HIV and Messenger RNA (Mrna) Vaccine
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Xfect™ RNA Transfection Reagent Protocol-At-A-Glance
Xfect™ RNA Transfection Reagent Protocol-At-A-Glance I. Introduction A. Summary This Protocol-At-A-Glance is provided for transfection of cells with RNA using the Xfect RNA Transfection Reagent (Cat. No. 631450). It describes transfection of mammalian cells with RNA (mRNA, sgRNA, microRNA, or shRNA) in a 12-well plate format. For formats other than 12-well plates, see Tables I and II for appropriate reaction volumes. Transfections can be carried out entirely in the presence of serum. The protocol is divided into two sections: Section II describes transfection of cells with RNA only, without the cotransfection of DNA. Section III describes cotransfection of cells with both RNA and DNA. NOTE: When transfecting cells with mRNA, expression may be increased by using serum-free medium during transfection, since serum may contain RNases that can reduce the amount of full-length mRNA. B. General Considerations Storage & handling Store the Xfect RNA Transfection Polymer at –20°C. Do not thaw until ready to use. Once thawed, store at 4°C for up to 12 months. NOTE: The Xfect RNA Transfection Polymer is a milky suspension and should be vortexed briefly prior to use to ensure that it is fully resuspended. Thaw Xfect Reaction Buffer at room temperature just prior to use. Vortex after thawing. Once thawed, store Xfect Reaction Buffer at 4°C for up to 12 months. Xfect Polymer The protocol for cotransfection with DNA (Section III) requires the use of the Xfect Polymer (not included; sold as part of the DNA Xfect Transfection Reagent, Cat. Nos. 631317, 631318). -

Split Deoxyribozyme Probe for Efficient Detection of Highly Structured RNA Targets
University of Central Florida STARS Honors Undergraduate Theses UCF Theses and Dissertations 2018 Split Deoxyribozyme Probe For Efficient Detection of Highly Structured RNA Targets Sheila Raquel Solarez University of Central Florida Part of the Biochemistry Commons, and the Biology Commons Find similar works at: https://stars.library.ucf.edu/honorstheses University of Central Florida Libraries http://library.ucf.edu This Open Access is brought to you for free and open access by the UCF Theses and Dissertations at STARS. It has been accepted for inclusion in Honors Undergraduate Theses by an authorized administrator of STARS. For more information, please contact [email protected]. Recommended Citation Solarez, Sheila Raquel, "Split Deoxyribozyme Probe For Efficient Detection of Highly Structured RNA Targets" (2018). Honors Undergraduate Theses. 311. https://stars.library.ucf.edu/honorstheses/311 SPLIT DEOXYRIBOZYME PROBE FOR EFFICIENT DETECTION OF HIGHLY STRUCTURED RNA TARGETS By SHEILA SOLAREZ A thesis submitted in partial fulfillment of the requirements for the Honors in the Major Program in Biological Sciences in the College of Sciences and the Burnett Honors College at the University of Central Florida Orlando, Florida Spring Term, 2018 Thesis Chair: Yulia Gerasimova, PhD ABSTRACT Transfer RNAs (tRNAs) are known for their role as adaptors during translation of the genetic information and as regulators for gene expression; uncharged tRNAs regulate global gene expression in response to changes in amino acid pools in the cell. Aminoacylated tRNAs play a role in non-ribosomal peptide bond formation, post-translational protein labeling, modification of phospholipids in the cell membrane, and antibiotic biosynthesis. [1] tRNAs have a highly stable structure that can present a challenge for their detection using conventional techniques. -

Mrna Vaccine Era—Mechanisms, Drug Platform and Clinical Prospection
International Journal of Molecular Sciences Review mRNA Vaccine Era—Mechanisms, Drug Platform and Clinical Prospection 1, 1, 2 1,3, Shuqin Xu y, Kunpeng Yang y, Rose Li and Lu Zhang * 1 State Key Laboratory of Genetic Engineering, Institute of Genetics, School of Life Science, Fudan University, Shanghai 200438, China; [email protected] (S.X.); [email protected] (K.Y.) 2 M.B.B.S., School of Basic Medical Sciences, Peking University Health Science Center, Beijing 100191, China; [email protected] 3 Shanghai Engineering Research Center of Industrial Microorganisms, Shanghai 200438, China * Correspondence: [email protected]; Tel.: +86-13524278762 These authors contributed equally to this work. y Received: 30 July 2020; Accepted: 30 August 2020; Published: 9 September 2020 Abstract: Messenger ribonucleic acid (mRNA)-based drugs, notably mRNA vaccines, have been widely proven as a promising treatment strategy in immune therapeutics. The extraordinary advantages associated with mRNA vaccines, including their high efficacy, a relatively low severity of side effects, and low attainment costs, have enabled them to become prevalent in pre-clinical and clinical trials against various infectious diseases and cancers. Recent technological advancements have alleviated some issues that hinder mRNA vaccine development, such as low efficiency that exist in both gene translation and in vivo deliveries. mRNA immunogenicity can also be greatly adjusted as a result of upgraded technologies. In this review, we have summarized details regarding the optimization of mRNA vaccines, and the underlying biological mechanisms of this form of vaccines. Applications of mRNA vaccines in some infectious diseases and cancers are introduced. It also includes our prospections for mRNA vaccine applications in diseases caused by bacterial pathogens, such as tuberculosis. -

Expanding the Genetic Code Lei Wang and Peter G
Reviews P. G. Schultz and L. Wang Protein Science Expanding the Genetic Code Lei Wang and Peter G. Schultz* Keywords: amino acids · genetic code · protein chemistry Angewandte Chemie 34 2005 Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim DOI: 10.1002/anie.200460627 Angew. Chem. Int. Ed. 2005, 44,34–66 Angewandte Protein Science Chemie Although chemists can synthesize virtually any small organic molecule, our From the Contents ability to rationally manipulate the structures of proteins is quite limited, despite their involvement in virtually every life process. For most proteins, 1. Introduction 35 modifications are largely restricted to substitutions among the common 20 2. Chemical Approaches 35 amino acids. Herein we describe recent advances that make it possible to add new building blocks to the genetic codes of both prokaryotic and 3. In Vitro Biosynthetic eukaryotic organisms. Over 30 novel amino acids have been genetically Approaches to Protein encoded in response to unique triplet and quadruplet codons including Mutagenesis 39 fluorescent, photoreactive, and redox-active amino acids, glycosylated 4. In Vivo Protein amino acids, and amino acids with keto, azido, acetylenic, and heavy-atom- Mutagenesis 43 containing side chains. By removing the limitations imposed by the existing 20 amino acid code, it should be possible to generate proteins and perhaps 5. An Expanded Code 46 entire organisms with new or enhanced properties. 6. Outlook 61 1. Introduction The genetic codes of all known organisms specify the same functional roles to amino acid residues in proteins. Selectivity 20 amino acid building blocks. These building blocks contain a depends on the number and reactivity (dependent on both limited number of functional groups including carboxylic steric and electronic factors) of a particular amino acid side acids and amides, a thiol and thiol ether, alcohols, basic chain. -

Malaria Parasites Both Repress Host CXCL10 and Use It As a Cue for Growth Acceleration
ARTICLE https://doi.org/10.1038/s41467-021-24997-7 OPEN Malaria parasites both repress host CXCL10 and use it as a cue for growth acceleration Yifat Ofir-Birin 1, Hila Ben Ami Pilo1, Abel Cruz Camacho1, Ariel Rudik1, Anna Rivkin1, Or-Yam Revach1, Netta Nir1, Tal Block Tamin1, Paula Abou Karam1, Edo Kiper1, Yoav Peleg2, Reinat Nevo1, Aryeh Solomon3, Tal Havkin-Solomon1, Alicia Rojas1, Ron Rotkopf 4, Ziv Porat 5, Dror Avni6,7, Eli Schwartz6,7, Thomas Zillinger 8, Gunther Hartmann 8, Antonella Di Pizio 9, Neils Ben Quashie 10,11, Rivka Dikstein1, Motti Gerlic12, Ana Claudia Torrecilhas 13, Carmit Levy14, Esther N. M. Nolte-‘t Hoen15, ✉ Andrew G. Bowie 16 & Neta Regev-Rudzki 1 1234567890():,; Pathogens are thought to use host molecular cues to control when to initiate life-cycle transitions, but these signals are mostly unknown, particularly for the parasitic disease malaria caused by Plasmodium falciparum. The chemokine CXCL10 is present at high levels in fatal cases of cerebral malaria patients, but is reduced in patients who survive and do not have complications. Here we show a Pf ‘decision-sensing-system’ controlled by CXCL10 concentration. High CXCL10 expression prompts P. falciparum to initiate a survival strategy via growth acceleration. Remarkably, P. falciparum inhibits CXCL10 synthesis in monocytes by disrupting the association of host ribosomes with CXCL10 transcripts. The underlying inhi- bition cascade involves RNA cargo delivery into monocytes that triggers RIG-I, which leads to HUR1 binding to an AU-rich domain of the CXCL10 3’UTR. These data indicate that when the parasite can no longer keep CXCL10 at low levels, it can exploit the chemokine as a cue to shift tactics and escape. -
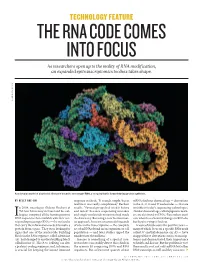
THE RNA CODE COMES INTO FOCUS As Researchers Open up to the Reality of RNA Modification, an Expanded Epitranscriptomics Toolbox Takes Shape
TECHNOLOGY FEATURE THE RNA CODE COMES INTO FOCUS As researchers open up to the reality of RNA modification, an expanded epitranscriptomics toolbox takes shape. LAGUNA DESIGN/SPL LAGUNA A molecular model of a bacterial ribosome bound to messenger RNA, a complex that is formed during protein synthesis. BY KELLY RAE CHI response in check. “It sounds simple, but in mRNAs harbour chemical tags — decorations real life it was really complicated,” Rechavi to the A, C, G and U nucleotides — that are n 2004, oncologist Gideon Rechavi at recalls. “Several groups had tried it before invisible to today’s sequencing technologies. Tel Aviv University in Israel and his col- and failed” because sequencing mistakes (Similar chemical tags, called epigenetic mark- leagues compared all the human genomic and single-nucleotide mutations had made ers, are also found on DNA.) Researchers aren’t IDNA sequences then available with their cor- the data noisy. But using a new bioinformat- sure what these chemical changes in RNA do, responding messenger RNAs — the molecules ics approach, his team uncovered thousands but they’re trying to find out. that carry the information needed to make a of sites in the transcriptome — the complete A wave of studies over the past five years — protein from a gene. They were looking for set of mRNAs found in an organism or cell many of which focus on a specific RNA mark signs that one of the nucleotide building population — and later studies upped the called N6-methyladenosine (m6A) — have blocks in the RNA sequence, called adenosine number into the millions1. -

Download Article (PDF)
DNA and RNA Nanotechnology 2015; 2: 42–52 Mini review Open Access Martin Panigaj*, Jakob Reiser Aptamer guided delivery of nucleic acid-based nanoparticles DOI 10.1515/rnan-2015-0005 Evolution of Ligands by Exponential enrichment) [4,5]. Received July 15, 2015; accepted October 3, 2015 Nucleic acid-based aptamers are especially well suited Abstract: Targeted delivery of bioactive compounds is a for the delivery of nucleic acid-based therapeutics. Any key part of successful therapies. In this context, nucleic nucleic acid with therapeutic potential can be linked acid and protein-based aptamers have been shown to to an aptamer sequence [6], resulting in a bivalent bind therapeutically relevant targets including receptors. molecule endowed with a targeting aptamer moiety and In the last decade, nucleic acid-based therapeutics a functional RNA/DNA moiety like a small interfering coupled to aptamers have emerged as a viable strategy for RNA (siRNA), a micro RNA (miRNA), a miRNA antagonist cell specific delivery. Additionally, recent developments (antimiR), deoxyribozymes (DNAzymes), etc. In addition in nucleic acid nanotechnology offer an abundance of to the specific binding, many aptamers upon receptor possibilities to rationally design aptamer targeted RNA recognition elicit antagonistic or agonistic responses that, or DNA nanoparticles involving combinatorial use of in combination with conjugated functional nucleic acids various intrinsic functionalities. Although a host of issues have the potential of synergism. Since the first report including stability, safety and intracellular trafficking describing an aptamer-siRNA delivery approach in 2006 remain to be addressed, aptamers as simple functional many functional RNAs and DNAs conjugated to aptamer chimeras or as parts of multifunctional self-assembled sequences have been tested in vitro and in vivo [7-9]. -

Mrna, Rrna and Trna Types of RNA: Mrna, Rrna and Trna
Types of RNA: mRNA, rRNA and tRNA Types of RNA: mRNA, rRNA and tRNA By Susha Cheriyedath, M.Sc. Reviewed by Michael Greenwood, M.Sc. RNA or ribonucleic acid is a polymer of nucleotides that is made up of a ribose sugar, a phosphate, and bases such as adenine, guanine, cytosine, and uracil. It plays a crucial role in gene expression by acting as the intermediate between the genetic information encoded by DNA and proteins. Designua | Shutterstock RNA has a structure very similar to that of DNA. The key difference in RNA structure is that the ribose sugar in RNA possesses a hydroxyl (OH) group that is absent in DNA. Types of RNA In both prokaryotes and eukaryotes, there are three main types of RNA – messenger RNA (mRNA), ribosomal RNA (rRNA), and transfer RNA (tRNA). These 3 types of RNA are discussed below. P Saved from URL: https://www.news-medical.net/life-sciences/-Types-of-RNA-mRNA-rRNA-and-tRNA.aspx 1/5 Types of RNA: mRNA, rRNA and tRNA Messenger RNA (mRNA) mRNA accounts for just 5% of the total RNA in the cell. mRNA is the most heterogeneous of the 3 types of RNA in terms of both base sequence and size. It carries complimentary genetic code copied, from DNA during transcription, in the form of triplets of nucleotides called codons. Each codon specifies a particular amino acid, though one amino acid may be coded for by many different codons. Although there are 64 possible codons or triplet bases in the genetic code, only 20 of them represent amino acids. -

Transcription Study Guide This Study Guide Is a Written Version of the Material You Have Seen Presented in the Transcription Unit
Transcription Study Guide This study guide is a written version of the material you have seen presented in the transcription unit. The cell’s DNA contains the instructions for carrying out the work of the cell. These instructions are used by the cell’s protein-making machinery to create proteins. If the cell’s DNA were directly read by the protein-making machinery, however, it could be damaged and the process would be slow and cumbersome. The cell avoids this problem by copying genetic information from its DNA into an intermediate called messenger RNA (mRNA). It is this mRNA that is read by the cell’s protein-making machinery. This process is called transcription. Components In this section you will be introduced to the components involved in the process of RNA synthesis, called transcription. This process requires an enzyme that uses many nucleotide bases to copy the instructions present in DNA into an intermediate messenger RNA molecule. RNA What is RNA? · Like DNA, RNA is a polymer made up of nucleotides. · Unlike DNA, which is composed of two strands of nucleotides twisted together, RNA is single-stranded. It can also sometimes fold into complex three-dimensional structures. · RNA contains the same nucleotides as DNA, with the substitution of uraciluridine (U) for thymidine (T). · RNA is chemically different from DNA so that the cell can easily tell the two apart. · In this animation, you will see one type of RNA, messenger RNA, being put together. · There are three types of RNA: mRNA, which you will read more about; tRNA, which is used in the translation process, and rRNA, which acts as a structural element in the ribosome (a translation component). -

Isolation of RNA with Properties of Messenger RNA from Cerebral Polyribosomes* Claire E
Proceedings of the National Academy of Sciences Vol. 67, No. 2, pp. 644-651, October 1970 Isolation of RNA with Properties of Messenger RNA from Cerebral Polyribosomes* Claire E. Zomzelyt, Sidney Roberts, and Susan Peache DEPARTMENT OF BIOLOGICAL CHEMISTRY, SCHOOL OF MEDICINE, AND THE BRAIN RESEARCH INSTI- TUTE, UNIVERSITY OF CALIFORNIA CENTER FOR THE HEALTH SCIENCES, LOS ANGELES, CALIFORNIA 90024 Communicated by H. W. Magoun, July 24, 1970 Abstract. RNA which dissociated from purified cerebral polyribosomes of adult rats in the presence of EDTA was isolated by fractionation in a dis- continuous sucrose gradient. The yield was 2% of the total polyribosomal RNA. The base composition resembled the complementary values for rat DNA and was very different from base compositions of ribosomal RNA and transfer RNA. This RNA fraction contained a large proportion of molecules which were rapidly labeled in vivo and hybridized to homologous DNA. The polyribosomal RNA preparation also exhibited high template activity in a cerebral cell-free system which had previously been stripped of the capacity to incorporate amino acids in the absence of added messenger RNA (mRNA). Sedimentation analysis revealed only two peaks, with coefficients of approximately 8 S and 16 S. The data indicate that RNA with the properties of mRNA can be selectively isolated from cerebral polyribosomes under mild conditions which avoid degradation. Numerous studies support the concept that alterations in RNA and protein synthesis are associated with information transfer and storage in the central nervous system. Activation of the synthesis of specific proteins is suggested by the reports that new RNA with altered base composition, presumably mRNA, is formed during the acquisition phase of learning. -

Why We Should Take a Second Look at RNA Technology
RNA THERAPEUTICS RNA Modulation: Gabriele Campi Why we should take a second look at RNA technology As a scientist first, but also as an investor, I have always hereditary ATTR amyloidosis with polyneuropathy, an believed in the potential of RNA molecules as therapeutics. autosomal dominant neurodegenerative disease. In 2018 While most current drugs aim to stop disease by modulating patisiran (Onpattro) was approved by the FDA. It is existing proteins, RNA molecules operate one step earlier. delivered to cells by lipid nanoparticles rather than GalNAc In the case of RNA interference (RNAi), they modulate the (N-Acetylgalactoseamine-siRNA conjugates). genes involved in different pathological processes. The RNA Since the end of 2018, investors have made a cautious molecules, small interfering RNA and microRNA, both return to the RNA therapy space with a targeted funding degrade messenger RNA (mRNA) and prevent it from being of existing RNA companies. In parallel, new companies are translated into proteins. entering the arena. An unexpected boost to the industry has I am the co-founder of the investment company AurorA come from the development and approval of the first mRNA Science, which recently led a Series B financing round for the vaccines to treat Covid-19 from Pfizer/BioNTech and Moderna. Dutch company InteRNA Technologies, which is developing I have always believed in the potency of RNA interference RNA therapeutics for the treatment of advanced solid and modulation techniques that allow a researcher to target tumours. This is an ambitious project, but with a potential specific messenger RNAs through the use of siRNAs, or for success. -

Efficient Sirna Delivery and Gene Silencing Using a Lipopolypeptide Hybrid Vector Mediated by a Caveolae-Mediated and Temperatur
Kasai et al. J Nanobiotechnol (2019) 17:11 https://doi.org/10.1186/s12951-019-0444-8 Journal of Nanobiotechnology RESEARCH Open Access Efcient siRNA delivery and gene silencing using a lipopolypeptide hybrid vector mediated by a caveolae‑mediated and temperature‑dependent endocytic pathway Hironori Kasai1, Kenji Inoue1, Kentaro Imamura1,2, Carlo Yuvienco3, Jin K. Montclare3,4,5,6 and Seiichi Yamano1* Abstract Background: We developed a non-viral vector, a combination of HIV-1 Tat peptide modifed with histidine and cysteine (mTat) and polyethylenimine, jetPEI (PEI), displaying the high efciency of plasmid DNA transfection with lit- tle toxicity. Since the highest efciency of INTERFERin (INT), a cationic amphiphilic lipid-based reagent, for small inter- fering RNA (siRNA) transfection among six commercial reagents was shown, we hypothesized that combining mTat/ PEI with INT would improve transfection efciency of siRNA delivery. To elucidate the efcacy of the hybrid vector for siRNA silencing, β-actin expression was measured after siRNA β-actin was transfected with mTat/PEI/INT or other vec- tors in HSC-3 human oral squamous carcinoma cells. Results: mTat/PEI/INT/siRNA produced signifcant improvement in transfection efciency with little cytotoxicity com- pared to other vectors and achieved 100% knockdown of β-actin expression compared to non-treated cells. The electric charge of mTat/PEI/INT/siRNA≈ was signifcantly higher than INT/siRNA. The particle size of mTat/PEI/INT/siRNA was signifcantly smaller than INT/siRNA. Filipin III and β-cyclodextrin, an inhibitor of caveolae-mediated endocyto- sis, signifcantly inhibited mTat/PEI/INT/siRNA transfection, while chlorpromazine, an inhibitor of clathrin-mediated endocytosis, did not inhibit mTat/PEI/INT/siRNA transfection.