Diptera Chironomidae
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

2020-11-12 Concord Station Final Agenda
Concord Station Community Development District Board of Supervisors’ Meeting November 12, 2020 District Office: 5844 Old Pasco Road, Suite 100 Wesley Chapel, Florida 33544 813.994.1615 www.concordstationcdd.com CONCORD STATION COMMUNITY DEVELOPMENT DISTRICT AGENDA Concord Station Clubhouse, located at 18636 Mentmore Boulevard, Land O’ Lakes, FL 34638 District Board of Supervisors Steven Christie Chairman Fred Berdeguez Vice Chairman Donna Matthias-Gorman Assistant Secretary Karen Hillis Assistant Secretary Jerica Ramirez Assistant Secretary District Manager Bryan Radcliff Rizzetta & Company, Inc. District Counsel John Vericker Straley Robin Vericker District Engineer Stephen Brletic JMT Engineering All Cellular phones and pagers must be turned off during the meeting. The Audience Comment portion of the agenda is where individuals may make comments on matters that concern the District. Individuals are limited to a total of three (3) minutes to make comments during this time. Pursuant to provisions of the Americans with Disabilities Act, any person requiring special accommodations to participate in this meeting/hearing/workshop is asked to advise the District Office at least forty-eight (48) hours before the meeting/hearing/workshop by contacting the District Manager at 813-933-5571. If you are hearing or speech impaired, please contact the Florida Relay Service by dialing 7-1- 1, or 1-800-955-8771 (TTY) 1-800-955-8770 (Voice), who can aid you in contacting the District Office. A person who decides to appeal any decision made at the meeting/hearing/workshop with respect to any matter considered at the meeting/hearing/workshop is advised that person will need a record of the proceedings and that accordingly, the person may need to ensure that a verbatim record of the proceedings is made including the testimony and evidence upon which the appeal is to be based. -

CHIRONOMUS Newsletter on Chironomidae Research
CHIRONOMUS Newsletter on Chironomidae Research No. 25 ISSN 0172-1941 (printed) 1891-5426 (online) November 2012 CONTENTS Editorial: Inventories - What are they good for? 3 Dr. William P. Coffman: Celebrating 50 years of research on Chironomidae 4 Dear Sepp! 9 Dr. Marta Margreiter-Kownacka 14 Current Research Sharma, S. et al. Chironomidae (Diptera) in the Himalayan Lakes - A study of sub- fossil assemblages in the sediments of two high altitude lakes from Nepal 15 Krosch, M. et al. Non-destructive DNA extraction from Chironomidae, including fragile pupal exuviae, extends analysable collections and enhances vouchering 22 Martin, J. Kiefferulus barbitarsis (Kieffer, 1911) and Kiefferulus tainanus (Kieffer, 1912) are distinct species 28 Short Communications An easy to make and simple designed rearing apparatus for Chironomidae 33 Some proposed emendations to larval morphology terminology 35 Chironomids in Quaternary permafrost deposits in the Siberian Arctic 39 New books, resources and announcements 43 Finnish Chironomidae 47 Chironomini indet. (Paratendipes?) from La Selva Biological Station, Costa Rica. Photo by Carlos de la Rosa. CHIRONOMUS Newsletter on Chironomidae Research Editors Torbjørn EKREM, Museum of Natural History and Archaeology, Norwegian University of Science and Technology, NO-7491 Trondheim, Norway Peter H. LANGTON, 16, Irish Society Court, Coleraine, Co. Londonderry, Northern Ireland BT52 1GX The CHIRONOMUS Newsletter on Chironomidae Research is devoted to all aspects of chironomid research and aims to be an updated news bulletin for the Chironomidae research community. The newsletter is published yearly in October/November, is open access, and can be downloaded free from this website: http:// www.ntnu.no/ojs/index.php/chironomus. Publisher is the Museum of Natural History and Archaeology at the Norwegian University of Science and Technology in Trondheim, Norway. -

Old Woman Creek National Estuarine Research Reserve Management Plan 2011-2016
Old Woman Creek National Estuarine Research Reserve Management Plan 2011-2016 April 1981 Revised, May 1982 2nd revision, April 1983 3rd revision, December 1999 4th revision, May 2011 Prepared for U.S. Department of Commerce Ohio Department of Natural Resources National Oceanic and Atmospheric Administration Division of Wildlife Office of Ocean and Coastal Resource Management 2045 Morse Road, Bldg. G Estuarine Reserves Division Columbus, Ohio 1305 East West Highway 43229-6693 Silver Spring, MD 20910 This management plan has been developed in accordance with NOAA regulations, including all provisions for public involvement. It is consistent with the congressional intent of Section 315 of the Coastal Zone Management Act of 1972, as amended, and the provisions of the Ohio Coastal Management Program. OWC NERR Management Plan, 2011 - 2016 Acknowledgements This management plan was prepared by the staff and Advisory Council of the Old Woman Creek National Estuarine Research Reserve (OWC NERR), in collaboration with the Ohio Department of Natural Resources-Division of Wildlife. Participants in the planning process included: Manager, Frank Lopez; Research Coordinator, Dr. David Klarer; Coastal Training Program Coordinator, Heather Elmer; Education Coordinator, Ann Keefe; Education Specialist Phoebe Van Zoest; and Office Assistant, Gloria Pasterak. Other Reserve staff including Dick Boyer and Marje Bernhardt contributed their expertise to numerous planning meetings. The Reserve is grateful for the input and recommendations provided by members of the Old Woman Creek NERR Advisory Council. The Reserve is appreciative of the review, guidance, and council of Division of Wildlife Executive Administrator Dave Scott and the mapping expertise of Keith Lott and the late Steve Barry. -
Diptera) of Finland
A peer-reviewed open-access journal ZooKeys 441: 37–46Checklist (2014) of the familes Chaoboridae, Dixidae, Thaumaleidae, Psychodidae... 37 doi: 10.3897/zookeys.441.7532 CHECKLIST www.zookeys.org Launched to accelerate biodiversity research Checklist of the familes Chaoboridae, Dixidae, Thaumaleidae, Psychodidae and Ptychopteridae (Diptera) of Finland Jukka Salmela1, Lauri Paasivirta2, Gunnar M. Kvifte3 1 Metsähallitus, Natural Heritage Services, P.O. Box 8016, FI-96101 Rovaniemi, Finland 2 Ruuhikosken- katu 17 B 5, 24240 Salo, Finland 3 Department of Limnology, University of Kassel, Heinrich-Plett-Str. 40, 34132 Kassel-Oberzwehren, Germany Corresponding author: Jukka Salmela ([email protected]) Academic editor: J. Kahanpää | Received 17 March 2014 | Accepted 22 May 2014 | Published 19 September 2014 http://zoobank.org/87CA3FF8-F041-48E7-8981-40A10BACC998 Citation: Salmela J, Paasivirta L, Kvifte GM (2014) Checklist of the familes Chaoboridae, Dixidae, Thaumaleidae, Psychodidae and Ptychopteridae (Diptera) of Finland. In: Kahanpää J, Salmela J (Eds) Checklist of the Diptera of Finland. ZooKeys 441: 37–46. doi: 10.3897/zookeys.441.7532 Abstract A checklist of the families Chaoboridae, Dixidae, Thaumaleidae, Psychodidae and Ptychopteridae (Diptera) recorded from Finland is given. Four species, Dixella dyari Garret, 1924 (Dixidae), Threticus tridactilis (Kincaid, 1899), Panimerus albifacies (Tonnoir, 1919) and P. przhiboroi Wagner, 2005 (Psychodidae) are reported for the first time from Finland. Keywords Finland, Diptera, species list, biodiversity, faunistics Introduction Psychodidae or moth flies are an intermediately diverse family of nematocerous flies, comprising over 3000 species world-wide (Pape et al. 2011). Its taxonomy is still very unstable, and multiple conflicting classifications exist (Duckhouse 1987, Vaillant 1990, Ježek and van Harten 2005). -

Chironomidae Hirschkopf
Literatur Chironomidae Gesäuse U.A. zur Bestimmung und Ermittlung der Autökologie herangezogene Literatur: Albu, P. (1972): Două specii de Chironomide noi pentru ştiinţă în masivul Retezat.- St. şi Cerc. Biol., Seria Zoologie, 24: 15-20. Andersen, T.; Mendes, H.F. (2002): Neotropical and Mexican Mesosmittia Brundin, with the description of four new species (Insecta, Diptera, Chironomidae).- Spixiana, 25(2): 141-155. Andersen, T.; Sæther, O.A. (1993): Lerheimia, a new genus of Orthocladiinae from Africa (Diptera: Chironomidae).- Spixiana, 16: 105-112. Andersen, T.; Sæther, O.A.; Mendes, H.F. (2010): Neotropical Allocladius Kieffer, 1913 and Pseudosmittia Edwards, 1932 (Diptera: Chironomidae).- Zootaxa, 2472: 1-77. Baranov, V.A. (2011): New and rare species of Orthocladiinae (Diptera, Chironomidae) from the Crimea, Ukraine.- Vestnik zoologii, 45(5): 405-410. Boggero, A.; Zaupa, S.; Rossaro, B. (2014): Pseudosmittia fabioi sp. n., a new species from Sardinia (Diptera: Chironomidae, Orthocladiinae).- Journal of Entomological and Acarological Research, [S.l.],46(1): 1-5. Brundin, L. (1947): Zur Kenntnis der schwedischen Chironomiden.- Arkiv för Zoologi, 39 A(3): 1- 95. Brundin, L. (1956): Zur Systematik der Orthocladiinae (Dipt. Chironomidae).- Rep. Inst. Freshwat. Drottningholm 37: 5-185. Casas, J.J.; Laville, H. (1990): Micropsectra seguyi, n. sp. du groupe attenuata Reiss (Diptera: Chironomidae) de la Sierra Nevada (Espagne).- Annls Soc. ent. Fr. (N.S.), 26(3): 421-425. Caspers, N. (1983): Chironomiden-Emergenz zweier Lunzer Bäche, 1972.- Arch. Hydrobiol. Suppl. 65: 484-549. Caspers, N. (1987): Chaetocladius insolitus sp. n. (Diptera: Chironomidae) from Lunz, Austria. In: Saether, O.A. (Ed.): A conspectus of contemporary studies in Chironomidae (Diptera). -
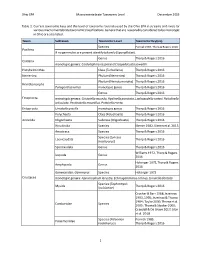
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Checklist of the Family Chironomidae (Diptera) of Finland
A peer-reviewed open-access journal ZooKeys 441: 63–90 (2014)Checklist of the family Chironomidae (Diptera) of Finland 63 doi: 10.3897/zookeys.441.7461 CHECKLIST www.zookeys.org Launched to accelerate biodiversity research Checklist of the family Chironomidae (Diptera) of Finland Lauri Paasivirta1 1 Ruuhikoskenkatu 17 B 5, FI-24240 Salo, Finland Corresponding author: Lauri Paasivirta ([email protected]) Academic editor: J. Kahanpää | Received 10 March 2014 | Accepted 26 August 2014 | Published 19 September 2014 http://zoobank.org/F3343ED1-AE2C-43B4-9BA1-029B5EC32763 Citation: Paasivirta L (2014) Checklist of the family Chironomidae (Diptera) of Finland. In: Kahanpää J, Salmela J (Eds) Checklist of the Diptera of Finland. ZooKeys 441: 63–90. doi: 10.3897/zookeys.441.7461 Abstract A checklist of the family Chironomidae (Diptera) recorded from Finland is presented. Keywords Finland, Chironomidae, species list, biodiversity, faunistics Introduction There are supposedly at least 15 000 species of chironomid midges in the world (Armitage et al. 1995, but see Pape et al. 2011) making it the largest family among the aquatic insects. The European chironomid fauna consists of 1262 species (Sæther and Spies 2013). In Finland, 780 species can be found, of which 37 are still undescribed (Paasivirta 2012). The species checklist written by B. Lindeberg on 23.10.1979 (Hackman 1980) included 409 chironomid species. Twenty of those species have been removed from the checklist due to various reasons. The total number of species increased in the 1980s to 570, mainly due to the identification work by me and J. Tuiskunen (Bergman and Jansson 1983, Tuiskunen and Lindeberg 1986). -

Table of Contents 2
Southwest Association of Freshwater Invertebrate Taxonomists (SAFIT) List of Freshwater Macroinvertebrate Taxa from California and Adjacent States including Standard Taxonomic Effort Levels 1 March 2011 Austin Brady Richards and D. Christopher Rogers Table of Contents 2 1.0 Introduction 4 1.1 Acknowledgments 5 2.0 Standard Taxonomic Effort 5 2.1 Rules for Developing a Standard Taxonomic Effort Document 5 2.2 Changes from the Previous Version 6 2.3 The SAFIT Standard Taxonomic List 6 3.0 Methods and Materials 7 3.1 Habitat information 7 3.2 Geographic Scope 7 3.3 Abbreviations used in the STE List 8 3.4 Life Stage Terminology 8 4.0 Rare, Threatened and Endangered Species 8 5.0 Literature Cited 9 Appendix I. The SAFIT Standard Taxonomic Effort List 10 Phylum Silicea 11 Phylum Cnidaria 12 Phylum Platyhelminthes 14 Phylum Nemertea 15 Phylum Nemata 16 Phylum Nematomorpha 17 Phylum Entoprocta 18 Phylum Ectoprocta 19 Phylum Mollusca 20 Phylum Annelida 32 Class Hirudinea Class Branchiobdella Class Polychaeta Class Oligochaeta Phylum Arthropoda Subphylum Chelicerata, Subclass Acari 35 Subphylum Crustacea 47 Subphylum Hexapoda Class Collembola 69 Class Insecta Order Ephemeroptera 71 Order Odonata 95 Order Plecoptera 112 Order Hemiptera 126 Order Megaloptera 139 Order Neuroptera 141 Order Trichoptera 143 Order Lepidoptera 165 2 Order Coleoptera 167 Order Diptera 219 3 1.0 Introduction The Southwest Association of Freshwater Invertebrate Taxonomists (SAFIT) is charged through its charter to develop standardized levels for the taxonomic identification of aquatic macroinvertebrates in support of bioassessment. This document defines the standard levels of taxonomic effort (STE) for bioassessment data compatible with the Surface Water Ambient Monitoring Program (SWAMP) bioassessment protocols (Ode, 2007) or similar procedures. -

(Chironomidae) in Heavy Metal Polluted Ponds
DNA-BARCODING OF NON-BITING MIDGE LARVAE (CHIRONOMIDAE) IN HEAVY METAL POLLUTED PONDS By Valeria Manfredi Canterbury Christ Church University Thesis submitted for the degree of MSc by Research 2019 Table of Contents List of Figures and Tables ................................................................................................................ 4 Abstract .............................................................................................................................................. 6 Introduction ....................................................................................................................................... 7 Water quality assessment ............................................................................................................ 7 Environmental Pollution ............................................................................................................. 8 Parys Mountain and pollution .................................................................................................. 10 Heavy metals in the sediments ................................................................................................. 12 Benthic invertebrates ................................................................................................................ 13 Chironomidae ........................................................................................................................... 14 Molecular ecology ................................................................................................................... -

Bibliograp Bibliography
BIBLIOGRAPHY 9.1 BIBLIOGRAPHY 9 Adam, J.I & O.A. Sæther. 1999. Revision of the records for the southern United States genus Nilothauma Kieffer, 1921 (Diptera: (Diptera: Chironomidae). J. Ga. Ent. Soc. Chironomidae). Ent. scand. Suppl. 56: 1- 15:69-73. 107. Beck, W.M., Jr. & E.C. Beck. 1966. Chironomidae Ali, A. 1991. Perspectives on management of pest- (Diptera) of Florida - I. Pentaneurini (Tany- iferous Chironomidae (Diptera), an emerg- podinae). Bull. Fla. St. Mus. Biol. Sci. ing global problem. J. Am. Mosq. Control 10:305-379. Assoc. 7: 260-281. Beck, W.M., Jr., & E.C. Beck. 1970. The immature Armitage, P., P.S. Cranston & L.C.V. Pinder (eds). stages of some Chironomini (Chiro- 1995. The Chironomidae. Biology and nomidae). Q.J. Fla. Acad. Sci. 33:29-42. ecology of non-biting midges. Chapman & Bilyj, B. 1984. Descriptions of two new species of Hall, London. 572 pp. Tanypodinae (Diptera: Chironomidae) from Ashe, P. 1983. A catalogue of chironomid genera Southern Indian Lake, Canada. Can. J. Fish. and subgenera of the world including syn- Aquat. Sci. 41: 659-671. onyms (Diptera: Chironomidae). Ent. Bilyj, B. 1985. New placement of Tanypus pallens scand. Suppl. 17: 1-68. Coquillett, 1902 nec Larsia pallens (Coq.) Barton, D.R., D.R. Oliver & M.E. Dillon. 1993. sensu Roback 1971 (Diptera: Chironomi- The first Nearctic record of Stackelbergina dae) and redescription of the holotype. Can. Shilova and Zelentsov (Diptera: Chironomi- Ent. 117: 39-42. dae): Taxonomic and ecological observations. Bilyj, B. 1988. A taxonomic review of Guttipelopia Aquatic Insects 15: 57-63. (Diptera: Chironomidae). -

DNA Barcoding
Full-time PhD studies of Ecology and Environmental Protection Piotr Gadawski Species diversity and origin of non-biting midges (Chironomidae) from a geologically young lake PhD Thesis and its old spring system Performed in Department of Invertebrate Zoology and Hydrobiology in Institute of Ecology and Environmental Protection Różnorodność gatunkowa i pochodzenie fauny Supervisor: ochotkowatych (Chironomidae) z geologicznie Prof. dr hab. Michał Grabowski młodego jeziora i starego systemu źródlisk Auxiliary supervisor: Dr. Matteo Montagna, Assoc. Prof. Łódź, 2020 Łódź, 2020 Table of contents Acknowledgements ..........................................................................................................3 Summary ...........................................................................................................................4 General introduction .........................................................................................................6 Skadar Lake ...................................................................................................................7 Chironomidae ..............................................................................................................10 Species concept and integrative taxonomy .................................................................12 DNA barcoding ...........................................................................................................14 Chapter I. First insight into the diversity and ecology of non-biting midges (Diptera: Chironomidae) -

“When We Went Onto the Land, the Elders Taught Me to Not Drink from a Lake If There Were No Insects, Because That Lake Was Dead.”
“When we went onto the land, the elders taught me to not drink from a lake if there were no insects, because that lake was dead.” - Inuvialuit community member, Personal communication, 2017 THE IMPACTS OF RECENT WILDFIRES ON STREAM WATER QUALITY AND MACROINVERTEBRATE ASSEMBLAGES IN SOUTHERN NORTHWEST TERRITORIES, CANADA By Caitlin Sarah Garner, B.Sc. (Hons.) A thesis submitted to the Faculty of Graduate Studies in partial fulfillment of the requirements of the degree of Master of Sustainability Brock University St. Catharines, ON ¬2017 Caitlin S. Garner Abstract High-latitude regions are currently undergoing rapid ecosystem change due to increasing temperatures and modified precipitation regimes. Since 2012, the Northwest Territories (Canada) has been experiencing severe drought and wildfire seasons. In 2014 alone, fires within the Northwest Territories consumed over 3.4 million hectares of forested land; 1.4 times larger than the national yearly average for Canada. Wildfire is one of the most important agents influencing age structure and composition of forest stands, as such, it is a critical factor in ecosystem dynamics. The impacts of wildfire on terrestrial systems garner more attention compared to aquatic habitats. This is especially true when considering aquatic ecosystems, specifically sub-arctic streams, where the impact of fires on stream ecology and chemistry are relatively understudied. Freshwater ecosystems, such as lakes and streams, are relied upon by northern communities for their cultural significance and economic and environmental goods and services they produce, including country foods. This study examines the impact of recent wildfire on freshwater streams within the North Slave, South Slave, and Dehcho regions of the Northwest Territories (Canada) through analysis of their water chemistry and benthic macroinvertebrate assemblages.