72 14.00 Extracellular Space Dbx2 13.68 13.32 13.99 Nucleus Itih3 13.93 13.93 13.94 Extracellular Space Htra1 17.12 16.91 13.92 Extracellular Space Atp1a2 14.59 14.48 13.73 Plasma Membrane Scg3 15.71 15.72 13.68 Extracellular Space F3 15.59 15.08 13.51 Plasma Membrane Mmd2 14.22 14.60 13.50 unknown Nrcam 13.73 13.88 13.47 Plasma Membrane Cldn10a 13.37 13.57 13.46 -

Downregulation of Nuclear and Cytoplasmic Chibby Is Associated with Advanced Cervical Cancer
6632 ONCOLOGY LETTERS 14: 6632-6644, 2017 Downregulation of nuclear and cytoplasmic Chibby is associated with advanced cervical cancer MING-CHANG YANG1,2*, SHANG-TAO CHIEN3,4*, TZU-FENG YANG2,5, SHIH-YI LIN3, TAI-MIN LEE3 and YI-REN HONG1,6,7 1 2 Department of Biological Sciences, National Sun Yat-sen University, Kaohsiung 80424; Laboratory of 3 Medical Research; Department of Pathology, Kaohsiung Armed Forces General Hospital, Kaohsiung 80284; 4Department of Medical Laboratory Sciences and Biotechnology, Fooyin University, Kaohsiung 83102; 5Department of Psychiatry, Kaohsiung Armed Forces General Hospital, Kaohsiung 80284; 6Department of Biochemistry, Faculty of Medicine, College of Medicine; 7Graduate Institute of Medicine, Kaohsiung Medical University, Kaohsiung 80708, Taiwan, R.O.C. Received March 11, 2014; Accepted June 9, 2017 DOI: 10.3892/ol.2017.7050 Abstract. Chibby has been identified as a putative tumor tumors, in which a significantly increased level of protein suppressor and antagonist to β-catenin, thereby controlling expression was detected. Chibby may be a tumor suppressor in the Wnt signaling pathway. Chibby is typically downregulated cervical cancer, since the dysregulation of Chibby expression is in numerous types of cancer and may be associated with associated with tumorigenesis in cervical cancer. Chibby and tumorigenesis. The present study aimed at clarifying the β-catenin expression together may potentially to a biomarker following: i) Whether Chibby antagonizes β-catenin in cervical for disease progression in cervical cancer. cancer; ii) whether Chibby and β-catenin mRNA expression is associated with cancer progression; and iii) whether Chibby Introduction and β-catenin expression may be used as a biomarker. -

ZNF226 Is Over-Expressed in Brain Metastatic Breast Cancer-PDF 111920
1 ZNF226 is over-expressed in brain metastatic breast cancer. 2 Shahan Mamoor East Islip, NY USA1 3 [email protected] 4 5 Metastasis to the brain is a clinical problem in patients with breast cancer1-3. We mined published 6 microarray data4,5 to compare primary and metastatic tumor transcriptomes to discover genes associated with brain metastasis in patients with metastatic breast cancer. We found that the zinc finger protein 226, 7 encoded by ZNF226 was among the genes whose expression was most different in the brain metastases of patients with brain metastatic breast cancer as compared to primary tumors of the breast. ZNF226 may be 8 relevant to processes underlying metastasis of primary tumor-derived cancer cells to the brain in humans 9 with metastatic breast cancer. 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 Keywords: breast cancer, brain metastases, central nervous system metastases, ZNF226, systems biology 26 of breast cancer, targeted therapeutics in breast cancer. 27 28 PAGE 1 1 One report described a 34% incidence of central nervous system metastases in trastuzumab- treated patients with breast cancer2. This alarmingly high frequency of CNS metastasis events demands 2 an enhanced understanding of the transcriptional makeup of brain metastatic tissues to support identification of therapeutic targets, whether they are treatment related or not. We performed a global 3 comparative analysis of primary and metastatic tumors in patients with brain metastatic breast cancer4,5. 4 We discovered significant differential and increased expression of ZNF226 in brain metastatic tissues of patients with metastatic breast cancer. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Genome-Wide DNA Methylation Profiling Identifies Differential Methylation in Uninvolved Psoriatic Epidermis
Genome-Wide DNA Methylation Profiling Identifies Differential Methylation in Uninvolved Psoriatic Epidermis Deepti Verma, Anna-Karin Ekman, Cecilia Bivik Eding and Charlotta Enerbäck The self-archived postprint version of this journal article is available at Linköping University Institutional Repository (DiVA): http://urn.kb.se/resolve?urn=urn:nbn:se:liu:diva-147791 N.B.: When citing this work, cite the original publication. Verma, D., Ekman, A., Bivik Eding, C., Enerbäck, C., (2018), Genome-Wide DNA Methylation Profiling Identifies Differential Methylation in Uninvolved Psoriatic Epidermis, Journal of Investigative Dermatology, 138(5), 1088-1093. https://doi.org/10.1016/j.jid.2017.11.036 Original publication available at: https://doi.org/10.1016/j.jid.2017.11.036 Copyright: Elsevier http://www.elsevier.com/ Genome-Wide DNA Methylation Profiling Identifies Differential Methylation in Uninvolved Psoriatic Epidermis Deepti Verma*a, Anna-Karin Ekman*a, Cecilia Bivik Edinga and Charlotta Enerbäcka *Authors contributed equally aIngrid Asp Psoriasis Research Center, Department of Clinical and Experimental Medicine, Division of Dermatology, Linköping University, Linköping, Sweden Corresponding author: Charlotta Enerbäck Ingrid Asp Psoriasis Research Center, Department of Clinical and Experimental Medicine, Linköping University SE-581 85 Linköping, Sweden Phone: +46 10 103 7429 E-mail: [email protected] Short title Differential methylation in psoriasis Abbreviations CGI, CpG island; DMS, differentially methylated site; RRBS, reduced representation bisulphite sequencing Keywords (max 6) psoriasis, epidermis, methylation, Wnt, susceptibility, expression 1 ABSTRACT Psoriasis is a chronic inflammatory skin disease with both local and systemic components. Genome-wide approaches have identified more than 60 psoriasis-susceptibility loci, but genes are estimated to explain only one third of the heritability in psoriasis, suggesting additional, yet unidentified, sources of heritability. -

Download The
PROBING THE INTERACTION OF ASPERGILLUS FUMIGATUS CONIDIA AND HUMAN AIRWAY EPITHELIAL CELLS BY TRANSCRIPTIONAL PROFILING IN BOTH SPECIES by POL GOMEZ B.Sc., The University of British Columbia, 2002 A THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE in THE FACULTY OF GRADUATE STUDIES (Experimental Medicine) THE UNIVERSITY OF BRITISH COLUMBIA (Vancouver) January 2010 © Pol Gomez, 2010 ABSTRACT The cells of the airway epithelium play critical roles in host defense to inhaled irritants, and in asthma pathogenesis. These cells are constantly exposed to environmental factors, including the conidia of the ubiquitous mould Aspergillus fumigatus, which are small enough to reach the alveoli. A. fumigatus is associated with a spectrum of diseases ranging from asthma and allergic bronchopulmonary aspergillosis to aspergilloma and invasive aspergillosis. Airway epithelial cells have been shown to internalize A. fumigatus conidia in vitro, but the implications of this process for pathogenesis remain unclear. We have developed a cell culture model for this interaction using the human bronchial epithelium cell line 16HBE and a transgenic A. fumigatus strain expressing green fluorescent protein (GFP). Immunofluorescent staining and nystatin protection assays indicated that cells internalized upwards of 50% of bound conidia. Using fluorescence-activated cell sorting (FACS), cells directly interacting with conidia and cells not associated with any conidia were sorted into separate samples, with an overall accuracy of 75%. Genome-wide transcriptional profiling using microarrays revealed significant responses of 16HBE cells and conidia to each other. Significant changes in gene expression were identified between cells and conidia incubated alone versus together, as well as between GFP positive and negative sorted cells. -

Cellular and Molecular Signatures in the Disease Tissue of Early
Cellular and Molecular Signatures in the Disease Tissue of Early Rheumatoid Arthritis Stratify Clinical Response to csDMARD-Therapy and Predict Radiographic Progression Frances Humby1,* Myles Lewis1,* Nandhini Ramamoorthi2, Jason Hackney3, Michael Barnes1, Michele Bombardieri1, Francesca Setiadi2, Stephen Kelly1, Fabiola Bene1, Maria di Cicco1, Sudeh Riahi1, Vidalba Rocher-Ros1, Nora Ng1, Ilias Lazorou1, Rebecca E. Hands1, Desiree van der Heijde4, Robert Landewé5, Annette van der Helm-van Mil4, Alberto Cauli6, Iain B. McInnes7, Christopher D. Buckley8, Ernest Choy9, Peter Taylor10, Michael J. Townsend2 & Costantino Pitzalis1 1Centre for Experimental Medicine and Rheumatology, William Harvey Research Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London, Charterhouse Square, London EC1M 6BQ, UK. Departments of 2Biomarker Discovery OMNI, 3Bioinformatics and Computational Biology, Genentech Research and Early Development, South San Francisco, California 94080 USA 4Department of Rheumatology, Leiden University Medical Center, The Netherlands 5Department of Clinical Immunology & Rheumatology, Amsterdam Rheumatology & Immunology Center, Amsterdam, The Netherlands 6Rheumatology Unit, Department of Medical Sciences, Policlinico of the University of Cagliari, Cagliari, Italy 7Institute of Infection, Immunity and Inflammation, University of Glasgow, Glasgow G12 8TA, UK 8Rheumatology Research Group, Institute of Inflammation and Ageing (IIA), University of Birmingham, Birmingham B15 2WB, UK 9Institute of -

And EZH2-Regulated Gene, Has Differential Roles in AR-Dependent and -Independent Prostate Cancer
www.impactjournals.com/oncotarget/ Oncotarget, Vol. 6, No.4 RUNX1, an androgen- and EZH2-regulated gene, has differential roles in AR-dependent and -independent prostate cancer Ken-ichi Takayama1,2, Takashi Suzuki3, Shuichi Tsutsumi4, Tetsuya Fujimura5, Tomohiko Urano1,2, Satoru Takahashi6, Yukio Homma5, Hiroyuki Aburatani4 and Satoshi Inoue1,2,7 1 Department of Anti-Aging Medicine, The University of Tokyo, Bunkyo-ku, Tokyo, Japan 2 Department of Geriatric Medicine, The University of Tokyo, Bunkyo-ku, Tokyo, Japan 3 Department of Pathology, Tohoku University Graduate School of Medicine, Sendai, Miyagi, Japan 4 Genome Science Division, Research Center for Advanced Science and Technology (RCAST), The University of Tokyo, Meguro-ku, Tokyo, Japan 5 Department of Urology, Graduate School of Medicine, The University of Tokyo, Bunkyo-ku, Tokyo, Japan 6 Department of Urology, Nihon University School of Medicine, Itabashi-ku, Tokyo, Japan 7 Division of Gene Regulation and Signal Transduction, Research Center for Genomic Medicine, Saitama Medical University, Hidaka, Saitama, Japan Correspondence to: Satoshi Inoue, email: [email protected] Keywords: RUNX1, androgen receptor, EZH2, prostate cancer Received: October 02, 2014 Accepted: December 09, 2014 Published: December 10, 2014 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. ABSTRACT Androgen receptor (AR) signaling is essential for the development of prostate cancer. Here, we report that runt-related transcription factor (RUNX1) could be a key molecule for the androgen-dependence of prostate cancer. We found RUNX1 is a target of AR and regulated positively by androgen. -

Supplementary Table S4. FGA Co-Expressed Gene List in LUAD
Supplementary Table S4. FGA co-expressed gene list in LUAD tumors Symbol R Locus Description FGG 0.919 4q28 fibrinogen gamma chain FGL1 0.635 8p22 fibrinogen-like 1 SLC7A2 0.536 8p22 solute carrier family 7 (cationic amino acid transporter, y+ system), member 2 DUSP4 0.521 8p12-p11 dual specificity phosphatase 4 HAL 0.51 12q22-q24.1histidine ammonia-lyase PDE4D 0.499 5q12 phosphodiesterase 4D, cAMP-specific FURIN 0.497 15q26.1 furin (paired basic amino acid cleaving enzyme) CPS1 0.49 2q35 carbamoyl-phosphate synthase 1, mitochondrial TESC 0.478 12q24.22 tescalcin INHA 0.465 2q35 inhibin, alpha S100P 0.461 4p16 S100 calcium binding protein P VPS37A 0.447 8p22 vacuolar protein sorting 37 homolog A (S. cerevisiae) SLC16A14 0.447 2q36.3 solute carrier family 16, member 14 PPARGC1A 0.443 4p15.1 peroxisome proliferator-activated receptor gamma, coactivator 1 alpha SIK1 0.435 21q22.3 salt-inducible kinase 1 IRS2 0.434 13q34 insulin receptor substrate 2 RND1 0.433 12q12 Rho family GTPase 1 HGD 0.433 3q13.33 homogentisate 1,2-dioxygenase PTP4A1 0.432 6q12 protein tyrosine phosphatase type IVA, member 1 C8orf4 0.428 8p11.2 chromosome 8 open reading frame 4 DDC 0.427 7p12.2 dopa decarboxylase (aromatic L-amino acid decarboxylase) TACC2 0.427 10q26 transforming, acidic coiled-coil containing protein 2 MUC13 0.422 3q21.2 mucin 13, cell surface associated C5 0.412 9q33-q34 complement component 5 NR4A2 0.412 2q22-q23 nuclear receptor subfamily 4, group A, member 2 EYS 0.411 6q12 eyes shut homolog (Drosophila) GPX2 0.406 14q24.1 glutathione peroxidase