Tests I Wish You'd Never Ordered
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Utility of the Digital Rectal Examination in the Emergency Department: a Review
The Journal of Emergency Medicine, Vol. 43, No. 6, pp. 1196–1204, 2012 Published by Elsevier Inc. Printed in the USA 0736-4679/$ - see front matter http://dx.doi.org/10.1016/j.jemermed.2012.06.015 Clinical Reviews UTILITY OF THE DIGITAL RECTAL EXAMINATION IN THE EMERGENCY DEPARTMENT: A REVIEW Chad Kessler, MD, MHPE*† and Stephen J. Bauer, MD† *Department of Emergency Medicine, Jesse Brown VA Medical Center and †University of Illinois-Chicago College of Medicine, Chicago, Illinois Reprint Address: Chad Kessler, MD, MHPE, Department of Emergency Medicine, Jesse Brown Veterans Hospital, 820 S Damen Ave., M/C 111, Chicago, IL 60612 , Abstract—Background: The digital rectal examination abdominal pain and acute appendicitis. Stool obtained by (DRE) has been reflexively performed to evaluate common DRE doesn’t seem to increase the false-positive rate of chief complaints in the Emergency Department without FOBTs, and the DRE correlated moderately well with anal knowing its true utility in diagnosis. Objective: Medical lit- manometric measurements in determining anal sphincter erature databases were searched for the most relevant arti- tone. Published by Elsevier Inc. cles pertaining to: the utility of the DRE in evaluating abdominal pain and acute appendicitis, the false-positive , Keywords—digital rectal; utility; review; Emergency rate of fecal occult blood tests (FOBT) from stool obtained Department; evidence-based medicine by DRE or spontaneous passage, and the correlation be- tween DRE and anal manometry in determining anal tone. Discussion: Sixteen articles met our inclusion criteria; there INTRODUCTION were two for abdominal pain, five for appendicitis, six for anal tone, and three for fecal occult blood. -

General Signs and Symptoms of Abdominal Diseases
General signs and symptoms of abdominal diseases Dr. Förhécz Zsolt Semmelweis University 3rd Department of Internal Medicine Faculty of Medicine, 3rd Year 2018/2019 1st Semester • For descriptive purposes, the abdomen is divided by imaginary lines crossing at the umbilicus, forming the right upper, right lower, left upper, and left lower quadrants. • Another system divides the abdomen into nine sections. Terms for three of them are commonly used: epigastric, umbilical, and hypogastric, or suprapubic Common or Concerning Symptoms • Indigestion or anorexia • Nausea, vomiting, or hematemesis • Abdominal pain • Dysphagia and/or odynophagia • Change in bowel function • Constipation or diarrhea • Jaundice “How is your appetite?” • Anorexia, nausea, vomiting in many gastrointestinal disorders; and – also in pregnancy, – diabetic ketoacidosis, – adrenal insufficiency, – hypercalcemia, – uremia, – liver disease, – emotional states, – adverse drug reactions – Induced but without nausea in anorexia/ bulimia. • Anorexia is a loss or lack of appetite. • Some patients may not actually vomit but raise esophageal or gastric contents in the absence of nausea or retching, called regurgitation. – in esophageal narrowing from stricture or cancer; also with incompetent gastroesophageal sphincter • Ask about any vomitus or regurgitated material and inspect it yourself if possible!!!! – What color is it? – What does the vomitus smell like? – How much has there been? – Ask specifically if it contains any blood and try to determine how much? • Fecal odor – in small bowel obstruction – or gastrocolic fistula • Gastric juice is clear or mucoid. Small amounts of yellowish or greenish bile are common and have no special significance. • Brownish or blackish vomitus with a “coffee- grounds” appearance suggests blood altered by gastric acid. -

The Differences Between ICD-9 and ICD-10
Preparing for the ICD-10 Code Set: Fact Sheet 2 October 1, 2015 Compliance Date Get the Facts to be Compliant Alert: The new ICD-10 compliance date is October 1, 2015. The Differences Between ICD-9 and ICD-10 This is the second fact sheet in a series and is focused on the differences between the ICD-9 and ICD-10 code sets. Collectively, the fact sheets will provide information, guidance, and checklists to assist you with understanding what you need to do to implement the ICD-10 code set. The ICD-10 code sets are not a simple update of the ICD-9 code set. The ICD-10 code sets have fundamental changes in structure and concepts that make them very different from ICD-9. Because of these differences, it is important to develop a preliminary understanding of the changes from ICD-9 to ICD-10. This basic understanding of the differences will then identify more detailed training that will be needed to appropriately use the ICD-10 code sets. In addition, seeing the differences between the code sets will raise awareness of the complexities of converting to the ICD-10 codes. Overall Comparisons of ICD-9 to ICD-10 Issues today with the ICD-9 diagnosis and procedure code sets are addressed in ICD-10. One concern today with ICD-9 is the lack of specificity of the information conveyed in the codes. For example, if a patient is seen for treatment of a burn on the right arm, the ICD-9 diagnosis code does not distinguish that the burn is on the right arm. -

HIGH-SENSITIVITY GUAIAC-BASED FECAL OCCULT BLOOD TEST (HSGFOBT) Anyone Can Get Colon Cancer
GET SCREENED: DETECT ANDDETECT PREVENT AND PREVENT COLON CANCER COLON CANCER TEST TYPE: HIGH-SENSITIVITY GUAIAC-BASED FECAL OCCULT BLOOD TEST (HSGFOBT) Anyone can get colon cancer. It can affect people of all racial and ethnic groups. Routine screening can help your health care provider find cancers earlier, when they are easier to treat. Screening may also prevent cancer, by finding and removing polyps or abnormal growths from the colon. COLON CANCER SCREENING There are different test options for screening. Talk with your provider to choose FACT SHEET the test that’s right for you. WHO? Adults who are at average risk for colon WHERE? You do this test at home. cancer may have an HSgFOBT. Talk with your health WHY? HSgFOBT detects signs of colon and rectal care provider about your risk and what age to begin cancer. It can also detect some polyps, which are screening. If you are at an increased risk of colon growths that could become cancer later. cancer, you may need screening early or this test may not be right for you. Discuss your medical and family HOW? You may have a restricted diet starting a medical history with your provider before choosing a few days before the test. You will get a kit from your test. Tell them if you have any of these risk factors: provider with instructions about how to take a sample A history of colon cancer or precancerous polyps of stool from three bowel movements in a row that A parent, sibling or child with colon cancer or you collect into a clean container. -

Unenhanced Areas Revealed by Contrast-Enhanced Abdominal Ultrasonography with Sonazoidtm Potentially Correspond to Colorectal Cancer
4012 EXPERIMENTAL AND THERAPEUTIC MEDICINE 12: 4012-4016, 2016 Unenhanced areas revealed by contrast-enhanced abdominal ultrasonography with SonazoidTM potentially correspond to colorectal cancer MINORU TOMIZAWA1, MIZUKI TOGASHI2, FUMINOBU SHINOZAKI3, RUMIKO HASEGAWA4, YOSHINORI SHIRAI4, MIDORI NORITAKE2, YUKIE MATSUOKA2, HIROAKI KAINUMA2, YASUJI IWASAKI2, KAZUNORI FUGO5, YASUFUMI MOTOYOSHI6, TAKAO SUGIYAMA7, SHIGENORI YAMAMOTO8, TAKASHI KISHIMOTO5 and NAOKI ISHIGE9 Departments of 1Gastroenterology; 2Clinical Laboratory; 3Radiology and 4Surgery, National Hospital Organization, Shimoshizu Hospital, Yotsukaido City, Chiba 284-0003; 5Department of Molecular Pathology, Chiba University Graduate School of Medicine, Chiba City, Chiba 260-8670; Departments of 6Neurology; 7Rheumatology; 8Pediatrics and 9Neurosurgery, National Hospital Organization, Shimoshizu Hospital, Yotsukaido, Yotsukaido City, Chiba 284-0003, Japan Received September 18, 2015; Accepted September 22, 2016 DOI: 10.3892/etm.2016.3868 Abstract. The present study investigated the potential utility of Introduction contrast-enhanced abdominal ultrasonography (CEUS), using SonazoidTM, in colorectal cancer (CRC). Three patients were Colorectal cancer (CRC) is commonly observed in clinical subjected to CEUS with SonazoidTM. Surgical specimens were settings (1). To improve the prognosis in patients with CRC, immunostained for CD31. Numbers of blood vessels positive prompt and accurate diagnosis is essential. Screening for CRC for CD31 were analyzed in each of five fields at x400 magnifi- is performed using fecal occult blood testing, and is diagnosed cation and averaged to determine blood vessel density. Blood with colonoscopy (2). vessel density was compared between non-tumorous and Abdominal ultrasound (US) is useful for the safe and tumorous areas. Prior to the administration of SonazoidTM, easy diagnosis of patients (3-6). During US screening of the CRC was illustrated as irregular-shaped wall thickening. -

Osteopathic Approach to the Spleen
Osteopathic approach to the spleen Luc Peeters and Grégoire Lason 1. Introduction the first 3 years to 4 - 6 times the birth size. The position therefore progressively becomes more lateral in place of The spleen is an organ that is all too often neglected in the original epigastric position. The spleen is found pos- the clinic, most likely because conditions of the spleen do tero-latero-superior from the stomach, its arterial supply is not tend to present a defined clinical picture. Furthermore, via the splenic artery and the left gastroepiploic artery it has long been thought that the spleen, like the tonsils, is (Figure 2). The venous drainage is via the splenic vein an organ that is superfluous in the adult. into the portal vein (Figure 2). The spleen is actually the largest lymphoid organ in the body and is implicated within the blood circulation. In the foetus it is an organ involved in haematogenesis while in the adult it produces lymphocytes. The spleen is for the blood what the lymph nodes are for the lymphatic system. The spleen also purifies and filters the blood by removing dead cells and foreign materials out of the circulation The function of red blood cell reserve is also essential for the maintenance of human activity. Osteopaths often identify splenic congestion under the influence of poor diaphragm function. Some of the symptoms that can be associated with dysfunction of the spleen are: Figure 2 – Position and vascularisation of the spleen Anaemia in children Disorders of blood development Gingivitis, painful and bleeding gums Swollen, painful tongue, dysphagia and glossitis Fatigue, hyperirritability and restlessness due to the anaemia Vertigo and tinnitus Frequent colds and infections due to decreased resis- tance Thrombocytosis Tension headaches The spleen is also considered an important organ by the osteopath as it plays a role in the immunity, the reaction of the circulation and oxygen transport during effort as well as in regulation of the blood pressure. -
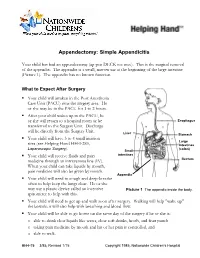
Appendectomy: Simple Appendicitis
Appendectomy: Simple Appendicitis Your child has had an appendectomy (ap pen DECK toe mee). This is the surgical removal of the appendix. The appendix is a small, narrow sac at the beginning of the large intestine (Picture 1). The appendix has no known function. What to Expect After Surgery . Your child will awaken in the Post Anesthesia Care Unit (PACU) near the surgery area. He or she may be in the PACU for 1 to 2 hours. After your child wakes up in the PACU, he or she will return to a hospital room or be Esophagus transferred to the Surgery Unit. Discharge will be directly from the Surgery Unit. Liver Stomach . Your child will have 3 to 4 small incision Large sites (see Helping Hand HH-I-283, Intestines Laparoscopic Surgery (colon) ). Small . Your child will receive fluids and pain intestines medicine through an intravenous line (IV). Rectum When your child can take liquids by mouth, pain medicine will also be given by mouth. Appendix . Your child will need to cough and deep-breathe often to help keep the lungs clear. He or she may use a plastic device called an incentive Picture 1 The appendix inside the body. spirometer to help with this. Your child will need to get up and walk soon after surgery. Walking will help "wake up" the bowels; it will also help with breathing and blood flow. Your child will be able to go home on the same day of the surgery if he or she is: o able to drink clear liquids like water, clear soft drinks, broth, and fruit punch o taking pain medicine by mouth and his or her pain is controlled, and o able to walk. -

17 Nutrition for Patients with Upper Gastrointestinal Disorders 403
84542_ch17.qxd 7/16/09 6:35 PM Page 402 Nutrition for Patients with Upper 17 Gastrointestinal Disorders TRUE FALSE 1 People who have nausea should avoid liquids with meals. 2 Thin liquids, such as clear juices and clear broths, are usually the easiest items to swallow for patients with dysphagia. 3 All patients with dysphagia are given solid foods in pureed form. 4 In people with GERD, the severity of the pain reflects the extent of esophageal damage. 5 High-fat meals may trigger symptoms of GERD. 6 People with esophagitis may benefit from avoiding spicy or acidic foods. 7 Alcohol stimulates gastric acid secretion. 8 A bland diet promotes healing of peptic ulcers. 9 People with dumping syndrome should avoid sweets and sugars. 10 Pernicious anemia is a potential complication of gastric surgery. UPON COMPLETION OF THIS CHAPTER, YOU WILL BE ABLE TO ● Give examples of ways to promote eating in people with anorexia. ● Describe nutrition interventions that may help maximize intake in people who have nausea. ● Compare the three levels of solid food textures included in the National Dysphagia Diet. ● Compare the four liquid consistencies included in the National Dysphagia Diet. ● Plan a menu appropriate for someone with GERD. ● Teach a patient about role of nutrition therapy in the treatment of peptic ulcer disease. ● Give examples of nutrition therapy recommendations for people experiencing dumping syndrome. utrition therapy is used in the treatment of many digestive system disorders. For many disorders, diet merely plays a supportive role in alleviating symptoms rather than alter- ing the course of the disease. -

Dysphagia - Pathophysiology of Swallowing Dysfunction, Symptoms, Diagnosis and Treatment
ISSN: 2572-4193 Philipsen. J Otolaryngol Rhinol 2019, 5:063 DOI: 10.23937/2572-4193.1510063 Volume 5 | Issue 3 Journal of Open Access Otolaryngology and Rhinology REVIEW ARTICLE Dysphagia - Pathophysiology of Swallowing Dysfunction, Symptoms, Diagnosis and Treatment * Bahareh Bakhshaie Philipsen Check for updates Department of Otorhinolaryngology-Head and Neck Surgery, Odense University Hospital, Denmark *Corresponding author: Dr. Bahareh Bakhshaie Philipsen, Department of Otorhinolaryngology-Head and Neck Surgery, Odense University Hospital, Sdr. Boulevard 29, 5000 Odense C, Denmark, Tel: +45 31329298, Fax: +45 66192615 the vocal folds adduct to prevent aspiration. The esoph- Abstract ageal phase is completely involuntary and consists of Difficulty swallowing is called dysphagia. There is a wide peristaltic waves [2]. range of potential causes of dysphagia. Because there are many reasons why dysphagia can occur, treatment Dysphagia is classified into the following major depends on the underlying cause. Thorough examination types: is important, and implementation of a treatment strategy should be based on evaluation by a multidisciplinary team. 1. Oropharyngeal dysphagia In this article, we will describe the mechanism of swallowing, the pathophysiology of swallowing dysfunction and different 2. Esophageal dysphagia causes of dysphagia, along with signs and symptoms asso- 3. Complex neuromuscular disorders ciated with dysphagia, diagnosis, and potential treatments. 4. Functional dysphagia Keywords Pathophysiology Dysphagia, Deglutition, Deglutition disorders, FEES, Video- fluoroscopy Swallowing is a complex process and many distur- bances in oropharyngeal and esophageal physiology including neurologic deficits, obstruction, fibrosis, struc- Introduction tural damage or congenital and developmental condi- Dysphagia is derived from the Greek phagein, means tions can result in dysphagia. Breathing difficulties can “to eat” [1]. -

Abdominal Pain - Gastroesophageal Reflux Disease
ACS/ASE Medical Student Core Curriculum Abdominal Pain - Gastroesophageal Reflux Disease ABDOMINAL PAIN - GASTROESOPHAGEAL REFLUX DISEASE Epidemiology and Pathophysiology Gastroesophageal reflux disease (GERD) is one of the most commonly encountered benign foregut disorders. Approximately 20-40% of adults in the United States experience chronic GERD symptoms, and these rates are rising rapidly. GERD is the most common gastrointestinal-related disorder that is managed in outpatient primary care clinics. GERD is defined as a condition which develops when stomach contents reflux into the esophagus causing bothersome symptoms and/or complications. Mechanical failure of the antireflux mechanism is considered the cause of GERD. Mechanical failure can be secondary to functional defects of the lower esophageal sphincter or anatomic defects that result from a hiatal or paraesophageal hernia. These defects can include widening of the diaphragmatic hiatus, disturbance of the angle of His, loss of the gastroesophageal flap valve, displacement of lower esophageal sphincter into the chest, and/or failure of the phrenoesophageal membrane. Symptoms, however, can be accentuated by a variety of factors including dietary habits, eating behaviors, obesity, pregnancy, medications, delayed gastric emptying, altered esophageal mucosal resistance, and/or impaired esophageal clearance. Signs and Symptoms Typical GERD symptoms include heartburn, regurgitation, dysphagia, excessive eructation, and epigastric pain. Patients can also present with extra-esophageal symptoms including cough, hoarse voice, sore throat, and/or globus. GERD can present with a wide spectrum of disease severity ranging from mild, intermittent symptoms to severe, daily symptoms with associated esophageal and/or airway damage. For example, severe GERD can contribute to shortness of breath, worsening asthma, and/or recurrent aspiration pneumonia. -

Acute Abdomen
Acute abdomen: Shaking down the Acute abdominal pain can be difficult to diagnose, requiring astute assessment skills and knowledge of abdominal anatomy 2.3 ANCC to discover its cause. We show you how to quickly and accurately CONTACT HOURS uncover the clues so your patient can get the help he needs. By Amy Wisniewski, BSN, RN, CCM Lehigh Valley Home Care • Allentown, Pa. The author has disclosed that she has no significant relationships with or financial interest in any commercial companies that pertain to this educational activity. NIE0110_124_CEAbdomen.qxd:Deepak 26/11/09 9:38 AM Page 43 suspects Determining the cause of acute abdominal rapidly, indicating a life-threatening process, pain is often complex due to the many or- so fast and accurate assessment is essential. gans in the abdomen and the fact that pain In this article, I’ll describe how to assess a may be nonspecific. Acute abdomen is a patient with acute abdominal pain and inter- general diagnosis, typically referring to se- vene appropriately. vere abdominal pain that occurs suddenly over a short period (usually no longer than What a pain! 7 days) and often requires surgical interven- Acute abdominal pain is one of the top tion. Symptoms may be severe and progress three symptoms of patients presenting in www.NursingMadeIncrediblyEasy.com January/February 2010 Nursing made Incredibly Easy! 43 NIE0110_124_CEAbdomen.qxd:Deepak 26/11/09 9:38 AM Page 44 the ED. Reasons for acute abdominal pain Visceral pain can be divided into three Your patient’s fall into six broad categories: subtypes: age may give • inflammatory—may be a bacterial cause, • tension pain. -

Flexible Sigmoidoscopy in Asymptomatic Patients with Negative Fecal Occult Blood Tests Joy Garrison Cauffman, Phd, Jimmy H
Flexible Sigmoidoscopy in Asymptomatic Patients with Negative Fecal Occult Blood Tests Joy Garrison Cauffman, PhD, Jimmy H. Hara, MD, Irving M. Rasgon, MD, and Virginia A. Clark, PhD Los Angeles, California Background. Although the American Cancer Society and tients with lesions were referred for colonoscopy; addi others haw established guidelines for colorectal cancer tional lesions were found in 14%. A total of 62 lesions screening, questions of who and how to screen still exist. were discovered, including tubular adenomas, villous Methods. A 60-crn flexible sigmoidoscopy was per adenomas, tubular villous adenomas (23 of the adeno formed on 1000 asymptomatic patients, 45 years of mas with atypia), and one adenocarcinoma. The high age or older, with negative fecal occult blood tests, est percentage of lesions discovered were in the sig who presented for routine physical examinations. Pa moid colon and the second highest percentage were in tients with clinically significant lesions were referred for the ascending colon. colonoscopy. The proportion of lesions that would not Conclusions. The 60-cm flexible sigmoidoscope was have been found if the 24-cm rigid or the 30-cm flexi able to detect more lesions than either the 24-cm or ble sigmoidoscope had been used was identified. 30-cm sigmoidoscope when used in asymptomatic pa Results. Using the 60-cm flexible sigmoidoscope, le tients, 45 years of age and over, with negative fecal oc sions were found in 3.6% of the patients. Eighty per cult blood tests. When significant lesions are discovered cent of the significant lesions were beyond the reach of by sigmoidoscopy, colonoscopy should be performed.