Autonomic Influence on Pharmacologically-Induced Cardiac Arrhythmogenesis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Cardioactive Agents : Metoprolol, Sotalol and Milrinone. Influence of Myocardial Content and Systolic Interval
3Õ' î'qt ACUTE HAEMODYNAMIC EFFECTS OF THREE CARDIOACTIVE AGENTS : METOPROLOL, SOTALOL AND MILRINONE. INFLUENCE OF MYOCARDIAL CONTENT AND SYSTOLIC INTERVAL. by Rebecca Helen Ritchie, B.Sc (Hons) A thesis submitted for the degree of Doctor of Philosophy ln The University of Adelaide (Faculty of Medicine) February 1994 Department of Medicine (Cardiology Unit, The Queen Elizabeth Hospital) The University of Adelaide Adelaide, SA, 5000. ll ¡ r -tL',. r,0';(', /1L.)/'t :.: 1 TABLE OF CONTENTS Table of contents 1 Declaration vtl Acknowledgements v111 Publications and communications to learned societies in support of thesis D( Summary xl Chapter 1: General Introduction 1 1.1 Overview 2 1.2 Acute effeots of cardioactive drugs 3 1.2.1 Drug effects 4 l.2.2Determnants of drug effects 5 1.3 Myocardial drug gPtake of cardioactive agents 8 1.3.1 Methods of assessment in humans invívo 9 1.3.2 Results of previous studies 10 1.4Influence of cardioactive drugs on contractile state 11 1.4. 1 Conventional indices 11 I.4.2 The staircase phenomenon t2 1.4.3 The mechanical restitution curve t2 1.5 The present study t4 1.5.1 Current relevant knowledge of the acute haemodynamic effects of the cardioactive drugs under investigation r4 1.5.1.1 Metoprolol 15 1.5.1.2 Sotalol 28 1.5.1.3 Milrinone 43 1.5.2 Cunent relevant knowledge of the short-term pharmacokinetics of the cardioactive drugs under investigation 59 1.5.2.1Metoprolol 59 1.5.2.2 Sotalol 7I ll 1.5.2.3 Milrinone 78 1.5.3 Current relevant knowledge of the potential for rate-dependence of the effects of these -

Adrenoceptors Regulating Cholinergic Activity in the Guinea-Pig Ileum 1978) G.M
- + ! ,' Br. J. Pharmac. (1978), 64, 293-300. F'(O t.,," e reab- ,ellular PHARMACOLOGICAL CHARACTERIZATION OF THE PRESYNAPTIC _-ADRENOCEPTORS REGULATING CHOLINERGIC ACTIVITY IN THE GUINEA-PIG ILEUM 1978) G.M. Departmentof Pharmacology,Allen and HzmburysResearchLimited, Ware, Hertfordshire,SG12 ODJ I The presynaptic ct-adrenoceptors located on the terminals of the cholinergic nerves of the guinea- pig myenteric plexus have been characterized according to their sensitivities to at-adrenoceptor agonists and antagonists. 2 Electrical stimulation of the cholinergic nerves supplying the longitudinal muscle of the guinea-pig ! ileum caused a twitch response. Clonidine caused a concentration-dependent inhibition of the twitch i response; the maximum inhibition obtained was 80 to 95_o of the twitch response. Oxymetazoline and xylazine were qualitatively similar to clonidine but were about 5 times less potent. Phenylephrine and methoxamine also inhibited the twitch response but were at least 10,000 times less potent than clonidine. 3 The twitch-inhibitory effects of clonidine, oxymetazoline and xylazine, but not those of phenyl- ephrine or methoxamine, were reversed by piperoxan (0.3 to 1.0 lag/ml). 4 Lysergic acid diethylamide (LSD) inhibited the twitch response, but also increased the basal tone of the ileum. Mepyramine prevented the increase in tone but did not affect the inhibitory action of LSD. Piperoxan or phentolamine only partially antagonized the inhibitory effect of LSD. 5 Phentolamine, yohimbine, piperoxan and tolazoline were potent, competitive antagonists of the inhibitory effect of clonidine with pA2 values of 8.51, 7.78, 7.64 and 6.57 respectively. 6 Thymoxamine was a weak antagonist of clonidine; it also antagonized the twitch-inhibitory effect of morphine. -

The Organic Chemistry of Drug Synthesis
The Organic Chemistry of Drug Synthesis VOLUME 2 DANIEL LEDNICER Mead Johnson and Company Evansville, Indiana LESTER A. MITSCHER The University of Kansas School of Pharmacy Department of Medicinal Chemistry Lawrence, Kansas A WILEY-INTERSCIENCE PUBLICATION JOHN WILEY AND SONS, New York • Chichester • Brisbane • Toronto Copyright © 1980 by John Wiley & Sons, Inc. All rights reserved. Published simultaneously in Canada. Reproduction or translation of any part of this work beyond that permitted by Sections 107 or 108 of the 1976 United States Copyright Act without the permission of the copyright owner is unlawful. Requests for permission or further information should be addressed to the Permissions Department, John Wiley & Sons, Inc. Library of Congress Cataloging in Publication Data: Lednicer, Daniel, 1929- The organic chemistry of drug synthesis. "A Wiley-lnterscience publication." 1. Chemistry, Medical and pharmaceutical. 2. Drugs. 3. Chemistry, Organic. I. Mitscher, Lester A., joint author. II. Title. RS421 .L423 615M 91 76-28387 ISBN 0-471-04392-3 Printed in the United States of America 10 987654321 It is our pleasure again to dedicate a book to our helpmeets: Beryle and Betty. "Has it ever occurred to you that medicinal chemists are just like compulsive gamblers: the next compound will be the real winner." R. L. Clark at the 16th National Medicinal Chemistry Symposium, June, 1978. vii Preface The reception accorded "Organic Chemistry of Drug Synthesis11 seems to us to indicate widespread interest in the organic chemistry involved in the search for new pharmaceutical agents. We are only too aware of the fact that the book deals with a limited segment of the field; the earlier volume cannot be considered either comprehensive or completely up to date. -

NATURE 199 Heart Was Removed with the Great Vessels Cut Close to the Heart, A~D the Organ Weighed
No. 49t5 January 11, 1964 NATURE 199 heart was removed with the great vessels cut close to the heart, a~d the organ weighed. The weights of each OH embryo and 1ts heart are seen tabulated in Fig. l. · CH • The largest embryos were seen at 37·5° C. The heart:; /\ /~_CH. CH2 N( . CH, from the group at 32·5° C were largest, even though the j 'H embryos bearing these hearts were smaller than those at Vv# the normal temperature. On gross examination we saw I that the increase in size at 32·5° C consisted of enlarge OH NR2 ment ~f the chambers of the heart as well as thickening IIa ~·~ - N(CH,.)2 of thmr walls. On microscopic section the increased CH . CH2 • N . R 2 heart tissue mass was found to be composed of heart I muscle and supporting tissues, not of an inflammatory exudate or cedoma. Two other gross changes were consistently present. In 00 II the low-temperature groups the intra-abdominal veins of the embryos as well as veins of the extra-embryonic to discover whether pronethalol also could produce local membranes were distended. Also the kidneys were en anresthesia. larged-both the mesonephric and metanephric kidneys. Guinea pigs were lightly anresthetized with sodium To seo whether the variation in the size of heart pentobarbitone (30 mgfkg intraperitoneally) and local resulting from the difference in temperature of incubation anresthetic potency determined by the intradermal weal would be observed in heart fragments transplanted to method of Biilbring and Wajda•. It was found that the chorioallantoic membrane, we removed the hearts of pronethalol is 1·8 times as active as procaine (log R = several 8-day-old embryos, pooled them, cut them into 0·270 ± 0·05). -

ACMD Advisory Council on the Misuse of Drugs
ACMD Advisory Council on the Misuse of Drugs Consideration of the naphthylpyrovalerone analogues and related compounds. 1 ACMD Advisory Council on the Misuse of Drugs Chair: Professor Les Iversen Secretary: Will Reynolds 3rd Floor (SW), Seacole Building 2 Marsham Street London SW1P 4DF Tel: 020 7035 0454 [email protected] Rt Hon Theresa May MP 2 Marsham Street London SW1P 4DF 7th July 2010 Dear Home Secretary, The ACMD indicated in its advice of 31st March 2010 on the cathinones that it would provide you with further advice on the naphthyl analogues of pyrovalerone (including naphyrone) and other such analogues. I have pleasure in attaching the Advisory Council on the Misuse of Drugs report on the ‘Consideration of the naphthylpyrovalerone analogues and related compounds’. The ACMD recognise the significant public health issue that ‘legal highs’ present. Our report references recent work on test purchases of a number of ‘legal highs’ that demonstrate their many and varied compositions. In this report the evidence highlights the dangers of purchasing compounds which are likely to contain harmful compounds and very often will not be the same as the material advertised and may be more harmful and illegal. Users of ‘legal highs’ should be acutely aware that just because it is being advertised as legal does not make a substance safe, nor may it be legal. Along with mephedrone and related compounds, the public health response should focus on the discrepancy between the compounds that are purported to be contained in the ‘legal high’ for sale and what the customer actually gets. -

Coronary Blood Flow Before and After Beta-Adrenergic Blockade
Brit. J. Pharmacol. (1966), 26, 713-722. EFFECTS OF ADRENALINE AND NORADRENALINE ON CORONARY BLOOD FLOW BEFORE AND AFTER BETA-ADRENERGIC BLOCKADE BY P. G. GAAL, A. A. KATTUS, A. KOLIN AND G. ROSS From the Departments of Medicine and Biophysics, University of California, Los Angeles (Received November 16, 1965) There are conflicting reports regarding the actions of adrenaline and noradrenaline on the coronary circulation. Some studies (Feinberg & Katz, 1958; Denison, Bardhanabaedya & Green, 1956) have indicated that these agents are solely coronary vasodilators, while others (Berne, 1958) have suggested that their primary action is vasoconstriction and that the increased coronary flow usually observed is secondary to metabolic changes associated with stimulation of the myocardium. Berne (1958) has stated that previous failures to demonstrate primary vasoconstriction may have been due to inadequate methodology. The present paper reports the use of electromagnetic flow meters and non-cannulating probes to determine the effects of adrenaline and noradrenaline on coronary arterial flow. The method enables rapid flow changes to be detected with minimum disturbance to the coronary vascular bed and also permits evaluation of changes in extravascular compression induced by the amines. Further clarification of the adrenergic mechanisms was obtained by comparing the effects of adrenaline and noradrenaline before and after blockade of the beta-adrenergic receptors by pronethalol and propranolol. METHODS Fourteen dogs weighing 18-28 kg were used. Technically satisfactory results were obtained in 12. Anaesthesia was produced by intravenous sodium pentobarbitone 30 mg/kg. After a median sternotomy, the pericardium was opened and the ascending aorta and 1-2 cm lengths of the proximal parts of the right coronary artery and the anterior descending branch of the left coronary artery were separated from adjacent tissues. -

New Approaches to the Uses of Beta Blocking Drugs in Hypertension
Journal of Human Hypertension (2000) 14, Suppl 1, S63–S68 2000 Macmillan Publishers Ltd All rights reserved 0950-9240/00 $15.00 www.nature.com/jhh New approaches to the uses of beta blocking drugs in hypertension BNC Prichard1, BR Graham1 and JM Cruickshank2 1Centre for Clinical Pharmacology, University College, 5 University Street, London WC1E 6JJ, UK; 242 Harefield, Long Melford, Suffolk CO10 9DE, UK After a slow start, beta-blockers have become widely dial infarction where they are often under used. There used as first-line agents in the treatment of hyperten- is some evidence that even in post-infarction patients sion, and recommended as such in recently published with co-existent chronic obstructive airways disease, guidelines. There is evidence that the beta1-selective usually regarded as a contra-indication, experience an agents are more efficacious than non-selective blockers improved 2-year survival with the use of beta-blockers. that inhibit both beta1 and beta2 receptors. Notwith- Recently they have also been demonstrated to improve standing some earlier evidence to the contrary, it prognosis in heart failure patients, previously regarded appears that beta1-selective drugs are equi-effective in as a contra-indication. Likewise, recent studies have young and elderly whites, younger, ie, under mid 60s, shown that atenolol was at least as effective as captopril blacks. It is with the combination of age and being black in improving the outlook in hypertensive patients with that beta-blockers are usually less useful than some non-insulin dependant diabetes. While earlier compari- other groups of antihypertensive drugs, most notably sons with the non-selective lipid soluble propranolol calcium antagonists and diuretics. -
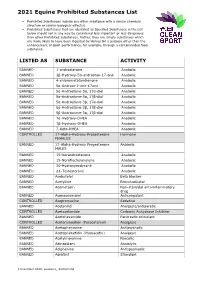
2021 Equine Prohibited Substances List
2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). Prohibited Substances that are identified as Specified Substances in the List below should not in any way be considered less important or less dangerous than other Prohibited Substances. Rather, they are simply substances which are more likely to have been ingested by Horses for a purpose other than the enhancement of sport performance, for example, through a contaminated food substance. LISTED AS SUBSTANCE ACTIVITY BANNED 1-androsterone Anabolic BANNED 3β-Hydroxy-5α-androstan-17-one Anabolic BANNED 4-chlorometatandienone Anabolic BANNED 5α-Androst-2-ene-17one Anabolic BANNED 5α-Androstane-3α, 17α-diol Anabolic BANNED 5α-Androstane-3α, 17β-diol Anabolic BANNED 5α-Androstane-3β, 17α-diol Anabolic BANNED 5α-Androstane-3β, 17β-diol Anabolic BANNED 5β-Androstane-3α, 17β-diol Anabolic BANNED 7α-Hydroxy-DHEA Anabolic BANNED 7β-Hydroxy-DHEA Anabolic BANNED 7-Keto-DHEA Anabolic CONTROLLED 17-Alpha-Hydroxy Progesterone Hormone FEMALES BANNED 17-Alpha-Hydroxy Progesterone Anabolic MALES BANNED 19-Norandrosterone Anabolic BANNED 19-Noretiocholanolone Anabolic BANNED 20-Hydroxyecdysone Anabolic BANNED Δ1-Testosterone Anabolic BANNED Acebutolol Beta blocker BANNED Acefylline Bronchodilator BANNED Acemetacin Non-steroidal anti-inflammatory drug BANNED Acenocoumarol Anticoagulant CONTROLLED Acepromazine Sedative BANNED Acetanilid Analgesic/antipyretic CONTROLLED Acetazolamide Carbonic Anhydrase Inhibitor BANNED Acetohexamide Pancreatic stimulant CONTROLLED Acetominophen (Paracetamol) Analgesic BANNED Acetophenazine Antipsychotic BANNED Acetophenetidin (Phenacetin) Analgesic BANNED Acetylmorphine Narcotic BANNED Adinazolam Anxiolytic BANNED Adiphenine Antispasmodic BANNED Adrafinil Stimulant 1 December 2020, Lausanne, Switzerland 2021 Equine Prohibited Substances List . Prohibited Substances include any other substance with a similar chemical structure or similar biological effect(s). -

Calorigenic Effect of Norepinephrine Correlated with Plasma Free Fatty Acid Turnover and Oxidation
Calorigenic Effect of Norepinephrine Correlated with Plasma Free Fatty Acid Turnover and Oxidation Daniel Steinberg, … , Elsworth R. Buskirk, Ronald H. Thompson J Clin Invest. 1964;43(2):167-176. https://doi.org/10.1172/JCI104901. Research Article Find the latest version: https://jci.me/104901/pdf Journal of Clinical Investigation Vol. 43, No. 2, 1964 Calorigenic Effect of Norepinephrine Correlated with Plasma Free Fatty Acid Turnover and Oxidation * DANIEL STEINBERG, PAUL J. NESTEL,t ELSWORTH R. BUSKIRK,4 AND RONALD H. THOMPSON (From the Laboratory of Metabolism, National Heart Institute, and the Metabolic Disease Branch, National Institute of Arthritis and Metabolic Diseases, Bethesda, Md.) The increase in oxygen consumption caused by consumption in man and that this is accompanied administration of epinephrine has been extensively by an increase in turnover of plasma FFA and an studied in animals and in man. Many physiologi- increase in rate of conversion of labeled plasma cal and biochemical correlations have been made FFA to respiratory C1402. All of these effects of and many different hypotheses have been ad- norepinephrine are abolished or radically re- vanced to explain this so-called calorigenic effect, duced when the patient has first been given an in- but there is still no clearly established consensus travenous dose of pronethalol [2-isopropylamino- regarding the mechanism or mechanisms involved 1-(2-naphthyl)ethanol hydrochloride], a drug that (1, 2). inhibits catecholamine stimulation of adrenergic In recent years it has become clear that one of ,8-receptors (8, 9) and that prevents the FFA the most striking metabolic effects of the cate- mobilization induced by catecholamines (10).1 A cholamines is their ability to stimulate mobiliza- preliminary report of these findings has been made tion of depot fat in the form of free fatty acids (11). -

Optimizing Drug Binding Affinity: (Semi) Empirical Studies
Approaches of Classical Medicinal Chemistry Optimizing Drug Binding Affinity: (Semi) Empirical Studies • • Drug Design: Optimizing Binding Interactions •increase activity and reduce dose levels •increase selectivity and reduce side effects Strategies: Vary alkyl substituents Vary aryl substituents Extension Chain extensions / contractions Ring expansions / contractions Ring variation Isosteres Simplification Rigidification Vary Alkyl Substituents Rationale : •Alkyl group in lead compound may interact with hydrophobic region in binding site •Vary length and bulk of group to optimize interaction Patrick: Medicinal Chemistry Vary Alkyl Substituents: Increasing selectivity Adrenaline β2 > β1 β1 > β2 Noradrenaline Salbutamol (Ventolin) (Anti-asthmatic) selective β2-receptor agonist Propranolol (β-Blocker) blocks the action of epinephrine and norepinephrine Patrick: Medicinal Chemistry on both β1- and β2-adrenergic receptors Chapter | 2 Lead Discovery and Lead Modification 75 noted by Richet,[326] Overton,[327] and Meyer[328] between concentration of the drug that produces some standard bio- lipid solubility and biological activity. He first set out to logical effect, was related by Hansch et al.[333] to its lipophi- measure the lipophilicities of various compounds then to licity by the parabolic expression shown in Eqn (2.7). determine lipophilicities of substituents. But how should the lipophilicities be measured? The most relevant method MPH # åL(MPH 0) K′ (MPH 0) Ḱ (2.7) would be to determine their solubility in membranes or On the basis of Eqn (2.6), it is apparent that if a compound vesicles. However, as an organic chemist, Hansch prob- is more soluble in water than in 1-octanol, P < 1, and, there- ably realized that if he set a scale of lipophilicities based fore, log P is negative. -

WO 2014/170786 Al 23 October 2014 (23.10.2014) P O P C T
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date WO 2014/170786 Al 23 October 2014 (23.10.2014) P O P C T (51) International Patent Classification: 06419 (US). MAGUIRE, Bruce; 17 Waterhouse Lane, C07D 401/14 (2006.01) C07D 471/04 (2006.01) Chester, Connecticut 06412 (US). MCCLURE, Kim F.; C07D 413/14 (2006.01) C07D 487/04 (2006.01) 12 Willow Drive, Mystic, Connecticut 06355 (US). A61K 31/4725 (2006.01) A61P 7/00 (2006.01) PETERSEN, Donna N.; 107 Forsyth Road, Salem, Con A61K 31/519 (2006.01) necticut 06420 (US). PIOTROWSKI, David W.; 19 Beacon Hill Drive, Waterford, Connecticut 06385 (US). (21) International Application Number: PCT/IB20 14/060407 (74) Agent: OLSON, A. Dean; Pfizer Inc., Eastern Point Road MS8260-2141, Groton, CT 06340 (US). (22) International Filing Date: 3 April 2014 (03.04.2014) (81) Designated States (unless otherwise indicated, for every kind of national protection available): AE, AG, AL, AM, (25) Filing Language: English AO, AT, AU, AZ, BA, BB, BG, BH, BN, BR, BW, BY, (26) Publication Language: English BZ, CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (30) Priority Data: HN, HR, HU, ID, IL, IN, IR, IS, JP, KE, KG, KN, KP, KR, 61/812,864 17 April 2013 (17.04.2013) US KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, ME, 61/880,336 20 September 2013 (20.09.2013) US MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, NO, NZ, 61/898,667 1 November 2013 (01. -

The Uníversity of Manltoba in Partial Fulflìlment
RECEPTOIì.S FOR ERC,OT ALKAIOIDS IN GUINEA-PIG II,IIIJM AND DOG URINARY BLADDM A Thesis Presented to The UnÍversity of Manltoba In Partial FulflÌlment of the Requirements for the Degree of ' Master of Science by Maqsud Ahmad Cheema 1965 RECEPTORS FOR ERGOT ALKALOIDS IN GUINEA- PIG ILEUM AND DOG URINARY BLADDER by Maqsud Ahmad Cheerna ABSTRACT Ergot alkaloids cause most smooth muscles to contract. It ls known that this actlon is due to combination with CY-adrenergic receptors in smooth muscles where noradrenaline is excitatory, e. g. rabbit aorta and uterus, dog retractor penis, cat nictitating membrane. There are many smooth muscles, however, rtrhere noradrenaline causes relaxatlon and yet ergot alkalolds are excitatory. The receptors on which ergot alka- Ioids act to produce their effect on tissues with these propertles have therefore been studied. TVo in vitro preparations were used' strips of dog urinary btadder which relaxed in response to noradrenaÌlne, and seg- ments of guinea-pig lleum taken from a region in which both CY- and p- adrenergic receptors subserve inhibitl-on. The resutts indicate that the ergot alkaloids cause contraction of these preparations by actlng on 5- hydroxytrypt amine recePtors . Four ergot alkaloids were tested, dihydroergotamlne, ergot- amine, ergonovine, and methylergonovine" Ergotamine and dlhydroergota- mlne did not cause guinea-pig ileum to contract, but inhiblted the con- tractile actlon of 5-hydroxytryptamine. Ergonovine and methyl-ergonovine behaved ag would be expected of partiat agonlsts on 5-hydroxytryptamine receptors, in that the maximal contractlon they could induce was smaller than that of 5-hydroxytryptamlne, and that they antagonlzed the actlon e /G77 of 5-hydroxytryptamine.