Differential Effects of Cocaine on Dopamine Neuron Firing in Awake and Anesthetized Rats
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Interactions Among Diameter, Myelination, and the Na/K Pump Affect Axonal Resilience to High-Frequency Spiking
Interactions among diameter, myelination, and the Na/K pump affect axonal resilience to high-frequency spiking Yunliang Zanga,b and Eve Mardera,b,1 aVolen National Center for Complex Systems, Brandeis University, Waltham, MA 02454; and bDepartment of Biology, Brandeis University, Waltham, MA 02454 Contributed by Eve Marder, June 22, 2021 (sent for review March 25, 2021; reviewed by Carmen C. Canavier and Steven A. Prescott) Axons reliably conduct action potentials between neurons and/or (16–21). It remains unknown how these directly correlated pro- other targets. Axons have widely variable diameters and can be cesses, fast spiking and slow Na+ removal, interact in the control myelinated or unmyelinated. Although the effect of these factors of reliable spike generation and propagation. Furthermore, it is on propagation speed is well studied, how they constrain axonal unclear whether reducing Na+-andK+-current overlap can con- resilience to high-frequency spiking is incompletely understood. sistently decrease the rate of Na+ influx and accordingly enhance Maximal firing frequencies range from ∼1Hzto>300 Hz across axonal reliability to propagate high-frequency spikes. Conse- neurons, but the process by which Na/K pumps counteract Na+ quently, by modeling the process of Na+ removal in unmyelinated influx is slow, and the extent to which slow Na+ removal is com- and myelinated axons, we systematically explored the effects of patible with high-frequency spiking is unclear. Modeling the pro- diameter, myelination, and Na/K pump density on spike propagation cess of Na+ removal shows that large-diameter axons are more reliability. resilient to high-frequency spikes than are small-diameter axons, because of their slow Na+ accumulation. -

The Pharmacologist 2 0 0 6 December
Vol. 48 Number 4 The Pharmacologist 2 0 0 6 December 2006 YEAR IN REVIEW The Presidential Torch is passed from James E. Experimental Biology 2006 in San Francisco Barrett to Elaine Sanders-Bush ASPET Members attend the 15th World Congress in China Young Scientists at EB 2006 ASPET Awards Winners at EB 2006 Inside this Issue: ASPET Election Online EB ’07 Program Grid Neuropharmacology Division Mixer at SFN 2006 New England Chapter Meeting Summary SEPS Meeting Summary and Abstracts MAPS Meeting Summary and Abstracts Call for Late-Breaking Abstracts for EB‘07 A Publication of the American Society for 121 Pharmacology and Experimental Therapeutics - ASPET Volume 48 Number 4, 2006 The Pharmacologist is published and distributed by the American Society for Pharmacology and Experimental Therapeutics. The Editor PHARMACOLOGIST Suzie Thompson EDITORIAL ADVISORY BOARD Bryan F. Cox, Ph.D. News Ronald N. Hines, Ph.D. Terrence J. Monks, Ph.D. 2006 Year in Review page 123 COUNCIL . President Contributors for 2006 . page 124 Elaine Sanders-Bush, Ph.D. Election 2007 . President-Elect page 126 Kenneth P. Minneman, Ph.D. EB 2007 Program Grid . page 130 Past President James E. Barrett, Ph.D. Features Secretary/Treasurer Lynn Wecker, Ph.D. Secretary/Treasurer-Elect Journals . Annette E. Fleckenstein, Ph.D. page 132 Past Secretary/Treasurer Public Affairs & Government Relations . page 134 Patricia K. Sonsalla, Ph.D. Division News Councilors Bryan F. Cox, Ph.D. Division for Neuropharmacology . page 136 Ronald N. Hines, Ph.D. Centennial Update . Terrence J. Monks, Ph.D. page 137 Chair, Board of Publications Trustees Members in the News . -

Energy Expenditure Computation of a Single Bursting Neuron
Cognitive Neurodynamics (2019) 13:75–87 https://doi.org/10.1007/s11571-018-9503-3 (0123456789().,-volV)(0123456789().,-volV) ORIGINAL ARTICLE Energy expenditure computation of a single bursting neuron 2 1,2 2 2 Fengyun Zhu • Rubin Wang • Xiaochuan Pan • Zhenyu Zhu Received: 17 October 2017 / Revised: 22 August 2018 / Accepted: 28 August 2018 / Published online: 3 September 2018 Ó The Author(s) 2018 Abstract Brief bursts of high-frequency spikes are a common firing pattern of neurons. The cellular mechanisms of bursting and its biological significance remain a matter of debate. Focusing on the energy aspect, this paper proposes a neural energy calculation method based on the Chay model of bursting. The flow of ions across the membrane of the bursting neuron with or without current stimulation and its power which contributes to the change of the transmembrane electrical potential energy are analyzed here in detail. We find that during the depolarization of spikes in bursting this power becomes negative, which was also discovered in previous research with another energy model. We also find that the neuron’s energy consumption during bursting is minimal. Especially in the spontaneous state without stimulation, the total energy con- sumption (2.152 9 10-7 J) during 30 s of bursting is very similar to the biological energy consumption (2.468 9 10-7 J) during the generation of a single action potential, as shown in Wang et al. (Neural Plast 2017, 2017a). Our results suggest that this property of low energy consumption could simply be the consequence of the biophysics of generating bursts, which is consistent with the principle of energy minimization. -

Characteristics of Hippocampal Primed Burst Potentiation in Vitro and in the Awake Rat
The Journal of Neuroscience, November 1988, 8(11): 4079-4088 Characteristics of Hippocampal Primed Burst Potentiation in vitro and in the Awake Rat D. M. Diamond, T. V. Dunwiddie, and G. M. Rose Medical Research Service, Veterans Administration Medical Center, and Department of Pharmacology, University of Colorado Health Sciences Center, Denver, Colorado 80220 A pattern of electrical stimulation based on 2 prominent (McNaughton, 1983; Lynch and Baudry, 1984; Farley and Al- physiological features of the hippocampus, complex spike kon, 1985; Byrne, 1987). Of these, a phenomenontermed long- discharge and theta rhythm, was used to induce lasting in- term potentiation (LTP) has received extensive attention be- creases in responses recorded in area CA1 of hippocampal causeit sharesa number of characteristicswith memory. For slices maintained in vitro and from the hippocampus of be- example, LTP has a rapid onset and long duration and is having rats. This effect, termed primed burst (PB) potentia- strengthenedby repetition (Barnes, 1979; Teyler and Discenna, tion, was elicited by as few as 3 stimuli delivered to the 1987). In addition, the decay of LTP is correlated with the time commissural/associational afferents to CAl. The patterns courseof behaviorally assessedforgetting (Barnes, 1979; Barnes of stimulus presentation consisted of a single priming pulse and McNaughton, 1985). The similarity of LTP and memory followed either 140 or 170 msec later by a high-frequency has fostered an extensive characterization of the conditions un- burst of 2-10 pulses; control stimulation composed of un- derlying the initiation of LTP, as well as its biochemical sub- primed high-frequency trains of up to 10 pulses had no en- strates (Lynch and Baudry, 1984; Wigstrijm and Gustafsson, during effect. -

The Logic Behind Neural Control of Breathing Pattern Alona Ben-Tal 1, Yunjiao Wang2 & Maria C
www.nature.com/scientificreports OPEN The logic behind neural control of breathing pattern Alona Ben-Tal 1, Yunjiao Wang2 & Maria C. A. Leite3 The respiratory rhythm generator is spectacular in its ability to support a wide range of activities and Received: 10 January 2019 adapt to changing environmental conditions, yet its operating mechanisms remain elusive. We show Accepted: 29 May 2019 how selective control of inspiration and expiration times can be achieved in a new representation of Published: xx xx xxxx the neural system (called a Boolean network). The new framework enables us to predict the behavior of neural networks based on properties of neurons, not their values. Hence, it reveals the logic behind the neural mechanisms that control the breathing pattern. Our network mimics many features seen in the respiratory network such as the transition from a 3-phase to 2-phase to 1-phase rhythm, providing novel insights and new testable predictions. Te mechanism for generating and controlling the breathing pattern by the respiratory neural circuit has been debated for some time1–6. In 1991, an area of the brainstem, the pre Bötzinger Complex (preBötC), was found essential for breathing7. An isolated single PreBötC neuron could generate tonic spiking (a non-interrupted sequence of action potentials), bursting (a repeating pattern that consists of a sequence of action potentials fol- lowed by a time interval with no action potentials) or silence (no action potentials)8. Tese signals are transmit- ted, through other populations of neurons, to spinal motor neurons that activate the respiratory muscle4,6. Te respiratory muscle contracts when it receives a sequence of action potentials from the motor neurons and relaxes when no action potentials arrive9. -

The Pharmacologist December
Vol. 55 Number 4 2013 The Pharmacologist December Also in this issue: • 2013 Year in Review • 2013 Contributors • 2014 Election Nominees • Program grid for ASPET Annual Meeting at EB 2014 • Holiday Gift Ideas for Pharmacologists • Meet the 2014 Washington Fellows • MAPS Chapter Meeting abstracts (online only) Katharine Dexter McCormick (Courtesy of the Santa Barbara Historical Museum) The Pharmacologist is published and distributed by the American Society for Pharmacology and Experimental Therapeu cs. Contents THE PHARMACOLOGIST PRODUCTION TEAM NEWS Gary Axelrod President's Corner 193 Suzie Thompson Judith A. Siuciak, Ph.D. 2013 Year in Review 194 Richard Dodenhoff 2013 Contributors 196 Danielle Jordan 2013 Corporate Contributors 197 COUNCIL 2014 Elec on Nominees 198 President Richard R. Neubig, M.D., Ph.D. ASPET Annual Mee ng at EB 2014 President-Elect Program Grid 200 Anne e E. Fleckenstein, Ph.D. Past President Important Dates, Informa on, and Links 202 John S. Lazo, Ph.D. Holiday Gi s for Pharmacologists 204 Secretary/Treasurer Sandra P. Welch, Ph.D. FEATURE Secretary/Treasurer-Elect Katharine Dexter McCormick and the Pill Paul A. Insel, M.D. by Rebecca J. Anderson, Ph.D. 206 Past Secretary/Treasurer Edward T. Morgan, Ph.D. DEPARTMENTS Councilors Journals 214 Charles P. France, Ph.D. Science Policy 215 John D. Schuetz, Ph.D. Kenneth E. Thummel, Ph.D. Social Media 221 Chair, Board of Publica ons Trustees Book Review: S ff : The Curious Lives of Human Cadavers 222 James E. Barre , Ph.D. Chair, Program Commi ee In the Spotlight: Interviews with ASPET Members 223 Sco Waldman, M.D., Ph.D. -

WCBR Program3
Welcome to the Thirty-Fifth Annual Winter Conference on Brain Research The Winter Conference on Brain Research (WCBR) was founded in 1968 to promote free exchange of information and ideas within neuroscience. It was the intent of the founders that both formal and informal interactions would occur between clinical and laboratory based neuroscientists. During the past thirty years neuroscience has grown and expanded to include many new fields and methodologies. This diversity is also reflected by WCBR participants and in our program. A primary goal of the WCBR is to enable participants to learn about the current status of areas of neuroscience other than their own. Another objective is to provide a vehicle for scientists with common interests to discuss current issues in an informal setting. On the other hand, WCBR is not designed for presentations limited to communicating the latest data to a small group of specialists; this is best done at national society meetings. The program includes panels (reviews for an audience not neces- sarily familiar with the area presented), workshops (informal discussions of current issues and data), and a number of posters. The annual conference lecture will be presented at the Sunday breakfast on Sunday, January 27. Our guest speaker will be Dr. Donald Kennedy, Editor-in-Chief of Science. On Tuesday, January 29, a town meeting will be held for the Aspen/Snowmass commu- nity at which Dr. George Ricaurte, and WCBR participants will discuss drug addiction and toxicity of addictive drugs. Also, participants in the WCBR Outreach Program will present sessions at local schools throughout the week to pique students’ interest in science. -
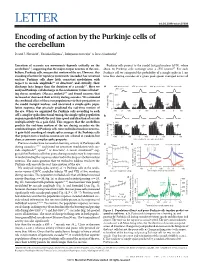
(2015) Encoding of Action by the Purkinje Cells of the Cerebellum
LETTER doi:10.1038/nature15693 Encoding of action by the Purkinje cells of the cerebellum David J. Herzfeld1, Yoshiko Kojima2, Robijanto Soetedjo2 & Reza Shadmehr1 Execution of accurate eye movements depends critically on the Purkinje cells project to the caudal fastigial nucleus (cFN), where cerebellum1–3, suggesting that the major output neurons of the cere- about 50 Purkinje cells converge onto a cFN neuron17. For each bellum, Purkinje cells, may predict motion of the eye. However, this Purkinje cell we computed the probability of a simple spike in 1-ms encoding of action for rapid eye movements (saccades) has remained time bins during saccades of a given peak speed, averaged across all unclear: Purkinje cells show little consistent modulation with respect to saccade amplitude4,5 or direction4, and critically, their discharge lasts longer than the duration of a saccade6,7.Herewe a 400° per s saccade 650° per s saccade 400° per s saccade 650° per s saccade analysed Purkinje-cell discharge in the oculomotor vermis of behav- 500° per s 8,9 200 ing rhesus monkeys (Macaca mulatta) and found neurons that N12 increased or decreased their activity during saccades. We estimated the combined effect of these two populations via their projections to 100 the caudal fastigial nucleus, and uncovered a simple-spike popu- lation response that precisely predicted the real-time motion of 0 the eye. When we organized the Purkinje cells according to each –1000 100 0 100 0 100 0 100 cell’s complex-spike directional tuning, the simple-spike population b 500° per s response predicted both the real-time speed and direction of saccade multiplicatively via a gain field. -

Analysis of Complex Bursting in Cortical Pyramidal Neuron Modelsଝ
Neurocomputing 32}33 (2000) 181}187 Analysis of complex bursting in cortical pyramidal neuron modelsଝ Adam Kepecs*, Xiao-Jing Wang Volen Center for Complex Systems, Brandeis University, Waltham, MA 02254, USA Accepted 13 January 2000 Abstract Burst "ring is a prominent feature of cortical pyramidal cells and is thought to have signi"cant functional roles in reliable signaling and synaptic plasticity. Modeling studies have successfully elucidated possible biophysical mechanisms underlying complex bursting in pyr- amidal cells. Based on these results (Pinsky, Rinzel, J. Comput. Neurosci. 1 (1994) 39}60), we have built a simpli"ed two-compartment burst model. Using the fast- and slow-variable analysis method, we show that complex bursting is an instance of square-wave bursting, where the dendritic slow potassium conductance is the single slow variable. The coupling parameters between the two compartments change the topological class of bursting thereby altering the "ring patterns of the neuron. These results explain the diverse set of "ring patterns seen with di!erent dendritic morphologies (Mainen, Sejnowski, Nature 382 (1996) 363}366). ( 2000 Elsevier Science B.V. All rights reserved. Keywords: Phase-plane analysis; Complex burst; Bifurcation diagram 1. Introduction Pyramidal cells in many cortical areas "re stereotyped bursts of action potentials termed complex spikes or complex bursts. These bursts consist of 2}7 action poten- tials occurring in a &30 ms window. First observed in hippocampal single-unit extracellular recordings, these were termed &complex spikes' and were later identi"ed by intracellular techniques. Burst "ring is thought to play an important role in reliable signaling [5,15] and synaptic plasticity [2]. -

Abnormal High-Frequency Burst Firing of Cerebellar Neurons in Rapid-Onset Dystonia-Parkinsonism
The Journal of Neuroscience, August 27, 2014 • 34(35):11723–11732 • 11723 Neurobiology of Disease Abnormal High-Frequency Burst Firing of Cerebellar Neurons in Rapid-Onset Dystonia-Parkinsonism Rachel Fremont,* D. Paola Calderon,* X Sara Maleki, and Kamran Khodakhah Dominick P. Purpura Department of Neuroscience, Albert Einstein College of Medicine, Bronx, New York 10461 Loss-of-function mutations in the ␣3 isoform of the Na ϩ/K ϩ ATPase (sodium pump) are responsible for rapid-onset dystonia parkin- sonism (DYT12). Recently, a pharmacological model of DYT12 was generated implicating both the cerebellum and basal ganglia in the disorder. Notably, partially blocking sodium pumps in the cerebellum was necessary and sufficient for induction of dystonia. Thus, a key question that remains is how partially blocking sodium pumps in the cerebellum induces dystonia. In vivo recordings from dystonic mice revealed abnormal high-frequency bursting activity in neurons of the deep cerebellar nuclei (DCN), which comprise the bulk of cerebellar output. In the same mice, Purkinje cells, which provide strong inhibitory drive to DCN cells, also fired in a similarly erratic manner. In vitro studies demonstrated that Purkinje cells are highly sensitive to sodium pump dysfunction that alters the intrinsic pacemaking of these neurons, resulting in erratic burst firing similar to that identified in vivo. This abnormal firing abates when sodium pump function is restored and dystonia caused by partial block of sodium pumps can be similarly alleviated. These findings suggest that persistent high-frequency burst firing of cerebellar neurons caused by sodium pump dysfunction underlies dystonia in this model of DYT12. Key words: basal ganglia; cerebellum Introduction sonism, otherwise referred to as DYT12 (de Carvalho et al., 2004; Dystonia is the third most common movement disorder after Brashear et al., 2007). -

Fast Rhythmic Bursting Can Be Induced in Layer 2/3 Cortical Neurons by Enhancing Persistent Naϩ Conductance Or by Blocking BK Channels
J Neurophysiol 89: 909–921, 2003; 10.1152/jn.00573.2002. Fast Rhythmic Bursting Can Be Induced in Layer 2/3 Cortical Neurons by Enhancing Persistent Naϩ Conductance or by Blocking BK Channels ROGER D. TRAUB,1 EBERHARD H. BUHL,2 TENGIS GLOVELI,2 AND MILES A. WHITTINGTON2 1Departments of Physiology and Pharmacology and Neurology, State University of New York Health Science Center, Brooklyn, New York 11203; and 2Division of Biomedical Sciences, The Worsley Building, University of Leeds, Leeds LS2 9NQ, United Kingdom Submitted 18 July 2002; accepted in final form 29 October 2002 Traub, Roger D., Eberhard H. Buhl, Tengis Gloveli, and Miles A. Cormick1996). These latter chattering cells responded to both Whittington. Fast rhythmic bursting can be induced in layer 2/3 ϩ visual stimulation and sufficiently large depolarizing pulses, cortical neurons by enhancing persistent Na conductance or with fast rhythmic bursts. In cat cortex in vivo, deep thalamic- by blocking BK channels. J Neurophysiol 89: 909–921, 2003; projecting neurons, and also aspiny (presumed inhibitory) neu- 10.1152/jn.00573.2002. Fast rhythmic bursting (or “chattering”) is a rons, could also exhibit fast rhythmic bursting during depolar- firing pattern exhibited by selected neocortical neurons in cats in vivo and in slices of adult ferret and cat brain. Fast rhythmic bursting (FRB) has izing current pulses of appropriate amplitude (Steriade et al. been recorded in certain superficial and deep principal neurons and in 1998); intralaminar thalamocortical neurons in vivo can also aspiny presumed local circuit neurons; it can be evoked by depolarizing discharge in this pattern (Steriade et al. -

WCBR Program 01
Welcome to the Thirty-Fourth Annual Winter Conference on Brain Research The Winter Conference on Brain Research (WCBR) was founded in 1968 to promote free exchange of information and ideas within neuroscience. It was the intent of the founders that both formal and informal interactions would occur between clinical and laboratory based neuroscientists. During the past thirty years neuroscience has grown and expanded to include many new fields and methodologies. This diversity is also reflected by WCBR participants and in our program. A primary goal of the WCBR is to enable participants to learn about the current status of areas of neuroscience other than their own. Another objective is to provide a vehicle for scientists with common interests to discuss current issues in an informal setting. On the other hand, WCBR is not designed for presentations limited to communicating the latest data to a small group of specialists; this is best done at national society meetings. The program includes panels (reviews for an audience not necessarily familiar with the area presented), workshops (informal discussions of current issues and data), and a number of posters. The annual confer- ence lecture will be presented at breakfast on Sunday, January 21. Our guest speaker will be Dr. Gerald Fischbach, Director of the National Institute for Neurological Disorders and Stroke at the National Institutes of Health. On Tuesday, January 23, a town meeting will be held for the Steamboat Springs community. Here Dr. Charles O Brien, and WCBR participants will discuss the neurobiological basis of mental illness. Also, participants in the WCBR outreach program will present sessions at local schools throughout the week to pique stu- dents interest in science.