Immunology in India: an Emerging Story
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Annual Report 2014 - 2015 Our Mission
ANNUAL REPORT 2014 - 2015 Our Mission By integrating the fields of medicine, science, engineering and technology into translational knowledge and making the resulting biomedical innovations accessible to public health, to improve the health of the most disadvantaged people in India and throughout the world. Our Vision As a networked organization linking many centers of excellence, THSTI is envisaged as a collective of scientists, engineers and physicians that will effectively enhance the quality of human life through integrating a culture of shared excellence in research, education and translational knowledge with the developing cohorts and studying the pathogenesis and the molecular mechanisms of disease to generate knowledge to complement the processes of designing interventions and technology development. CONTENTS 3 THE ORGANIZATION Society Governing Body Leadership From the Executive Director’s Desk 11 RESEARCH PROGRAMS Vaccine and Infectious Disease Research Centre Pediatric Biology Centre Centre for Biodesign and Diagnostics Policy Center for Biomedical Research Drug Discovery Research Centre Centre for Human Microbial Ecology Population Science Partnership Centre Clinical Development Services Agency 178 ACADEMIA 184 ADMINISTRATION Organization INTRAMURAL CENTRES Vaccine & Infectious Disease Research Centre (VIDRC) Pediatric Biology Centre (PBC) Centre for Biodesign & Diagnostics (CBD) Centre for Human Microbial Ecology (CHME) Policy Centre for Biomedical Research (PCBR) Drug Discovery Research Centre (DDRC) PARTNERSHIP CENTRE Population Science Partnership Centre (PSPC) EXTRAMURAL CENTRE Clinical Development Services Agency (CDSA) THSTI Society 1 2 3 4 5 6 7 8 9 10 11 12 1. Dr. G. Padmanaban 5. Dr. T.S. Rao 9. Dr. J. Gowrishankar Distinguished Professor, Nodal Officer, THSTI, Sr. Advisor, Director, IISc Bangalore Department of Biotechnology, Centre for DNA Fingerprinting President New Delhi & Diagnostics, Member Ex-officio Hyderabad 2. -
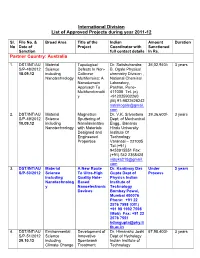
International Division List of Approved Projects During Year 2011-12
International Division List of Approved Projects during year 2011-12 Sl. File No. & Broad Area Title of the Indian Amount Duration No Date of Project Coordinator with Sanctioned Sanction full contact details In Rs. Partner Country: Australia 1. DST/INT/A U Material Topological Dr. Satishchandra 36,02,940/- 3 years S/P-48/2012 Science Defects In Non- B. Ogale Physical 18.09.12 including Collinear chemistry Division , Nanotechnology Multiferroics: A National Chemical Nanodomain Laboratory, Approach To Pashan, Pune- Multifunctionalit 411008 Tel. (o) y +912025902260 (M) 91-9822628242 satishogale@gmail. com 2. DST/INT/AU Material Magnetron Dr. V.K. Srivastava 39,36,600/- 3 years S/P-49/2012 Science Sputtering of Dept. of Mechanical 10.09.12 including Nanolaminates Engg., Banaras Nanotechnology with Materials Hindu University Designed and Institute Of Engineered Technology Properties Varanasi – 221005 Tel.(+91) 9453915551 Fax: (+91) 542 2368428 vijayks210@gmail. com 3. DST/INT/AU Material A New Route Dr. Kantimay Das Under 3 years S/P-50/2012 Science To Ultra-High Gupta Dept of Process including Quality Hole- Physics Indian Nanotechnolog Based Institute of y Nanoelectronic Technology Devices Bombay Powai, Mumbai 400076 Phone: +91 22 2576 7598 (Off.) +91 98 1992 7598 (Mob) Fax: +91 22 2576 7551 [email protected] tb.ac.in 4. DST/INT/AU Environmental Development of Dr. Himanshu Joshi 67,98,400/- 3 years S/P-51/2012 Science Innovative Dept of Hydrology 29.10.12 including Spentwash Indian Institute of Climate Change Treatment Technology Research Systems and Roorkee– 247667, Resource Uttarakhand Recovery himanshujoshi58@ Options for gmail.com; Distillery himanshu_joshi58 Application @yahoo.co.in; [email protected] n 5. -

Dr Sanjeev Galande Joins As Dean of School of Natural Sciences at SNU
Dr Sanjeev Galande joins as Dean of School of Natural Sciences at SNU 02 June 2021 | News Dr Galande brings with him over two decades of experience as a scientist and academic Shiv Nadar University Delhi-NCR has appointed cell biologist and epigeneticist Dr Sanjeev Galande as Dean of the School of Natural Sciences. Dr Galande joins the university from the Indian Institute of Science Education & Research (IISER)-Pune, where he served as Professor of Biology, and Dean of Research & Development. Dr Galande brings with him over two decades of experience as a scientist and academic. Dr Galande is credited for having established a multidisciplinary programme engaged at the interface of biochemistry, molecular biology, bioinformatics, cell biology, proteomics, and genomics. Dr Galande was the recipient of the International Senior Research Fellowship, Wellcome Trust, UK, 2005-10, DBT National Bioscience Award 2006, DST Swarnajayanti Fellowship 2007, the coveted CSIR Shanti Swarup Bhatnagar Prize 2010, GD Birla Award for Scientific Excellence 2015, and the SERB JC Bose Fellowship 2019. He has been a Fellow of the Indian Academy of Sciences since 2010, of the Indian National Science Academy since 2012, and of the National Academy of Sciences since 2017. He was an honorary associate faculty at the University of Sydney, Australia, and a visiting faculty at the University of Turku, Finland. Dr Rupamanjari Ghosh, Vice-Chancellor of Shiv Nadar University Delhi-NCR, said, “Dr Galande is a great fit to lead and build upon its success, as we continue to elevate the University’s position to the world stage as an ‘Institution of Eminence.'" Dr Galande said, “I am delighted to be part of a university that aspires to be among the leading research-based, multidisciplinary universities of global stature and impact. -

Touching New Heights Editor’S Note
ENSEMBLEENSEMBLE Volume 4 (1) | January – February 2016 Newsletter of the Indo-French Centre for the Promotion of Advanced Research India-France Relations Touching New Heights editor’s note Dear Readers, India and France are witnessing the emergence of a new era of collaborative efforts between the two countries in various sectors. In November/December 2015, France hosted a successful global scale diplomatic event for adopting an agreement by many countries on climate change where India was also an active participant. After the visit of Honourable Prime Minister of India Shri Narendra Modi to France in April 2015, the French President H. E. Dr. Mukesh Kumar Mr. François Hollande visited India in January, 2016 as a Chief Guest on the Director, CEFIPRA occasion of 67th Republic Day of India. During his visit, Indian and French scientific as well as technological communities from academia and industry sectors, joined hands through several Agreements/MoUs signed by the two countries. CEFIPRA had the privilege of hosting the officials / signatories of three of such Agreements/MoU signed on 25 January 2016. CEFIPRA, since its evolution as a unique institutional platform and collaborative mechanism, is contributing through its various interventions in a diverse range of S&T domains. These collaborative efforts are making it possible to generate significant knowledge that has a potential to translate discovery science into solution science. I sincerely wish that all these MoUs will create new pathways to further strengthen the Indo-French collaborative research efforts. inside Editor-in-Chief Dr. Mukesh Kumar ii | editor’s note GD Birla Award x | Director, CEFIPRA Dr. -

Annual Reports 2011-12
Contents 1 Preamble 3 2 Mandate of the Centre 4 3 Scientific reports 5 Structural Biology of Regulatory Events in Physiological Processes 7 Mechanisms of Cell Division and Cellular Dynamics 10 Engineering of Nanomaterials for Biomedical Applications 16 Structural Biology of Bacterial Surface Proteins 20 Investigating Molecular Mechanism in the Ubiquitin Mediated 22 Signalling in Cellular Pathways Studies on Biology of Infectious and Idiopathic Inflammation of the Gut 25 Pathophysiology of Hemolysis and Thrombosis 29 Intrinsic Signals that Regulate Skeletal Muscle Structure and Function 31 4 Profiles of Faculty Members joining during 2011-2012 33 5 Scientific Activities and Achievements 37 RCB Colloquia 39 Workshops conducted by RCB 40 Seminars delivered by visiting scientists at RCB 41 Lectures delivered / Conferences attended/ Visits abroad 46 Membership of professional/ Academic bodies/ Editorial Boards 48 Distinctions, Honours and Awards 50 6. Infrastructure development 51 Administrative activities 53 Interim Laboratories in NCR, Gurgaon 53 Permanent Campus of the NCR-BSC Project at Faridabad 53 7 Audited Statement of Accounts of the Centre 57 8 Institutional Information 63 Committees of Regional Centre for Biotechnology 65 Staff of the Regional Centre 69 1 2 Preamble It is indeed a great pleasure to report substantial developments and growth at the Regional Centre for Biotechnology this year considering the limitations of functioning from an interim campus in Gurgaon. While the construction of the laboratory building in the Faridabad campus is in full swing, consolidation of academic and research programmes at the Gurgaon campus is underway. As efforts are being made to foray into new multidisciplinary facets of biotech science, the small beginning made in the field of biomedical sciences last year got accelerated. -

IISER Pune Annual Report 2015-16 Chairperson Pune, India Prof
dm{f©H$ à{VdoXZ Annual Report 2015-16 ¼ããäÌãÓ¾ã ãä¶ã¹ã¥ã †Ìãâ Êãà¾ã „ÞÞã¦ã½ã ½ãÖ¦Ìã ‡ãŠñ †‡ãŠ †ñÔãñ Ìãõ—ãããä¶ã‡ãŠ ÔãâÔ©ãã¶ã ‡ãŠãè Ô©ãã¹ã¶ãã ãä•ãÔã½ãò ‚㦾ãã£ãìãä¶ã‡ãŠ ‚ã¶ãìÔãâ£ãã¶ã Ôããä֦㠂㣾ãã¹ã¶ã †Ìãâ ãäÍãàã¥ã ‡ãŠã ¹ãî¥ãùã Ôãñ †‡ãŠãè‡ãŠÀ¥ã Öãñý ãä•ã—ããÔãã ¦ã©ãã ÀÞã¶ã㦽ã‡ãŠ¦ãã Ôãñ ¾ãì§ãŠ ÔãÌããó§ã½ã Ôã½ãã‡ãŠÊã¶ã㦽ã‡ãŠ ‚㣾ãã¹ã¶ã ‡ãñŠ ½ã㣾ã½ã Ôãñ ½ããõãäÊã‡ãŠ ãäÌã—ãã¶ã ‡ãŠãñ ÀãñÞã‡ãŠ ºã¶ãã¶ããý ÊãÞããèÊãñ †Ìãâ Ôããè½ããÀãäÖ¦ã / ‚ãÔããè½ã ¹ã㟿ã‰ãŠ½ã ¦ã©ãã ‚ã¶ãìÔãâ£ãã¶ã ¹ããäÀ¾ããñ•ã¶ãã‚ããò ‡ãñŠ ½ã㣾ã½ã Ôãñ œãñ›ãè ‚ãã¾ãì ½ãò Öãè ‚ã¶ãìÔãâ£ãã¶ã àãñ¨ã ½ãò ¹ãÆÌãñÍãý Vision & Mission Establish scientific institution of the highest caliber where teaching and education are totally integrated with state-of-the- art research Make learning of basic sciences exciting through excellent integrative teaching driven by curiosity and creativity Entry into research at an early age through a flexible borderless curriculum and research projects Annual Report 2015-16 Governance Correct Citation Board of Governors IISER Pune Annual Report 2015-16 Chairperson Pune, India Prof. T.V. Ramakrishnan (till 03/12/2015) Emeritus Professor of Physics, DAE Homi Bhabha Professor, Department of Physics, Indian Institute of Science, Bengaluru Published by Dr. K. Venkataramanan (from 04/12/2015) Director and President (Engineering and Construction Projects), Dr. -

Centenary Celebration of Bose Institute
C E N T E N A R Y C E L E B R AT I O N CCEENNTTEENNAARRYY CCEELLEEBBRRAATTIIOONN OOFF BBOOSSEE IINNSSTTIITTUUTTEE 1 9 1 7 - 2 0 1 7 List of Posters (24.11.2017–28.11.2017) Poster Faculty Programme Title Poster Faculty Programme Title No No A-1 Amita Pal I Molecular characterization of VmMAPK1 and deciphering its role in restricting D-10 Tanya Das IV Is cancer a stem cell disease? MYMIV multiplication in tobacco Poulami Khan, Apoorva Bhattacharya, Shruti Banerjee, Swastika Paul, Abhishek Dutta, Anju Patel, Pankaj Kumar Singh, Shubho Chaudhuri and Amita Pal Dipanwita Dutta Chowdhury, Udit Basak, Apratim Dutta, Arijit Bhowmik, Devdutt A-2 Anupama Ghosh I Induction of apoptosis-like cell death and clearance of stress-induced intracellular Mazumdar, Aparajita Das, Sourio Chakraborty and Tanya Das protein aggregates: dual roles for Ustilago maydis metacaspase Mca1. E-1 Abhrajyoti Ghosh V Deciphering the code behind prokaryotic stress responses and ecophysiology A-1 Dibya Mukherjee, Sayandeep Gupta, Saran N, Rahul Datta, Anupama Ghosh Mousam Roy, Sayandeep Gupta, Chandrima Bhattacharyya, Shayantan Mukherji, A-3 Debabrata Basu I A multifaceted approach to unravel the signalling components of 'Black Spot' Abhrajyoti Ghosh A-2 disease resistance in oilseed mustard E-2 Srimonti Sarkar V The minimal ESCRT machinery of Giardia lamblia has altered inter-subunit Mrinmoy Mazumder, Amrita Mukherjee, Banani Mondal, Swagata Ghosh, Aishee De interactions within the ESCRT-II and ESCRT-III complexes A-3 and Debabrata Basu Nabanita Saha, Somnath Dutta and Srimonti Sarkar A-4 I Gaurab Gangyopadhyay Towards broadening the gene pool of few crop plants through molecular and E-3 Subrata Sau V Identification, purification and characterization of a cyclophilin from A-4 transgenic breeding Staphylococcus aureus Debabrata Dutta, Soumili Pal, Marufa Sultana, Vivek Arora and Gaurab Soham Seal, Debabrata Sinha, Subrata Sau Gangopadhyay E-4 Sujoy Kr. -

List of Life Members As on 20Th January 2021
LIST OF LIFE MEMBERS AS ON 20TH JANUARY 2021 10. Dr. SAURABH CHANDRA SAXENA(2154) ALIGARH S/O NAGESH CHANDRA SAXENA POST HARDNAGANJ 1. Dr. SAAD TAYYAB DIST ALIGARH 202 125 UP INTERDISCIPLINARY BIOTECHNOLOGY [email protected] UNIT, ALIGARH MUSLIM UNIVERSITY ALIGARH 202 002 11. Dr. SHAGUFTA MOIN (1261) [email protected] DEPT. OF BIOCHEMISTRY J. N. MEDICAL COLLEGE 2. Dr. HAMMAD AHMAD SHADAB G. G.(1454) ALIGARH MUSLIM UNIVERSITY 31 SECTOR OF GENETICS ALIGARH 202 002 DEPT. OF ZOOLOGY ALIGARH MUSLIM UNIVERSITY 12. SHAIK NISAR ALI (3769) ALIGARH 202 002 DEPT. OF BIOCHEMISTRY FACULTY OF LIFE SCIENCE 3. Dr. INDU SAXENA (1838) ALIGARH MUSLIM UNIVERSITY, ALIGARH 202 002 HIG 30, ADA COLONY [email protected] AVANTEKA PHASE I RAMGHAT ROAD, ALIGARH 202 001 13. DR. MAHAMMAD REHAN AJMAL KHAN (4157) 4/570, Z-5, NOOR MANZIL COMPOUND 4. Dr. (MRS) KHUSHTAR ANWAR SALMAN(3332) DIDHPUR, CIVIL LINES DEPT. OF BIOCHEMISTRY ALIGARH UP 202 002 JAWAHARLAL NEHRU MEDICAL COLLEGE [email protected] ALIGARH MUSLIM UNIVERSITY ALIGARH 202 002 14. DR. HINA YOUNUS (4281) [email protected] INTERDISCIPLINARY BIOTECHNOLOGY UNIT ALIGARH MUSLIM UNIVERSITY 5. Dr. MOHAMMAD TABISH (2226) ALIGARH U.P. 202 002 DEPT. OF BIOCHEMISTRY [email protected] FACULTY OF LIFE SCIENCES ALIGARH MUSLIM UNIVERSITY 15. DR. IMTIYAZ YOUSUF (4355) ALIGARH 202 002 DEPT OF CHEMISTRY, [email protected] ALIGARH MUSLIM UNIVERSITY, ALIGARH, UP 202002 6. Dr. MOHAMMAD AFZAL (1101) [email protected] DEPT. OF ZOOLOGY [email protected] ALIGARH MUSLIM UNIVERSITY ALIGARH 202 002 ALLAHABAD 7. Dr. RIAZ AHMAD(1754) SECTION OF GENETICS 16. -

PE PGRS33 Contributes to Mycobacterium Tuberculosis Entry in Macrophages Through Interaction with TLR2
RESEARCH ARTICLE PE_PGRS33 Contributes to Mycobacterium tuberculosis Entry in Macrophages through Interaction with TLR2 Ivana Palucci1, Serena Camassa1, Alessandro Cascioferro3, Michela Sali1, Saber Anoosheh3, Antonella Zumbo1, Mariachiara Minerva1, Raffaella Iantomasi1, Flavio De Maio1, Gabriele Di Sante2, Francesco Ria2, Maurizio Sanguinetti1, Giorgio Palù3, Michael J. Brennan4, Riccardo Manganelli3, Giovanni Delogu1* 1 Institute of Microbiology, Università Cattolica del Sacro Cuore, L.go A. Gemelli, 8–00168, Rome, Italy, 2 Institute of General Pathology, Università Cattolica del Sacro Cuore, L.go A. Gemelli, 8–00168, Rome, Italy, 3 Department of Molecular Medicine, University of Padua, Via A. Gabelli, 63–35121, Padua, Italy, 4 Aeras, Rockville (MD), United States of America * [email protected] Abstract PE_PGRS represent a large family of proteins typical of pathogenic mycobacteria whose members are characterized by an N-terminal PE domain followed by a large Gly-Ala repeat- OPEN ACCESS rich C-terminal domain. Despite the abundance of PE_PGRS-coding genes in the Myco- Citation: Palucci I, Camassa S, Cascioferro A, Sali bacterium tuberculosis (Mtb) genome their role and function in the biology and pathogene- M, Anoosheh S, Zumbo A, et al. (2016) PE_PGRS33 sis still remains elusive. In this study, we generated and characterized an Mtb H37Rv Contributes to Mycobacterium tuberculosis Entry in mutant (MtbΔ33) in which the structural gene of PE_PGRS33, a prototypical member of the Macrophages through Interaction with TLR2. PLoS ONE 11(3): e0150800. doi:10.1371/journal. protein family, was inactivated. We showed that this mutant entered macrophages with an pone.0150800 efficiency up to ten times lower than parental or complemented strains, while its efficiency in Editor: Joyoti Basu, Bose Institute, INDIA infecting pneumocytes remained unaffected. -
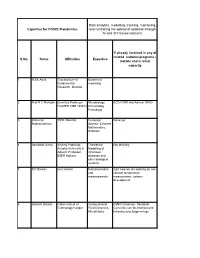
Data Analytics and Modeling
Data analytics, modelling, tracking, monitoring, Expertise for COVID /Pandemics and controlling the spread of outbreak through AI and IOT-based solutions If already involved in any of related national programs : S.No. Name Affiliation Expertise Details and in what capacity 1 H. M. Antia Tata Institute of Numerical Fundamental modelling Research, Mumbai 2 Prof R C Mahajan Emeritus Professor Microbiology ECD ICMR and Advisor WHO PGIMER CHD 160012 Immunology Parasitogy 3 Jaikumar TIFR, Mumbai Computer None yet. Radhakrishnan Science, Discrete Mathematics, Software 4 Somdatta Sinha Visiting Professor, Theoretical Not officially. Ashoka University & Modelling of Adjunct Professor, infectious IISER Kolkata diseases and other biological systems 5 S K Biswas iiser mohali Instrumentation right now we are working on non and contact temperature measurements measurement system development 6 Gautam Biswas Indian Intitute of Computational GIAN (Chairman, Sectional Technology Kanpur Fluid Dynamics, Committee on Mechanical and Microfluidics Infrastructure Engineering) 7 Ram Ramaswamy IIT Delhi Modeling - 8 Sandip Paul Ramanujan Fellow NGS data NA analysis, adaptive evolution 9 Amish kumar Ph.D. Student Bioinformatics After submission of my Ph.D thesis I am working as visitor research student at Department of plant sciences under Project entitled as "TIGR2ESS: Transforming India's Green Revolution by Research and Empowerment for Sustainable food Supplies". 10 Surajit Assistant Professor, Immunology, None Bhattacharjee Department of Infection Biology Molecular -

List of Officers Who Attended Courses at NCRB
List of officers who attened courses at NCRB Sr.No State/Organisation Name Rank YEAR 2000 SQL & RDBMS (INGRES) From 03/04/2000 to 20/04/2000 1 Andhra Pradesh Shri P. GOPALAKRISHNAMURTHY SI 2 Andhra Pradesh Shri P. MURALI KRISHNA INSPECTOR 3 Assam Shri AMULYA KUMAR DEKA SI 4 Delhi Shri SANDEEP KUMAR ASI 5 Gujarat Shri KALPESH DHIRAJLAL BHATT PWSI 6 Gujarat Shri SHRIDHAR NATVARRAO THAKARE PWSI 7 Jammu & Kashmir Shri TAHIR AHMED SI 8 Jammu & Kashmir Shri VIJAY KUMAR SI 9 Maharashtra Shri ABHIMAN SARKAR HEAD CONSTABLE 10 Maharashtra Shri MODAK YASHWANT MOHANIRAJ INSPECTOR 11 Mizoram Shri C. LALCHHUANKIMA ASI 12 Mizoram Shri F. RAMNGHAKLIANA ASI 13 Mizoram Shri MS. LALNUNTHARI HMAR ASI 14 Mizoram Shri R. ROTLUANGA ASI 15 Punjab Shri GURDEV SINGH INSPECTOR 16 Punjab Shri SUKHCHAIN SINGH SI 17 Tamil Nadu Shri JERALD ALEXANDER SI 18 Tamil Nadu Shri S. CHARLES SI 19 Tamil Nadu Shri SMT. C. KALAVATHEY INSPECTOR 20 Uttar Pradesh Shri INDU BHUSHAN NAUTIYAL SI 21 Uttar Pradesh Shri OM PRAKASH ARYA INSPECTOR 22 West Bengal Shri PARTHA PRATIM GUHA ASI 23 West Bengal Shri PURNA CHANDRA DUTTA ASI PC OPERATION & OFFICE AUTOMATION From 01/05/2000 to 12/05/2000 1 Andhra Pradesh Shri LALSAHEB BANDANAPUDI DY.SP 2 Andhra Pradesh Shri V. RUDRA KUMAR DY.SP 3 Border Security Force Shri ASHOK ARJUN PATIL DY.COMDT. 4 Border Security Force Shri DANIEL ADHIKARI DY.COMDT. 5 Border Security Force Shri DR. VINAYA BHARATI CMO 6 CISF Shri JISHNU PRASANNA MUKHERJEE ASST.COMDT. 7 CISF Shri K.K. SHARMA ASST.COMDT. -

ANNUAL REPORT April 2009 to March 2010
Ôããè ¡ãè †¹ãŠ ¡ãè CDFD ÌãããäÓãÇ㊠¹ãÆãä¦ãÌãñª¶ã ‚ã¹ãÆõÊã 2009 Ôãñ ½ããÞãà 2010 ¦ã‡ãŠ ANNUAL REPORT April 2009 to March 2010 ¡ãè †¶ã † ãä¹ãâŠØãÀãä¹ãÆâãä›âØã †Ìãâ ãä¶ãªã¶ã ‡ãñŠ¶³ ¶ãã½ã¹ãÊÊããè, ÖõªÀãºã㪠- 500 001 Centre for DNA Fingerprinting and Diagnostics Nampally, Hyderabad - 500 001 1 2 CONTENTS I Mandate 5 II From the Director’s Desk 9 III Services 1. Laboratory of DNA Fingerprinting Services 15 2. Diagnostic Services 18 IV Research 1. Laboratory of Molecular Genetics 27 2. Laboratory of Genomics and Profiling Applications 35 3. Laboratory of Fungal Pathogenesis 40 4. Laboratory of Immunology 44 5. Laboratory of Bacterial Genetics 48 6. Laboratory of Computational Biology 53 7. Laboratory of Molecular Cell Biology 57 8. Laboratory of Structural Biology 61 9. Laboratory of Mammalian Genetics 65 10. Laboratory of Molecular Oncology 69 11. Laboratory of Cancer Biology 74 12. Laboratory of Computational and Functional Genomics 78 13. Laboratory of Transcription 83 14. Laboratory of Cell Signalling 87 15. Laboratory of Plant Microbe Interaction 91 16. Other Scientific Services / Facilities a. Bioinformatics 97 b. Instrumentation 98 V Publications 99 VI Human Resource Development 107 VII Lectures, Meetings, Workshops, and Important Events 111 VIII Senior Staff and Officers of CDFD 115 IX Deputations Abroad of CDFD Personnel 119 X Committees of the Institute 123 XI Budget and Finance 133 XII Auditor’s Report 137 XIII Photo Gallery 227 3 4 Mandate 5 6 MANDATE The objectives for which the Centre for DNA Fingerprinting and Diagnostics (CDFD) was established as enumerated in Memorandum of Association and Rules and Regulations of CDFD Society are as follows: i.