Lecture 4 - Citric Acid Cycle
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Cross-Talk Between Methylation and Phosphorylation in Lymphoid-Specific Helicase Drives Cancer Stem-Like Properties
Signal Transduction and Targeted Therapy www.nature.com/sigtrans ARTICLE OPEN The cross-talk between methylation and phosphorylation in lymphoid-specific helicase drives cancer stem-like properties Na Liu1,2,3, Rui Yang1,2, Ying Shi1,2, Ling Chen1,2, Yating Liu1,2, Zuli Wang1,2, Shouping Liu1,2, Lianlian Ouyang4, Haiyan Wang1,2, Weiwei Lai1,2, Chao Mao1,2, Min Wang1,2, Yan Cheng5, Shuang Liu4, Xiang Wang6, Hu Zhou7, Ya Cao1,2, Desheng Xiao1 and Yongguang Tao1,2,6 Posttranslational modifications (PTMs) of proteins, including chromatin modifiers, play crucial roles in the dynamic alteration of various protein properties and functions including stem-cell properties. However, the roles of Lymphoid-specific helicase (LSH), a DNA methylation modifier, in modulating stem-like properties in cancer are still not clearly clarified. Therefore, exploring PTMs modulation of LSH activity will be of great significance to further understand the function and activity of LSH. Here, we demonstrate that LSH is capable to undergo PTMs, including methylation and phosphorylation. The arginine methyltransferase PRMT5 can methylate LSH at R309 residue, meanwhile, LSH could as well be phosphorylated by MAPK1 kinase at S503 residue. We further show that the accumulation of phosphorylation of LSH at S503 site exhibits downregulation of LSH methylation at R309 residue, which eventually promoting stem-like properties in lung cancer. Whereas, phosphorylation-deficient LSH S503A mutant promotes the accumulation of LSH methylation at R309 residue and attenuates stem-like properties, indicating the critical roles of LSH PTMs in modulating stem-like properties. Thus, our study highlights the importance of the crosstalk between LSH PTMs in determining its activity and function in lung cancer stem-cell maintenance. -

82870547.Pdf
ORIGINAL RESEARCH published: 06 March 2017 doi: 10.3389/fpls.2017.00291 Integrated Transcriptome and Metabolic Analyses Reveals Novel Insights into Free Amino Acid Metabolism in Huangjinya Tea Cultivar Qunfeng Zhang 1, 2, Meiya Liu 1, 2 and Jianyun Ruan 1, 2* 1 Tea Research Institute, Chinese Academy of Agricultural Sciences, Hangzhou, China, 2 Key Laboratory for Plant Biology and Resource Application of Tea, The Ministry of Agriculture, Hangzhou, China The chlorotic tea variety Huangjinya, a natural mutant, contains enhanced levels of free amino acids in its leaves, which improves the drinking quality of its brewed tea. Consequently, this chlorotic mutant has a higher economic value than the non-chlorotic Edited by: varieties. However, the molecular mechanisms behind the increased levels of free amino Basil J. Nikolau, Iowa State University, USA acids in this mutant are mostly unknown, as are the possible effects of this mutation Reviewed by: on the overall metabolome and biosynthetic pathways in tea leaves. To gain further John A. Morgan, insight into the effects of chlorosis on the global metabolome and biosynthetic pathways Purdue University, USA Vinay Kumar, in this mutant, Huangjinya plants were grown under normal and reduced sunlight, Central University of Punjab, India resulting in chlorotic and non-chlorotic leaves, respectively; their leaves were analyzed *Correspondence: using transcriptomics as well as targeted and untargeted metabolomics. Approximately Jianyun Ruan 5,000 genes (8.5% of the total analyzed) and ca. 300 metabolites (14.5% of the total [email protected] detected) were significantly differentially regulated, thus indicating the occurrence of Specialty section: marked effects of light on the biosynthetic pathways in this mutant plant. -

Effect of Citric Acid Cycle Genetic Variants and Their Interactions With
cancers Article Effect of Citric Acid Cycle Genetic Variants and Their Interactions with Obesity, Physical Activity and Energy Intake on the Risk of Colorectal Cancer: Results from a Nested Case-Control Study in the UK Biobank Sooyoung Cho 1 , Nan Song 2,3 , Ji-Yeob Choi 2,4,5 and Aesun Shin 1,2,* 1 Department of Preventive Medicine, Seoul National University College of Medicine, Seoul 03080, Korea; [email protected] 2 Cancer Research Institute, Seoul National University, Seoul 03080, Korea; [email protected] (N.S.); [email protected] (J.-Y.C.) 3 Department of Epidemiology and Cancer Control, St. Jude Children’s Research Hospital, Memphis, TN 38105, USA 4 Department of Biomedical Sciences, Graduate School of Seoul National University, Seoul 03080, Korea 5 Medical Research Center, Institute of Health Policy and Management, Seoul National University, Seoul 03080, Korea * Correspondence: [email protected]; Tel.: +82-2-740-8331; Fax: +82-2-747-4830 Received: 18 August 2020; Accepted: 9 October 2020; Published: 12 October 2020 Simple Summary: The citric acid cycle has a central role in the cellular energy metabolism and biosynthesis of macromolecules in the mitochondrial matrix. We identified the single nucleotide polymorphisms (SNPs) of the citrate acid cycle with colorectal cancer susceptibility in UK population. Furthermore, we found the significant interaction of SNPs in the citric acid cycle with the contributors to energy balance and SNP-SNP interactions. Our findings provide clues to the etiology in cancer development related to energy metabolism and evidence on identification of the population at high risk of colorectal cancer. -

Nutrition and Metabolism
NUTRITION AND METABOLISM Metabolism - the sum of the chemical changes that occur in the cell and involve the breakdown (catabolism) and synthesis (anabolism) of stored energy sources. Basal Metabolic Rate is dened as the rate of energy production by the body measured under a dened set of conditions which is usually at rest (physical and mental), room temperature, 12 hours after a meal. The result is produced as a percentage of a standard value which is derived from studies of normal healthy people. Measurement of the metabolic rate takes place using a method called calorimetry. This may be done directly by measuring the amount of heat produced by the body in an Atwater chamber, the metabolic rate is the amount of heat produced per hour. More commonly the metabolic rate is determined indirectly by putting people on a closed circuit breathing system, with CO2 removed by a soda lime scrubber and the rate of oxygen consumption measured by change in volume. Oxygen consumption is proportional to the metabolic rate because most of the energy in the body is derived from oxidative phosphorylation, which uses a set amount of oxygen to produce a set amount of energy. For every litre of oxygen consumed the body produces (uses) 4.82 kcals of energy. If the oxygen consumption is 250ml/min (15L/hr) then the metabolic rate is 72.3 kcals/hr. This is often further rened by dividing the gure by the body surface area which for a 70kg male is 1.73m2. This gives an average BMR of approximately 40 kcal/m2/hr. -

ATP-Citrate Lyase Has an Essential Role in Cytosolic Acetyl-Coa Production in Arabidopsis Beth Leann Fatland Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 2002 ATP-citrate lyase has an essential role in cytosolic acetyl-CoA production in Arabidopsis Beth LeAnn Fatland Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Molecular Biology Commons, and the Plant Sciences Commons Recommended Citation Fatland, Beth LeAnn, "ATP-citrate lyase has an essential role in cytosolic acetyl-CoA production in Arabidopsis " (2002). Retrospective Theses and Dissertations. 1218. https://lib.dr.iastate.edu/rtd/1218 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. ATP-citrate lyase has an essential role in cytosolic acetyl-CoA production in Arabidopsis by Beth LeAnn Fatland A dissertation submitted to the graduate faculty in partial fulfillment of the requirements for the degree of DOCTOR OF PHILOSOPHY Major: Plant Physiology Program of Study Committee: Eve Syrkin Wurtele (Major Professor) James Colbert Harry Homer Basil Nikolau Martin Spalding Iowa State University Ames, Iowa 2002 UMI Number: 3158393 INFORMATION TO USERS The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleed-through, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted. -

Mutation of the Fumarase Gene in Two Siblings with Progressive Encephalopathy and Fumarase Deficiency T
Mutation of the Fumarase Gene in Two Siblings with Progressive Encephalopathy and Fumarase Deficiency T. Bourgeron,* D. Chretien,* J. Poggi-Bach, S. Doonan,' D. Rabier,* P. Letouze,I A. Munnich,* A. R6tig,* P. Landneu,* and P. Rustin* *Unite de Recherches sur les Handicaps Genetiques de l'Enfant, INSERM U393, Departement de Pediatrie et Departement de Biochimie, H6pital des Enfants-Malades, 149, rue de Sevres, 75743 Paris Cedex 15, France; tDepartement de Pediatrie, Service de Neurologie et Laboratoire de Biochimie, Hopital du Kremlin-Bicetre, France; IFaculty ofScience, University ofEast-London, UK; and IService de Pediatrie, Hopital de Dreux, France Abstract chondrial enzyme (7). Human tissue fumarase is almost We report an inborn error of the tricarboxylic acid cycle, fu- equally distributed between the mitochondria, where the en- marase deficiency, in two siblings born to first cousin parents. zyme catalyzes the reversible hydration of fumarate to malate They presented with progressive encephalopathy, dystonia, as a part ofthe tricarboxylic acid cycle, and the cytosol, where it leucopenia, and neutropenia. Elevation oflactate in the cerebro- is involved in the metabolism of the fumarate released by the spinal fluid and high fumarate excretion in the urine led us to urea cycle. The two isoenzymes have quite homologous struc- investigate the activities of the respiratory chain and of the tures. In rat liver, they differ only by the acetylation of the Krebs cycle, and to finally identify fumarase deficiency in these NH2-terminal amino acid of the cytosolic form (8). In all spe- two children. The deficiency was profound and present in all cies investigated so far, the two isoenzymes have been found to tissues investigated, affecting the cytosolic and the mitochon- be encoded by a single gene (9,10). -

Phosphorylation of Chicken Protein Tyrosine Phosphatase 1 by Casein Kinase II in Vitro
EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 29, No 4, 229-233, December 1997 Phosphorylation of chicken protein tyrosine phosphatase 1 by casein kinase II in vitro Eun Joo Jung,1 Kee Ryeon Kang1 and Introduction Yoon-Se Kang1,2 The phosphorylation of protein tyrosine residues is an early event in signal transduction initiated by binding of 1 Department of Biochemistry and Gyeongsang Institute of Cancer growth factors and hormones to their cognate receptors Research, College of Medicine, Gyeongsang National University, and it leads to regulation of cellular activities which include Chinju 660-280, Korea proliferation, differentiation, and also malignant transfor- 2 Corresponding author mation of cells (Hunter, 1989; Ullirich and Schlessinger, Accepted 17 November 1997 1990; Cantley et al., 1991). Under normal conditions, the level of tyrosine phosphorylation within a cell is determined by a balance between the actions of protein tyrosine Abbreviations: CPTP, chicken protein tyrosine phosphatase; HPTP1B, human placenta kinases (PTKs) and protein tyrosine phosphatases (PTPs) protein tyrosine phosphatase 1B; CKII, casein kinase II; MAP kinase, mitogen-activated (Hunter, 1989; Fischer et al., 1991; Trowbridge, 1991). protein kinase; GST, glutathione S-transferase; pNPP, p-nitrophenyl phosphate; EGF, PTPs do not simply reverse the action of tyrosine kinases, epidermal growth factor but rather, PTP itself may play a central role in cellular regulation. PTPs are generally classified as transmem- brane (receptor-type) and nontransmembrane (nonrecep- tor-type) enzymes based on the presence or absence of extracellular and transmembrane portions of their predicted sequence (Fischer et al., 1991). Because the activity of Abstract tyrosine kinase can be controlled by phosphorylation, it has been postulated that PTP activity may be regulated The phosphorylation and dephosphorylation of by phosphorylation as well. -

Tricarboxylic Acid (TCA) Cycle Intermediates: Regulators of Immune Responses
life Review Tricarboxylic Acid (TCA) Cycle Intermediates: Regulators of Immune Responses Inseok Choi , Hyewon Son and Jea-Hyun Baek * School of Life Science, Handong Global University, Pohang, Gyeongbuk 37554, Korea; [email protected] (I.C.); [email protected] (H.S.) * Correspondence: [email protected]; Tel.: +82-54-260-1347 Abstract: The tricarboxylic acid cycle (TCA) is a series of chemical reactions used in aerobic organisms to generate energy via the oxidation of acetylcoenzyme A (CoA) derived from carbohydrates, fatty acids and proteins. In the eukaryotic system, the TCA cycle occurs completely in mitochondria, while the intermediates of the TCA cycle are retained inside mitochondria due to their polarity and hydrophilicity. Under cell stress conditions, mitochondria can become disrupted and release their contents, which act as danger signals in the cytosol. Of note, the TCA cycle intermediates may also leak from dysfunctioning mitochondria and regulate cellular processes. Increasing evidence shows that the metabolites of the TCA cycle are substantially involved in the regulation of immune responses. In this review, we aimed to provide a comprehensive systematic overview of the molecular mechanisms of each TCA cycle intermediate that may play key roles in regulating cellular immunity in cell stress and discuss its implication for immune activation and suppression. Keywords: Krebs cycle; tricarboxylic acid cycle; cellular immunity; immunometabolism 1. Introduction The tricarboxylic acid cycle (TCA, also known as the Krebs cycle or the citric acid Citation: Choi, I.; Son, H.; Baek, J.-H. Tricarboxylic Acid (TCA) Cycle cycle) is a series of chemical reactions used in aerobic organisms (pro- and eukaryotes) to Intermediates: Regulators of Immune generate energy via the oxidation of acetyl-coenzyme A (CoA) derived from carbohydrates, Responses. -

Citric Acid Cycle
CHEM464 / Medh, J.D. The Citric Acid Cycle Citric Acid Cycle: Central Role in Catabolism • Stage II of catabolism involves the conversion of carbohydrates, fats and aminoacids into acetylCoA • In aerobic organisms, citric acid cycle makes up the final stage of catabolism when acetyl CoA is completely oxidized to CO2. • Also called Krebs cycle or tricarboxylic acid (TCA) cycle. • It is a central integrative pathway that harvests chemical energy from biological fuel in the form of electrons in NADH and FADH2 (oxidation is loss of electrons). • NADH and FADH2 transfer electrons via the electron transport chain to final electron acceptor, O2, to form H2O. Entry of Pyruvate into the TCA cycle • Pyruvate is formed in the cytosol as a product of glycolysis • For entry into the TCA cycle, it has to be converted to Acetyl CoA. • Oxidation of pyruvate to acetyl CoA is catalyzed by the pyruvate dehydrogenase complex in the mitochondria • Mitochondria consist of inner and outer membranes and the matrix • Enzymes of the PDH complex and the TCA cycle (except succinate dehydrogenase) are in the matrix • Pyruvate translocase is an antiporter present in the inner mitochondrial membrane that allows entry of a molecule of pyruvate in exchange for a hydroxide ion. 1 CHEM464 / Medh, J.D. The Citric Acid Cycle The Pyruvate Dehydrogenase (PDH) complex • The PDH complex consists of 3 enzymes. They are: pyruvate dehydrogenase (E1), Dihydrolipoyl transacetylase (E2) and dihydrolipoyl dehydrogenase (E3). • It has 5 cofactors: CoASH, NAD+, lipoamide, TPP and FAD. CoASH and NAD+ participate stoichiometrically in the reaction, the other 3 cofactors have catalytic functions. -

Differential Regulation of Cysteine Oxidative Post-Translational
www.nature.com/scientificreports OPEN Diferential regulation of cysteine oxidative post-translational modifcations in high and low Received: 16 April 2018 Accepted: 9 October 2018 aerobic capacity Published: xx xx xxxx Rodrigo W. A. Souza1, Christiano R. R. Alves 1,2, Alessandra Medeiros3, Natale Rolim4, Gustavo J. J. Silva4, José B. N. Moreira4, Marcia N. Alves4, Martin Wohlwend4, Mohammed Gebriel5, Lars Hagen5, Animesh Sharma 5, Lauren G. Koch6, Steven L. Britton7,8, Geir Slupphaug5, Ulrik Wisløf4,9 & Patricia C. Brum 1 Given the association between high aerobic capacity and the prevention of metabolic diseases, elucidating the mechanisms by which high aerobic capacity regulates whole-body metabolic homeostasis is a major research challenge. Oxidative post-translational modifcations (Ox-PTMs) of proteins can regulate cellular homeostasis in skeletal and cardiac muscles, but the relationship between Ox-PTMs and intrinsic components of oxidative energy metabolism is still unclear. Here, we evaluated the Ox-PTM profle in cardiac and skeletal muscles of rats bred for low (LCR) and high (HCR) intrinsic aerobic capacity. Redox proteomics screening revealed diferent cysteine (Cys) Ox-PTM profle between HCR and LCR rats. HCR showed a higher number of oxidized Cys residues in skeletal muscle compared to LCR, while the opposite was observed in the heart. Most proteins with diferentially oxidized Cys residues in the skeletal muscle are important regulators of oxidative metabolism. The most oxidized protein in the skeletal muscle of HCR rats was malate dehydrogenase (MDH1). HCR showed higher MDH1 activity compared to LCR in skeletal, but not cardiac muscle. These novel fndings indicate a clear association between Cys Ox-PTMs and aerobic capacity, leading to novel insights into the role of Ox- PTMs as an essential signal to maintain metabolic homeostasis. -
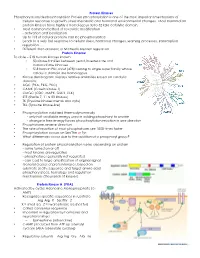
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -
Chapter 9 (Part 2): Glycolysis & Krebs Cycle (9.2 & 9.3)
NOTES: Chapter 9 (Part 2): Glycolysis & Krebs Cycle (9.2 & 9.3) ● CELLULAR RESPIRATION: reactions in living cells in which sugars are broken down and energy is released Mitochondria in a Liver Cell!! Glucose + oxygen carbon dioxide + water + ENERGY C6H12O6 + 6O2 6CO2 + 6H2O + energy ● Food (glucose), like fuel, is “burned” by our cells for energy; however if it is burned all at once, too much energy is released. ● So, the reaction is broken down into many small steps controlled by ENZYMES ● the energy is transferred to the bonds of ATP which stores and releases the energy in usable amounts (packets) to be used by the cell Recall: the ATP cycle -Glucose = “large denomination” ($100) -ATP = “small change” ($1) *For each molecule of glucose, the cell can make approximately 36-38 ATP. Steps of Cellular Respiration: Phase of Occurs Starts Ends # of ATP Resp. where? with? with? made cyto- Glycoly- plasm 1 2 2 sis glucose pyruvate; NADH Krebs cycle (Citric Acid Cyc) E.T.C. (Resp Chain) & oxidative phosphor Steps of Cellular Respiration: Phase of Occurs Starts Ends # of ATP Resp. where? with? with? made cyto- Glycoly- plasm 1 2 2 sis glucose pyruvate; NADH Krebs inner 2 4 CO2, cycle matrix of pyruvate NADH, 2 (Citric Acid mito- FADH2 Cyc) chondria E.T.C. (Resp Chain) & oxidative phosphor Steps of Cellular Respiration: Phase of Occurs Starts Ends # of ATP Resp. where? with? with? made cyto- Glycoly- plasm 1 2 2 sis glucose pyruvate; NADH Krebs inner 2 4 CO2, cycle matrix of pyruvate NADH, 2 (Citric Acid mito- FADH2 Cyc) chondria E.T.C.