Substitution of Amaranth As Dye in Wicking Test
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Application A603 Supporting Document 3 FOOD TECHNOLOGY
Application A603 Supporting Document 3 FOOD TECHNOLOGY REPORT Summary The Applicant, Golding Handcrafts, claims that there is a technical need to extend the use of the red food colouring erythrosine in Australia and New Zealand from preserved cherries to food colouring preparations used to colour icing and other cake decorations for the technological purpose of achieving colour enhancement. This has been substantiated with claims that food colourings preparations containing erythrosine possess superior colouring characteristics to alternative red food colourings including colour strength, longevity, lack of bleeding and quality of the finished product. Consequently this allow for a greater range of colouring effects to be achieved including wider ranges of pinks, lavenders, violets, royal blue and true black. The Applicant notes that commercial products are currently available for this purpose in the United States of America (USA) and seeks to import and sell these colourings preparations for cake icing and related products for use in Australia and New Zealand. The merit of using erythrosine is strongly supported by some companies, in particular, AmericolorTM Corporation and CK Products, two of the largest companies who use erythrosine in products for cake decorating purposes. They have advised the Applicant that the purpose of adding erythrosine to food is to achieve a precise visual effect and unique shades which are unattainable by using any other food colours. The colour hue and intensity is directly affected by the amounts of erythrosine added and the proposed amounts are consistent in achieving the intended result – to colour the food. Colour combinations are individually weighed and added to the mix for each single recipe so that a consistent final effect is obtained. -

Artificial Food Colours and Children Why We Want to Limit and Label Foods Containing the ‘Southampton Six’ Food Colours on the UK Market Post-Brexit
Artificial food colours and children Why we want to limit and label foods containing the ‘Southampton Six’ food colours on the UK market post-Brexit November 2020 FIRST STEPS NUTRITIONArtificial food coloursTRUST and children: page Artificial food colours and children: Why we want to limit and label foods containing the‘Southampton Six’ food colours on the UK market post-Brexit November 2020 Published by First Steps Nutrition Trust. A PDF of this resource is available on the First Steps Nutrition Trust website. www.firststepsnutrition.org The text of this resource, can be reproduced in other materials provided that the materials promote public health and make no profit, and an acknowledgement is made to First Steps Nutrition Trust. This resource is provided for information only and individual advice on diet and health should always be sought from appropriate health professionals. First Steps Nutrition Trust Studio 3.04 The Food Exchange New Covent Garden Market London SW8 5EL Registered charity number: 1146408 First Steps Nutrition Trust is a charity which provides evidence-based and independent information and support for good nutrition from pre-conception to five years of age. For more information, see our website: www.firststepsnutrition.org Acknowledgements This report was written by Rachael Wall and Dr Helen Crawley. We would like to thank Annie Seeley, Sarah Weston, Erik Millstone and Anna Rosier for their help and support with this report. Artificial food colours and children: page 1 Contents Page Executive summary 3 Recommendations -

Global Regulations of Food Colors Each Region Has Its Own Definitions of What Constitutes a Color Additive, with Related Use Requirements and Restrictions
Global Regulations of Food Colors Each region has its own definitions of what constitutes a color additive, with related use requirements and restrictions. Sue Ann McAvoy Sensient Colors LLC t is said, we eat with our eyes . Since antiq - food colors was soon recognized as a threat Iuity, humans have used the color of a to public health. Of concern was that some food to discern its quality. Color provides of the substances were known to be poi - a way to judge ripeness, perceive flavor and sonous and were often incorporated to hide assess quality of food. poor quality, add bulk to foods and to pass Ancient civilizations introduced color off imitation foods as real. into their foods. The ancient Egyptians col - On February 1, 1899, the executive com - ored their food yellow with saffron, and the mittee of the National Confectioners’ Asso - ancient Mayans used annatto to color their ciation published an official circular which Sue Ann McAvoy is food orange-red. Wealthy Romans ate bread was “to throw light upon the vexed question global regulatory scien - that had been whitened by adding alum to of what colors may be safely used in confec - tist for Sensient Food the flour. Color could be used to enhance tionery” as “there may at times be a doubt in Colors LLC. She has worked at Sensient the mind of the honest confectioner as to the physical appearance of the product. since 1979. However, if it made the food appear to be which colors, flavors, or ingredients he may of better quality than it was, that was con - safely use and which he may reject.” This list sidered a deceitful practice. -

Regulatory Information Sheet
Regulatory Information Sheet Approved Drug Colourants Listed by the European Union Colour Index Colour E Number Alternate Names Number Allura Red AC (a) E129 16035 FD&C Red #40 Aluminum*** E173 77000 -- Amaranth*** (a) E123 16185 Delisted FD&C Red #2 Annatto*** E160b 75120 Bixin, norbixin Anthocyanins (a) E163 -- -- Beetroot Red E162 -- Betanin Beta APO-8´-Carotenal E160e 40820 -- Brilliant Black BN (a) E151 28440 Black BN Brilliant Blue FCF (a) E133 42090 FD&C Blue #1 Brown HT (a) E155 20285 -- Calcium Carbonate E170 77220 -- Canthaxanthin* E161g 40850 -- Caramel,-Plain E150a -- -- Caramel,-Caustic Sulphite E150b -- -- Caramel,-Ammonia E150c -- -- Caramel, Sulphite Ammonia E150d -- -- Carmine (a) E120 75470 Carminic Acid, Cochineal Carmoisine (a) E122 14720 Azorubine Carotenes E160a 40800 / 75130 -- Chlorophylls/Chlorophyllins E140 75810 / 75815 -- Copper Complexes of E141 75815 -- Chlorophylls/Chlorophyllins(a) Curcumin (a) E100 75300 Turmeric Erythrosine*** (a) E127 45430 FD&C Red #3 Gold*** E175 77480 -- Green S (a) E142 44090 Acid Brilliant Green BS Indigotine (a) E132 73015 FD&C Blue #2, Indigo Carmine 77491 / 77492 / Iron Oxides & Hydroxides E172 Iron Oxide Red, Yellow, Black 77499 Litholrubine BK*** (a) E180 -- -- Lutein E161b -- -- Lycopene*** E160d 75125 -- Paprika Extract E160c -- Capsanthin, Capsorubin Patent Blue V (a) E131 42051 Acid Blue 3 Ponceau 4R (a) E124 16255 Cochineal Red A Page 1 of 2 Document Reference No.: GLO-10107, revision 2 Effective Date: September 2014 Reviewed Date: November 2017 This document is valid at the time of distribution. Distributed 24-Sep-2021 (UTC) E Colour Index Colour Alternate Names Number Number Quinoline Yellow** (a) E104 47005 China Yellow Riboflavins (a) E101 -- -- Silver*** E174 -- -- Sunset Yellow FCF (a) E110 15985 FD&C Yellow #6, Orange Yellow S Tartrazine (a) E102 19140 FD&C Yellow #5 Titanium Dioxide E171 77891 -- Vegetable Carbon E153 77268:1 Carbo Medicinalis Vegetalis The above list is derived from Part B, List of All Additives, from Annex II to Regulation (EC) No 1333/2008 on food additives. -

Role of Microorganisms in Biodegradation of Food Additive Azo Dyes: a Review
Vol. 19(11), pp.799-805, November, 2020 DOI: 10.5897/AJB2020.17250 Article Number: F63AA1865367 ISSN: 1684-5315 Copyright ©2020 Author(s) retain the copyright of this article African Journal of Biotechnology http://www.academicjournals.org/AJB Review Role of microorganisms in biodegradation of food additive Azo dyes: A review Fatimah Alshehrei Department of Biology, Jamum College University, Umm AlQura University, Makkah24382, Saudi Arabia. Received 22 September, 2020; Accepted 27 October, 2020 Food additives Azo dyes are synthetic compounds added to foods to impart color and improve their properties. Some azo dyes have been banned as food additives due to toxic, mutagenic, and carcinogenic side effects. Long exposure to foods containing azo dye leads to chronic toxicity. Some microorganisms are capable to degrade these dyes and convert them to aromatic amines. In human body, microbiota can play a vital role in biodegradation of azo dyes by producing azo reductase. Aromatic amines are toxic, water-soluble and well absorbed via human intestine. In the current study, the role of microorganisms in biodegradation of six dyes related to azo group was discussed. These dyes are: Tartrazine E102, Sunset Yellow E110, Ponceau E124, Azorubine E122, Amaranth E123, and Allura Red E129 which are classified as the most harmful food additive dyes. Key word: Food additive, azo dyes, microorganisms, azo reductase, aromatic amines. INTRODUCTION Food additives are synthetic compounds added to food In the USA and European countries, some azo dyes have for many proposes such as maintaining the product from been banned as food additives due to toxic, mutagenic, deterioration or improving its safety, freshness, taste, and carcinogenic side effects (Chung, 2000). -

Food Additives As Inhibitors of Intestinal Drug Transporter Oatp2b1
1 FOOD ADDITIVES AS INHIBITORS OF INTESTINAL DRUG TRANSPORTER OATP2B1 Alli Tikkanen1, Estelle Pierrot1, Feng Deng1,2, Virginia Barras Sánchez1, Marja Hagström1, Jan B. Koenderink3, Heidi Kidron1* 1Division of Pharmaceutical Biosciences, Faculty of Pharmacy, University of Helsinki, P.O. Box 56, 00014 Helsinki, Finland 2 Current address: Department of Clinical Pharmacology, Faculty of Medicine, University of Helsinki 3 Radboud University Medical Center, Department of Pharmacology and Toxicology, Nijmegen, The Netherlands *corresponding author Telephone: +358405666660 Email: [email protected] 2 FOR TABLE OF CONTENTS USE ONLY FOOD ADDITIVES AS INHIBITORS OF INTESTINAL DRUG TRANSPORTER OATP2B1 Alli Tikkanen, Estelle Pierrot, Feng Deng, Virginia Barras Sánchez, Marja Hagström, Jan B. Koenderink, Heidi Kidron 3 ABSTRACT Food additives are compounds that are added to food and beverage to improve taste, color, preservation or composition. Generally, food additives are considered safe for human use due to safety evaluation conducted by food safety authorities and high safety margins applied to permitted usage levels. However, the interaction potential of food additives with simultaneously administered medication has not received much attention. Even though many food additives are poorly absorbed into systemic circulation, high concentrations could exist in the intestinal lumen making intestinal drug transporters, such as the uptake transporter organic anion transporting polypeptide 2B1 (OATP2B1), a possible site of food additive–drug interaction. In the present work, we aimed to characterize the interaction of a selection of 25 food additives including colorants, preservatives and sweeteners with OATP2B1 in vitro. In HEK293 cells transiently overexpressing OATP2B1 or control, uptake of dibromofluorescein was studied with and without 50 µM food additive at pH 7.4. -

Biological Effects of Food Coloring in in Vivo and in Vitro Model Systems
foods Article Biological Effects of Food Coloring in In Vivo and In Vitro Model Systems Rocío Merinas-Amo, María Martínez-Jurado, Silvia Jurado-Güeto, Ángeles Alonso-Moraga and Tania Merinas-Amo * Department of Genetics, University of Córdoba, 14071 Córdoba, Spain; [email protected] (R.M.-A.); [email protected] (M.M.-J.); [email protected] (S.J.-G.); [email protected] (Á.A.-M.) * Correspondence: [email protected] Received: 18 April 2019; Accepted: 22 May 2019; Published: 24 May 2019 Abstract: (1) Background: The suitability of certain food colorings is nowadays in discussion because of the effects of these compounds on human health. For this reason, in the present work, the biological effects of six worldwide used food colorings (Riboflavin, Tartrazine, Carminic Acid, Erythrosine, Indigotine, and Brilliant Blue FCF) were analyzed using two model systems. (2) Methods: In vivo toxicity, antitoxicity, and longevity assays using the model organism Drosophila melanogaster and in vitro cytotoxicity, DNA fragmentation, and methylation status assays using HL-60 tumor human cell line were carried out. (3) Results: Our in vivo results showed safe effects in Drosophila for all the food coloring treatments, non-significant protective potential against an oxidative toxin, and different effects on the lifespan of flies. The in vitro results in HL-60 cells, showed that the tested food colorings increased tumor cell growth but did not induce any DNA damage or modifications in the DNA methylation status at their acceptable daily intake (ADI) concentrations. (4) Conclusions: From the in vivo and in vitro studies, these results would support the idea that a high chronic intake of food colorings throughout the entire life is not advisable. -

Analysis of Synthetic Dyes in Food Samples by Capillary Zone Electrophoresis
Analysis of synthetic dyes in food samples by capillary zone electrophoresis Application Note Foods and Flavors Authors Rainer Schuster, Angelika Gratzfeld-Hüsgen Agilent Technologies, Waldbronn, Germany Abstract Synthetic food dyes were separated by capillary zone electrophoresis using an alkaline phosphate buffer as background electrolyte. The precision had a relative standard deviation of less than 0.5 % for the run-to-run migration times and 2 % for the peak areas with buffer replenishment after each run. The detection limit for the individual dyes was about 1 ng using a 50-µm internal diameter Agilent extended path length capillary. Compounds were detected at different wavelengths— 215, 410, 520 and 598 nm—and the identities of the individual dyes were confirmed using peak-purity routines and a UV-visible spectral library. Introduction Azo Dyes Triphenylmethane Dyes Color is a vital constituent of foods OH NaO3S SO Na and probably the first characteristic 3 HO perceived by the human senses. NaO3S NN - Almost all foods— from raw agricultu- SO3 ral commodities to finished produc- ts—have an associated color. Further, NaO3S many tests have shown that color can Amaranth E123 (FD&C Red No. 2) + organoleptically dominate the flavor of (C2 H5 )N N (C 2 H5 ) a food. Colors have been added to Patent blue E131 foods since ancient times and include HO chlorophylls, carotinoids, flavonoids O O NaO3 S NN and anthocyans extracted from diffe- C C SO2 Na rent plants. Today synthetic dyes have C=C NaO3 S HN HN widely replaced natural colors. These SO Na dyes are used to supplement and 3 Sulfonated Indigo enhance the natural colors destroyed Sunset yellow FCF E110 (FD & C Yellow No. -
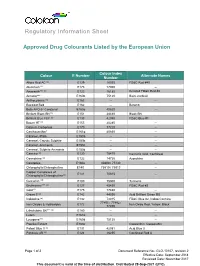
Regulatory Information Sheet
Regulatory Information Sheet Approved Drug Colourants Listed by the European Union Colour Index Colour E Number Alternate Names Number Allura Red AC (a) E129 16035 FD&C Red #40 Aluminum*** E173 77000 -- Amaranth*** (a) E123 16185 Delisted FD&C Red #2 Annatto*** E160b 75120 Bixin, norbixin Anthocyanins (a) E163 -- -- Beetroot Red E162 -- Betanin Beta APO-8´-Carotenal E160e 40820 -- Brilliant Black BN (a) E151 28440 Black BN Brilliant Blue FCF (a) E133 42090 FD&C Blue #1 Brown HT (a) E155 20285 -- Calcium Carbonate E170 77220 -- Canthaxanthin* E161g 40850 -- Caramel,-Plain E150a -- -- Caramel,-Caustic Sulphite E150b -- -- Caramel,-Ammonia E150c -- -- Caramel, Sulphite Ammonia E150d -- -- Carmine (a) E120 75470 Carminic Acid, Cochineal Carmoisine (a) E122 14720 Azorubine Carotenes E160a 40800 / 75130 -- Chlorophylls/Chlorophyllins E140 75810 / 75815 -- Copper Complexes of E141 75815 -- Chlorophylls/Chlorophyllins(a) Curcumin (a) E100 75300 Turmeric Erythrosine*** (a) E127 45430 FD&C Red #3 Gold*** E175 77480 -- Green S (a) E142 44090 Acid Brilliant Green BS Indigotine (a) E132 73015 FD&C Blue #2, Indigo Carmine 77491 / 77492 / Iron Oxides & Hydroxides E172 Iron Oxide Red, Yellow, Black 77499 Litholrubine BK*** (a) E180 -- -- Lutein E161b -- -- Lycopene*** E160d 75125 -- Paprika Extract E160c -- Capsanthin, Capsorubin Patent Blue V (a) E131 42051 Acid Blue 3 Ponceau 4R (a) E124 16255 Cochineal Red A Page 1 of 2 Document Reference No.: GLO-10107, revision 2 Effective Date: September 2014 Reviewed Date: November 2017 This document is valid at the time of distribution. Distributed 28-Sep-2021 (UTC) E Colour Index Colour Alternate Names Number Number Quinoline Yellow** (a) E104 47005 China Yellow Riboflavins (a) E101 -- -- Silver*** E174 -- -- Sunset Yellow FCF (a) E110 15985 FD&C Yellow #6, Orange Yellow S Tartrazine (a) E102 19140 FD&C Yellow #5 Titanium Dioxide E171 77891 -- Vegetable Carbon E153 77268:1 Carbo Medicinalis Vegetalis The above list is derived from Part B, List of All Additives, from Annex II to Regulation (EC) No 1333/2008 on food additives. -

Organic Colouring Agents in the Pharmaceutical Industry
DOI: 10.1515/fv-2017-0025 FOLIA VETERINARIA, 61, 3: 32—46, 2017 ORGANIC COLOURING AGENTS IN THE PHARMACEUTICAL INDUSTRY Šuleková, M.1, Smrčová, M.1, Hudák, A.1 Heželová, M.2, Fedorová, M.3 1Department of Chemistry, Biochemistry and Biophysics, Institute of Pharmaceutical Chemistry University of Veterinary Medicine and Pharmacy, Komenského 73, 041 81 Košice 2Faculty of Metallurgy, Institute of Recycling Technologies Technical University in Košice, Letná 9, 042 00 Košice 3Department of Pharmacy and Social Pharmacy University of Veterinary Medicine and Pharmacy, Komenského 73, 041 81 Košice Slovakia [email protected] ABSTRACT INTRODUCTION Food dyes are largely used in the process of manufac- In addition to the active ingredients, various additives turing pharmaceutical products. The aim of such a pro- are used in the manufacture of pharmaceuticals. This group cedure is not only to increase the attractiveness of prod- of compounds includes dyes. A colour additive is any dye, ucts, but also to help patients distinguish between phar- pigment, or other substance that imparts colour to food, maceuticals. Various dyes, especially organic colouring drink or any non-food applications including pharma- agents, may in some cases have a negative impact on the ceuticals. Moreover, a colour additive is also any chemical human body. They are incorporated into pharmaceuti- compound that reacts with another substance and causes cal products including tablets, hard gelatine capsules or the formation of a colour [22, 56]. The pharmaceutical in- soft gelatine capsules, lozenges, syrups, etc. This article dustry employs various inorganic and, especially, organic provides an overview of the most widely used colouring dyes for this purpose. -

Automation of AOAC 988.13 for the Identification of FD&C Color
May, 2012 Automation of AOAC 988.13 for the Identification of FD&C Color Additives in Foods using Solid Phase Extraction Application Note FB0112 Keywords GX-274 ASPEC™, AOAC, Spectrophotometer, Food and Beverage, Solid Phase Extraction, SPE, Color Additives, FD&C, FDA, TRILUTION ® LH, Liquid Handling Introduction A color additive is any dye, pigment or other substance which imparts color to a food, drug or cosmetic or to the human body (1). Color additives for food are commonly found in expected places, such as candies and powdered drink mixes, but also can be added to fruit skins to make them look more appealing. The addition of synthetic color additives is regulated closely by the FDA and is examined from the manufacturing of the pigment itself, through to its use and appropriate product labeling. Color additives have come under scrutiny recently because of their potential adverse physical and mental health effects that may be linked to ingestion, especially in children. AOAC method 988.13 qualitatively tests for the presence of eight synthetic color additives, one of which is now banned. These color additives are FD&C colors approved for use in food, drugs and cosmetics. Additives are extracted from the sample matrix using solid phase extraction (SPE), and then identified by spectrum analysis on a spectrophotometer. The color additives examined by the method – FD&C Red Nos. 3 and 40, Blue Nos. 1 and 2, Yellow Nos. 5 and 6, Green No. 3 and the now banned Red No. 2 – are listed in Table 1, along with the E Number used in the EU and UK, common chemical name and chemical structure. -

Tattoos and Permanent Make-Up State of Play and Trends in Tattoo Practices
Safety of tattoos and permanent make-up State of play and trends in tattoo practices Report on Work Package 2 Administrative Arrangement N. 2014 -33617 Analysis conducted on behalf of DG JUST Paola Picc inini, Laura Contor, Sazan Pakalin, Tim Raemaekers, Chiara Senaldi 2 0 1 5 LIMITED ReportDISTRIBUTION EUR 27528 EN This publication is a Technical report by the Joint Research Centre, the European Commission’s in-house science service. It aims to provide evidence-based scientific support to the European policy-making process. The scientific output expressed does not imply a policy position of the European Commission. Neither the European Commission nor any person acting on behalf of the Commission is responsible for t he use which might be made of this publication. JRC Science Hub https://ec.europa.eu/jrc JRC96808 EUR 27528 EN ISBN 978-92-79-52789-0 (PDF) ISSN 1831-9424 (online) doi:10.2788/924128 (online) © European Union, 2015 Reproduction is authorised provided the source is acknowledged. All images © European Union 2015, except: [cover page, Kolidzei, image #65391434], 2015. Source: [Fotolia.com] How to cite: Authors; title; EUR; doi (Paola Piccinini, Laura Contor, Sazan Pakalin, Tim Raemaekers, Chiara Senaldi; Safety of tattoos and permanent make-up. State of play and trends in tattoo practices; EUR 27528 EN ; 10.2788/924128) Safety of tattoos and permanent make-up State of play and trends in tattoo practices Table of contents Abstract 1 1. Introduction 3 2. Tattoos and Permanent Make-up Project 4 3. Methodology 5 3.1 Questionnaires 5 3.2 Web search 7 3.3 RAPEX notifications 7 3.4 National studies and surveys 8 3.5 Literature 8 3.6 Presentations 8 4.