Methacrylic Acid and Esters, Supp. B
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Modeling of the Free-Radical Copolymerization Kinetics With
Revista de la Sociedad Química de México, Vol. 47, Núm. 1 (2003) 22-33 Investigación Modeling of the Free-Radical Copolymerization Kinetics with Crosslinking of Methyl Methacrylate / Ethylene Glycol Dimethacrylate Up to High Conversions and Considering Thermal Effects Eduardo Vivaldo-Lima*, Rosalba García-Pérez, and Oswaldo J. Celedón-Briones Departamento de Ingeniería Química, Facultad de Química, Conjunto E, Universidad Nacional Autónoma de México, Ciudad Universitaria, México D.F., CP 04510, México. Tel.: 5622-5256; Fax: 5622-5355; E-mail: [email protected] Recibido el 13 de marzo del 2002; aceptado el 28 de enero del 2003 Resumen. Se usa un modelo matemático para copolimerización por Abstract. A mathematical model for the free radical copolymeriza- radicales libres con entrecruzamiento para modelar el comportamien- tion kinetics with crosslinking of vinyl / divinyl monomers is used to to del sistema de copolimerización metacrilato de metilo (MMA) / model the free-radical copolymerization of methyl methacrylate dimetacrilato de etilén glicol (EGDMA). Las predicciones del mode- (MMA)/ethylene glycol dimethacrylate (EGDMA). Good agreement lo se comparan contra datos experimentales de literatura, teniendo between model predictions and experimental data from the literature buena concordancia a bajas conversiones, y bajas concentraciones de is obtained at low conversions, and low crosslinker concentrations. agente de entrecruzamiento. Si se consideran en el modelo los efectos The agreement is also good at high conversions, and intermediate térmicos asociados a la polimerización con temperatura no uniforme crosslinker concentrations, if thermal effects in ampoule copolyme- en ampolletas de más de 0.5 mm de diámetro externo, la concordan- rization are incorporated into the model, through an energy balance. -

FULL PAPER Upon the Α-Methylenation of Methyl
FULL PAPER Upon the α-methylenation of methyl propanoate via catalytic dehydrogenation of methanol Patrizia Lorusso,[a] Jacorien Coetzee,[a] Graham R. Eastham[b] and David J. Cole-Hamilton*[a] Abstract: A one-pot system for the conversion of methyl propanoate afford methyl 3-hydroxy-2-methylpropanoate as an intermediate, (MeP) to methyl methacrylate (MMA) has been investigated. In which, in turn, would dehydrate to MMA (Scheme 1). particular, this study is focused on the possibility of performing catalytic dehydrogenation of methanol for the in situ production of anhydrous formaldehyde, which is then consumed in a one-pot base-catalysed condensation with MeP to afford methyl 3-hydroxy-2- methylpropanoate, which spontaneously dehydrogenates to MMA, some of which is subsequently hydrogenated to methyl 2- methypropanoate (MiBu). Scheme 1. Proposed one-pot formation of methyl methacrylate (MMA) from methyl propanote (MeP) and methanol Introduction Alpha methylenation of simple esters can be achieved in low Formaldehyde is a chemical used widely in several industrial yield using condensation of the ester with formaldehyde processes including the manufacture of building materials. A catalysed by caesium oxide on silica at high temperature[3], or by remarkable example in this area is represented by the innovative a complex series of reactions using Meldrum’s acid two-step Alpha technology developed by Lucite for the large Eschemoser’s iodide salt (dimethylmethyleneimmonium scale production of methyl methacrylate (MMA), the essential iodide).[4] -

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

Assessment of Portable HAZMAT Sensors for First Responders
The author(s) shown below used Federal funds provided by the U.S. Department of Justice and prepared the following final report: Document Title: Assessment of Portable HAZMAT Sensors for First Responders Author(s): Chad Huffman, Ph.D., Lars Ericson, Ph.D. Document No.: 246708 Date Received: May 2014 Award Number: 2010-IJ-CX-K024 This report has not been published by the U.S. Department of Justice. To provide better customer service, NCJRS has made this Federally- funded grant report available electronically. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. Department of Justice. Assessment of Portable HAZMAT Sensors for First Responders DOJ Office of Justice Programs National Institute of Justice Sensor, Surveillance, and Biometric Technologies (SSBT) Center of Excellence (CoE) March 1, 2012 Submitted by ManTech Advanced Systems International 1000 Technology Drive, Suite 3310 Fairmont, West Virginia 26554 Telephone: (304) 368-4120 Fax: (304) 366-8096 Dr. Chad Huffman, Senior Scientist Dr. Lars Ericson, Director UNCLASSIFIED This project was supported by Award No. 2010-IJ-CX-K024, awarded by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice. The opinions, findings, and conclusions or recommendations expressed in this publication are those of the author(s) and do not necessarily reflect those of the Department of Justice. This document is a research report submitted to the U.S. Department of Justice. This report has not been published by the Department. Opinions or points of view expressed are those of the author(s) and do not necessarily reflect the official position or policies of the U.S. -

Methyl Methacrylate
METHY METHACRYLATE This substance was considered by a previous Working Group, in February 1978 (IARC, 1979). Since that time, new data have become available, and these have been incorporated into the monograph and taken into consideration in the present evaluation. 1. Exposure Data 1.1 Chemical and physical data 1.1.1 Nomenclature Chem. Abstr. Sem Reg. No.: 80-62-6 Chem. Abstr. Name: 2-Methyl-2-propenoic acid, methyl ester ¡UPAC Systematic Name: Methacrylic acid, methyl ester Synonyms: 2-(Methoxycarbonyl)-1-propene; methyl 2-methylacrylate; methyl 2-methyl- 2-propenoate; MMA 1.1.2 Structural and molecular formulae and relative molecular mass o Il H2C = c- C- 0- CH3 1 CH3 CsHS02 Relative molecular mass: 100.12 1.1.3 Chemical and physical properties of the pure substance (a) Description: Colourless liquid (CYRO Industries, 1987), with a fruity, pungent odour (American Conference of Governmental Industrial Hygienists, 1991) (b) Boiling-point: 100-101 °C (Lide, 1991) (c) Meltng-point: -48 °C (Lide, 1991) (d) Density: 0.9440 at 20 °C/4 °C (Lide, 1991) (e) Spectroscopy data: Infrared (2226), ultraviolet, nucIear magnetic resonance and mass spectral data have been reported (Sadtler Research Laboratories, 1991; US National Library of Medicine, 1993a). if Solubilty: Slightly soluble in water (1.6 gllOO ml at 20 °C), glycerine and ethylene glycol (CYRO Industries, 1987; Bauer, 1990); soluble in acetone, diethyl ether and ethanol (Lide, 1991) -445- 446 IARC MONOGRAPHS VOLUME 60 (g) Volatility: Vapour pressure, 3.87 kPa at 20°C (Ba uer, 1990; Rohm & Haas Co., 1993); relative vapour density (air = 1),3.45 (Verschueren, 1983) (h) Stability: Highly inflammable vapours (Mannsville Chemical Products Corp., 1987); lower explosive limit, 2.1 voL. -
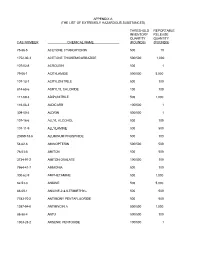
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

Cyanohydrin - Wikipedia, the Free Encyclopedia
Cyanohydrin - Wikipedia, the free encyclopedia http://en.wikipedia.org/wiki/Cyanohydrin Cyanohydrin From Wikipedia, the free encyclopedia A cyanohydrin is a functional group found in organic compounds. Cyanohydrins have the formula R2C(OH)CN, where R is H, alkyl, or aryl. Cyanohydrins are industrially important precursors to carboxylic acids and some amino acids. Cyanohydrins can be formed by the cyanohydrin reaction, which involves treating a ketone or an aldehyde with hydrogen cyanide (HCN) in the presence of excess amounts of sodium cyanide (NaCN) as a catalyst: The structure of a general RR’C=O + HCN → RR’C(OH)CN cyanohydrin. In this reaction, the nucleophilic CN− ion attacks the electrophilic carbonyl carbon in the ketone, followed by protonation by HCN, thereby regenerating the cyanide anion. Cyanohydrins are also prepared by displacement of sulfite by cyanide salts:[1] Cyanohydrins are intermediates in the Strecker amino acid synthesis. Contents 1 Acetone cyanohydrins 2 Other cyanohydrins 3 References 4 External links Acetone cyanohydrins Acetone cyanohydrin, (CH3)2C(OH)CN is the cyanohydrin of acetone. It is generated as an intermediate in the industrial production of methyl methacrylate.[2] In the laboratory, this liquid serves as a source of HCN, which is inconveniently volatile.[3] Thus, acetone cyanohydrin can be used for the preparation other cyanohydrins, for of HCN to Michael acceptors, and for the formylation of arenes. Treatment of this cyanohydrin with lithium hydride affords anhydrous lithium cyanide: 1 of 3 6/3/10 6:07 PM Cyanohydrin - Wikipedia, the free encyclopedia http://en.wikipedia.org/wiki/Cyanohydrin Other cyanohydrins Mandelonitrile, with the formula C6H5CH(OH)CN, occurs in small amounts in the pits of some fruits.[1] Related cyanogenic glycosides are known, such as amygdalin. -

Process for Producing Methyl Methacrylate Verfahren Zur Herstellung Von Methylmethacrylat Procede Pour La Fabrication De Methacrylate De Methyle
~™ II 1 1 III II II 1 1 II II I Ml II I II I II (19) J European Patent Office Office europeen des brevets (11) EP 0 406 676 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publicationation and mention (51 ) |nt. CI.6: C07C 69/54, C07C 67/20 of the grant of the patent: 27.03.1996 Bulletin 1996/13 (21) Application number: 90112194.7 (22) Date of filing: 27.06.1990 (54) Process for producing methyl methacrylate Verfahren zur Herstellung von Methylmethacrylat Procede pour la fabrication de methacrylate de methyle (84) Designated Contracting States: • Ebata, Shuji, DE ES FR GB IT NL C/o Mitsubishi Gas Chem. Com. Tayuhama, Niigata-shi, Niigata-ken (JP) (30) Priority: 04.07.1989 JP 171190/89 (74) Representative: Turk, Gille, Hrabal, Leifert (43) Date of publication of application: Brucknerstrasse 20 09.01.1991 Bulletin 1991/02 D-40593 Dusseldorf (DE) (73) Proprietor: MITSUBISHI GAS CHEMICAL (56) References cited: COMPANY, INC. DE-A- 3 436 608 Chiyoda-ku, Tokyo (JP) • PATENT ABSTRACTS OF JAPAN vol. 14, no. 68 (72) Inventors: (C- 686)(401 1 ), 8 February 1 990; & JP-A-1 290653 • Higuchi, Hirofumi, (MITSUBISHI GAS CHEM) 22.11. 1989 C/o Mitsubishi Gas Chem. Com. Tayuhama, Niigata-shi, Niigata-ken (JP) Remarks: • Kida, Koichi, The file contains technical information submitted C/o Mitsubishi Gas Chem. Com. after the application was filed and not included in this Tayuhama, Niigata-shi, Niigata-ken (JP) specification CO CO CO o Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. -

Provisional Peer-Reviewed Toxicity Values for Acetone Cyanohydrin (Casrn 75-86-5)
EPA/690/R-12/001F l Final 10-01-2012 Provisional Peer-Reviewed Toxicity Values for Acetone Cyanohydrin (CASRN 75-86-5) Superfund Health Risk Technical Support Center National Center for Environmental Assessment Office of Research and Development U.S. Environmental Protection Agency Cincinnati, OH 45268 AUTHORS, CONTRIBUTORS, AND REVIEWERS CHEMICAL MANAGER Scott C. Wesselkamper, PhD National Center for Environmental Assessment, Cincinnati, OH DRAFT DOCUMENT PREPARED BY ICF International 9300 Lee Highway Fairfax, VA 22031 PRIMARY INTERNAL REVIEWERS Zheng (Jenny) Li, PhD, DABT National Center for Environmental Assessment, Washington, DC Anuradha Mudipalli, MSc, PhD National Center for Environmental Assessment, Research Triangle Park, NC This document was externally peer reviewed under contract to Eastern Research Group, Inc. 110 Hartwell Avenue Lexington, MA 02421-3136 Questions regarding the contents of this document may be directed to the U.S. EPA Office of Research and Development’s National Center for Environmental Assessment, Superfund Health Risk Technical Support Center (513-569-7300). ii Acetone cyanohydrin TABLE OF CONTENTS COMMONLY USED ABBREVIATIONS ................................................................................... iv BACKGROUND .............................................................................................................................1 DISCLAIMERS ...............................................................................................................................1 QUESTIONS REGARDING -

Methyl Methacrylate
Health Council of the Netherlands Methyl methacrylate Health-based recommended occupational exposure limit Gezondheidsraad Health Council of the Netherlands Aan de staatssecretaris van Sociale Zaken en Werkgelegenheid Onderwerp : aanbieding advies over Methyl methacrylate Uw kenmerk : DGA/G/TOS/93/07732A Ons kenmerk : U-6899/JR/bp/459-U66 Bijlagen : 1 Datum : 16 december 2011 Geachte staatssecretaris, Graag bied ik u hierbij aan het advies over de gevolgen van beroepsmatige blootstelling aan methylmethacrylaat. Dit advies maakt deel uit van een uitgebreide reeks, waarin gezondheidskundige advies- waarden worden afgeleid voor concentraties van stoffen op de werkplek. Het genoemde advies is opgesteld door de Commissie Gezondheid en beroepsmatige blootstelling aan stoffen (GBBS) van de Gezondheidsraad en beoordeeld door de Beraadsgroep Gezondheid en omgeving. Ik heb dit advies vandaag ter kennisname toegezonden aan de staatssecretaris van Infra- structuur en Milieu en aan de minister van Volksgezondheid, Welzijn en Sport. Met vriendelijke groet, prof. dr. L.J. Gunning-Schepers, voorzitter Bezoekadres Postadres Parnassusplein 5 Postbus 16052 2511 VX Den Haag 2500 BB Den Haag Telefoon (070) 340 70 04 www.gr.nl E-mail: [email protected] Methyl methacrylate Health-based recommended occupational exposure limit Dutch Expert Committee on Occupational Safety A Committee of the Health Council of the Netherlands to: the State Secretary of Social Affairs and Employment No. 2011/38, The Hague, December 16, 2011 The Health Council of the Netherlands, established in 1902, is an independent scientific advisory body. Its remit is “to advise the government and Parliament on the current level of knowledge with respect to public health issues and health (services) research...” (Section 22, Health Act). -
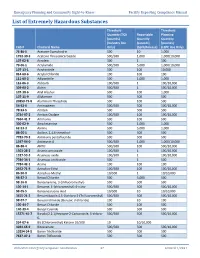
List of Extremely Hazardous Substances
Emergency Planning and Community Right-to-Know Facility Reporting Compliance Manual List of Extremely Hazardous Substances Threshold Threshold Quantity (TQ) Reportable Planning (pounds) Quantity Quantity (Industry Use (pounds) (pounds) CAS # Chemical Name Only) (Spill/Release) (LEPC Use Only) 75-86-5 Acetone Cyanohydrin 500 10 1,000 1752-30-3 Acetone Thiosemicarbazide 500/500 1,000 1,000/10,000 107-02-8 Acrolein 500 1 500 79-06-1 Acrylamide 500/500 5,000 1,000/10,000 107-13-1 Acrylonitrile 500 100 10,000 814-68-6 Acrylyl Chloride 100 100 100 111-69-3 Adiponitrile 500 1,000 1,000 116-06-3 Aldicarb 100/500 1 100/10,000 309-00-2 Aldrin 500/500 1 500/10,000 107-18-6 Allyl Alcohol 500 100 1,000 107-11-9 Allylamine 500 500 500 20859-73-8 Aluminum Phosphide 500 100 500 54-62-6 Aminopterin 500/500 500 500/10,000 78-53-5 Amiton 500 500 500 3734-97-2 Amiton Oxalate 100/500 100 100/10,000 7664-41-7 Ammonia 500 100 500 300-62-9 Amphetamine 500 1,000 1,000 62-53-3 Aniline 500 5,000 1,000 88-05-1 Aniline, 2,4,6-trimethyl- 500 500 500 7783-70-2 Antimony pentafluoride 500 500 500 1397-94-0 Antimycin A 500/500 1,000 1,000/10,000 86-88-4 ANTU 500/500 100 500/10,000 1303-28-2 Arsenic pentoxide 100/500 1 100/10,000 1327-53-3 Arsenous oxide 100/500 1 100/10,000 7784-34-1 Arsenous trichloride 500 1 500 7784-42-1 Arsine 100 100 100 2642-71-9 Azinphos-Ethyl 100/500 100 100/10,000 86-50-0 Azinphos-Methyl 10/500 1 10/10,000 98-87-3 Benzal Chloride 500 5,000 500 98-16-8 Benzenamine, 3-(trifluoromethyl)- 500 500 500 100-14-1 Benzene, 1-(chloromethyl)-4-nitro- 500/500 -

Crosslinking Kinetics in Emulsion Copolymerization of Methyl Methacrylate/Ethylene Glycol Dimethacrylate
Crosslinking kinetics in emulsion copolymerization of methyl methacrylate/ethylene glycol dimethacrylate Hidetaka Tobita*, Kazuhiro Kimura, Kazumi Fujita and Mamoru Nomura Department of Materials Science and Engineering, Fukui University, Fukui, 910 Japan (Received 29 June 1992; revised 24 August 1992) A newly developed kinetic model for network structure development during emulsion crosslinking copolymerization of vinyl and divinyl monomer is used to investigate the kinetics of emulsion crosslinking copolymerization of methyl methacrylate and ethylene glycol dimethacrylate. The agreement of predicted with measured development of crosslinking density and that of monomer concentration in the polymer particles is found to be satisfactory. It is shown that the crosslinking density is fairly high even from very early stages of polymerization and that the produced polymeric networks tend to be highly heterogeneous. The kinetics of crosslinking and resulting network structure are essentially different from those for polymerizations in homogeneous media such as bulk and solution polymerization. (Keywords: polymeric network; gel; mathematical model; crosslinking density distribution; free-radical copolymerization) INTRODUCTION reactions in free-radical copolymerization. This theory The kinetics of crosslinking in emulsion polymerization proves the existence and permits calculation of the have received very little attention from academic and crosslinking density distribution, although most statistical industrial researchers, considering the ever-growing