Hemophilia a and B (Factor VIII (8) and Factor IX (9)
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Guideline on Clinical Investigation of Recombinant and Human Plasma-Derived 9 Factor IX Products’ (EMA/CHMP/BPWP/144552/2009 Rev
1 15 November 2018 2 EMA/CHMP/BPWP/144552/2009 rev. 2 Corr. 1 3 Committee for medicinal products for human use (CHMP) 4 Guideline on clinical investigation of recombinant and 5 human plasma-derived factor IX products 6 Draft Draft Agreed by Blood Products Working Party (BPWP) August 2018 Adopted by Committee for Medicinal Products for Human Use (CHMP) 15 November 2018 Start of public consultation 3 December 2018 End of public consultation 30 June 2019 7 8 This guideline replaces ‘Guideline on clinical investigation of recombinant and human plasma-derived 9 factor IX products’ (EMA/CHMP/BPWP/144552/2009 Rev. 1, Corr. 1) 10 Comments should be provided using this template. The completed comments form should be sent to [email protected] 11 Keywords Recombinant factor IX, plasma-derived factor IX, efficacy, safety, immunogenicity, inhibitor, thrombogenicity, anaphylactic reactions, potency assays 30 Churchill Place ● Canary Wharf ● London E14 5EU ● United Kingdom Telephone +44 (0)20 3660 6000 Facsimile +44 (0)20 3660 5555 Send a question via our website www.ema.europa.eu/contact An agency of the European Union © European Medicines Agency, 2019. Reproduction is authorised provided the source is acknowledged. 12 Guideline on the clinical investigation of recombinant and 13 human plasma-derived factor IX products 14 Table of contents 15 Executive summary ..................................................................................... 4 16 1. Introduction (background) ..................................................................... -

MONONINE (“Difficulty ® Monoclonal Antibody Purified in Concentrating”; Subject Recovered)
CSL Behring IU/kg (n=38), 0.98 ± 0.45 K at doses >95-115 IU/kg (n=21), 0.70 ± 0.38 K at doses >115-135 IU/kg (n=2), 0.67 K at doses >135-155 IU/kg (n=1), and 0.73 ± 0.34 K at doses >155 IU/kg (n=5). Among the 36 subjects who received these high doses, only one (2.8%) Coagulation Factor IX (Human) reported an adverse experience with a possible relationship to MONONINE (“difficulty ® Monoclonal Antibody Purified in concentrating”; subject recovered). In no subjects were thrombo genic complications MONONINE observed or reported.4 only The manufacturing procedure for MONONINE includes multiple processing steps that DESCRIPTION have been designed to reduce the risk of virus transmission. Validation studies of the Coagulation Factor IX (Human), MONONINE® is a sterile, stable, lyophilized concentrate monoclonal antibody (MAb) immunoaffinity chromatography/chemical treatment step and of Factor IX prepared from pooled human plasma and is intended for use in therapy nanofiltration step used in the production of MONONINE doc ument the virus reduction of Factor IX deficiency, known as Hemophilia B or Christmas disease. MONONINE is capacity of the processes employed. These studies were conducted using the rel evant purified of extraneous plasma-derived proteins, including Factors II, VII and X, by use of enveloped and non-enveloped viruses. The results of these virus validation studies utilizing immunoaffinity chromatography. A murine monoclonal antibody to Factor IX is used as an a wide range of viruses with different physicochemical properties are summarized in Table affinity ligand to isolate Factor IX from the source material. -

Familial Multiple Coagulation Factor Deficiencies
Journal of Clinical Medicine Article Familial Multiple Coagulation Factor Deficiencies (FMCFDs) in a Large Cohort of Patients—A Single-Center Experience in Genetic Diagnosis Barbara Preisler 1,†, Behnaz Pezeshkpoor 1,† , Atanas Banchev 2 , Ronald Fischer 3, Barbara Zieger 4, Ute Scholz 5, Heiko Rühl 1, Bettina Kemkes-Matthes 6, Ursula Schmitt 7, Antje Redlich 8 , Sule Unal 9 , Hans-Jürgen Laws 10, Martin Olivieri 11 , Johannes Oldenburg 1 and Anna Pavlova 1,* 1 Institute of Experimental Hematology and Transfusion Medicine, University Clinic Bonn, 53127 Bonn, Germany; [email protected] (B.P.); [email protected] (B.P.); [email protected] (H.R.); [email protected] (J.O.) 2 Department of Paediatric Haematology and Oncology, University Hospital “Tzaritza Giovanna—ISUL”, 1527 Sofia, Bulgaria; [email protected] 3 Hemophilia Care Center, SRH Kurpfalzkrankenhaus Heidelberg, 69123 Heidelberg, Germany; ronald.fi[email protected] 4 Department of Pediatrics and Adolescent Medicine, University Medical Center–University of Freiburg, 79106 Freiburg, Germany; [email protected] 5 Center of Hemostasis, MVZ Labor Leipzig, 04289 Leipzig, Germany; [email protected] 6 Hemostasis Center, Justus Liebig University Giessen, 35392 Giessen, Germany; [email protected] 7 Center of Hemostasis Berlin, 10789 Berlin-Schöneberg, Germany; [email protected] 8 Pediatric Oncology Department, Otto von Guericke University Children’s Hospital Magdeburg, 39120 Magdeburg, Germany; [email protected] 9 Division of Pediatric Hematology Ankara, Hacettepe University, 06100 Ankara, Turkey; Citation: Preisler, B.; Pezeshkpoor, [email protected] B.; Banchev, A.; Fischer, R.; Zieger, B.; 10 Department of Pediatric Oncology, Hematology and Clinical Immunology, University of Duesseldorf, Scholz, U.; Rühl, H.; Kemkes-Matthes, 40225 Duesseldorf, Germany; [email protected] B.; Schmitt, U.; Redlich, A.; et al. -

081999 Disseminated Intravascular Coagulation
The New England Journal of Medicine Current Concepts Systemic activation+ of coagulation DISSEMINATED INTRAVASCULAR COAGULATION Intravascular+ Depletion of platelets+ deposition of fibrin and coagulation factors MARCEL LEVI, M.D., AND HUGO TEN CATE, M.D. Thrombosis of small+ Bleeding and midsize vessels+ ISSEMINATED intravascular coagulation is and organ failure characterized by the widespread activation Dof coagulation, which results in the intravas- Figure 1. The Mechanism of Disseminated Intravascular Coag- cular formation of fibrin and ultimately thrombotic ulation. occlusion of small and midsize vessels.1-3 Intravascu- Systemic activation of coagulation leads to widespread intra- lar coagulation can also compromise the blood sup- vascular deposition of fibrin and depletion of platelets and co- agulation factors. As a result, thrombosis of small and midsize ply to organs and, in conjunction with hemodynam- vessels may occur, contributing to organ failure, and there may ic and metabolic derangements, may contribute to be severe bleeding. the failure of multiple organs. At the same time, the use and subsequent depletion of platelets and coag- ulation proteins resulting from the ongoing coagu- lation may induce severe bleeding (Fig. 1). Bleeding may be the presenting symptom in a patient with disseminated intravascular coagulation, a factor that can complicate decisions about treatment. TABLE 1. COMMON CLINICAL CONDITIONS ASSOCIATED WITH DISSEMINATED ASSOCIATED CLINICAL CONDITIONS INTRAVASCULAR COAGULATION. AND INCIDENCE Sepsis Infectious Disease Trauma Serious tissue injury Disseminated intravascular coagulation is an ac- Head injury Fat embolism quired disorder that occurs in a wide variety of clin- Cancer ical conditions, the most important of which are listed Myeloproliferative diseases in Table 1. -

Gene Therapy Expression Vectors Based on the Clotting Factor IX Promoter
Gene Therapy (1999) 6, 1584–1589 1999 Stockton Press All rights reserved 0969-7128/99 $15.00 http://www.stockton-press.co.uk/gt Gene therapy expression vectors based on the clotting Factor IX promoter H Hoag, J Gore, D Barry and CR Mueller Department of Biochemistry and Cancer Research Laboratories, Queen’s University, Kingston, Ontario, Canada The liver is one of the prime targets for gene therapy, and moter. Introduction of this element increases promoter the correction of defects in a variety of clotting factor genes activity at least 20-fold over the proximal promoter alone is one of the main goals of liver-directed therapies. The use when assayed in the human liver cell line Hep G2. This of transcriptional regulatory elements derived from these optimized promoter is significantly more active than the genes may provide for the optimal expression of trans- SV40 enhancer/early promoter. The expression of the opti- duced genes. We have applied our knowledge of the pro- mized Factor IX promoter is also more persistent in the moter structure of the clotting Factor IX gene to design short term. The inclusion of a liver-specific locus control optimized expression vectors for use in gene therapy. The region, derived from the apolipoprotein E/C locus, did not activity of the proximal promoter has been augmented by further augment expression levels. These Factor IX vectors the introduction of a multimerized upstream site which we also exhibit a high degree of tissue specificity, as meas- have previously shown to be a prime regulator of the pro- ured by transfection into breast and muscle cell lines. -

What Everyone Should Know to Stop Bleeding After an Injury
What Everyone Should Know to Stop Bleeding After an Injury THE HARTFORD CONSENSUS The Joint Committee to Increase Survival from Active Shooter and Intentional Mass Casualty Events was convened by the American College of Surgeons in response to the growing number and severity of these events. The committee met in Hartford Connecticut and has produced a number of documents with rec- ommendations. The documents represent the consensus opinion of a multi-dis- ciplinary committee involving medical groups, the military, the National Security Council, Homeland Security, the FBI, law enforcement, fire rescue, and EMS. These recommendations have become known as the Hartford Consensus. The overarching principle of the Hartford Consensus is that no one should die from uncontrolled bleeding. The Hartford Consensus recommends that all citizens learn to stop bleeding. Further information about the Hartford Consensus and bleeding control can be found on the website: Bleedingcontrol.org 2 SAVE A LIFE: What Everyone Should Know to Stop Bleeding After an Injury Authors: Peter T. Pons, MD, FACEP Lenworth Jacobs, MD, MPH, FACS Acknowledgements: The authors acknowledge the contributions of Michael Cohen and James “Brooks” Hart, CMI to the design of this manual. Some images adapted from Adam Wehrle, EMT-P and NAEMT. © 2017 American College of Surgeons CONTENTS SECTION 1 3 ■ Introduction ■ Primary Principles of Trauma Care Response ■ The ABCs of Bleeding SECTION 2 5 ■ Ensure Your Own Safety SECTION 3 6 ■ A – Alert – call 9-1-1 SECTION 4 7 ■ B – Bleeding – find the bleeding injury SECTION 5 9 ■ C – Compress – apply pressure to stop the bleeding by: ■ Covering the wound with a clean cloth and applying pressure by pushing directly on it with both hands, OR ■Using a tourniquet, OR ■ Packing (stuff) the wound with gauze or a clean cloth and then applying pressure with both hands SECTION 6 13 ■ Summary 2 SECTION 1: INTRODUCTION Welcome to the Stop the Bleed: Bleeding Control for the Injured information booklet. -
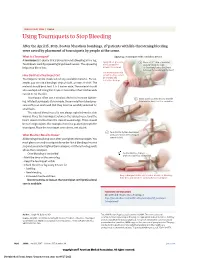
Using Tourniquets to Stop Bleeding
JAMA PATIENT PAGE | Trauma Using Tourniquets to Stop Bleeding After the April 15, 2013, Boston Marathon bombings, 27 patients with life-threatening bleeding were saved by placement of tourniquets by people at the scene. What Is a Tourniquet? Applying a tourniquet with a windlass device A tourniquet is a device that is placed around a bleeding arm or leg. Apply direct pressure 1 Place a 2-3” strip of material Tourniquets work by squeezing large blood vessels. The squeezing to the wound for about 2” from the edge helps stop blood loss. at least 15 minutes. of the wound over a long bone between the wound and the heart. Use a tourniquet only How Do I Put a Tourniquet On? when bleeding cannot be stopped and Tourniquets can be made out of any available material. For ex- is life threatening. ample, you can use a bandage, strip of cloth, or even a t-shirt. The material should be at least 2 to 3 inches wide. The material should also overlap itself. Using thin straps or material less than 2 inches wide can rip or cut the skin. Tourniquets often use a windlass device to increase tighten- 2 Insert a stick or other strong, straight ing. Inflated tourniquets (for example, those made from blood pres- item into the knot to act as a windlass. sure cuffs) can work well. But they must be carefully watched for small leaks. The injured blood vessel is not always right below the skin wound. Place the tourniquet between the injured vessel and the heart, about 2 inches from the closest wound edge. -

A Guide for People Living with Von Willebrand Disorder CONTENTS
A guide for people living with von Willebrand disorder CONTENTS What is von Willebrand disorder (VWD)?................................... 3 Symptoms............................................................................................... 5 Types of VWD...................................................................................... 6 How do you get VWD?...................................................................... 7 VWD and blood clotting.................................................................... 11 Diagnosis................................................................................................. 13 Treatment............................................................................................... 15 Taking care of yourself or your child.............................................. 19 (Education, information, first aid/medical emergencies, medication to avoid) Living well with VWD......................................................................... 26 (Sport, travel, school, telling others, work) Special issues for women and girls.................................................. 33 Connecting with others..................................................................... 36 Can I live a normal life with von Willebrand disorder?............. 37 More information................................................................................. 38 2 WHAT IS VON WILLEBRAND DISORDER (VWD)? Von Willebrand disorder (VWD) is an inherited bleeding disorder. People with VWD have a problem with a protein -

Plasma Levels of Plasminogen Activator Inhibitor Type 1, Factor VIII
Plasma Levels of Plasminogen Activator Inhibitor Type 1, Factor VIII, Prothrombin ,Activation Fragment 1؉2, Anticardiolipin and Antiprothrombin Antibodies are Risk Factors for Thrombosis in Hemodialysis Patients Daniela Molino,*,†,‡ Natale G. De Santo,* Rosa Marotta,*,†,§ Pietro Anastasio,* Mahrokh Mosavat,† and Domenico De Lucia† Patients with end-stage renal disease are prone to hemorrhagic complications and simul- taneously are at risk for a variety of thrombotic complications such as thrombosis of dialysis blood access, the subclavian vein, coronary arteries, cerebral vessel, and retinal veins, as well as priapism. The study was devised for the following purposes: (1) to identify the markers of thrombophilia in hemodialyzed patients, (2) to establish a role for antiphos- pholipid antibodies in thrombosis of the vascular access, (3) to characterize phospholipid antibodies in hemodialysis patients, and (4) to study the effects of dialysis on coagulation cascade. A group of 20 hemodialysis patients with no thrombotic complications (NTC) and 20 hemodialysis patients with thrombotic complications (TC) were studied along with 400 volunteer blood donors. Patients with systemic lupus erythematosus and those with nephrotic syndrome were excluded. All patients underwent a screening prothrombin time, activated partial thromboplastin time, fibrinogen (Fg), coagulation factors of the intrinsic and extrinsic pathways, antithrombin III (AT-III), protein C (PC), protein S (PS), resistance to activated protein C, prothrombin activation fragment 1؉2 (F1؉2), plasminogen, tissue type plasminogen activator (t-PA), plasminogen tissue activator inhibitor type-1 (PAI-1), anticardiolipin antibodies type M and G (ACA-IgM and ACA-IgG), lupus anticoagulant antibodies, and antiprothrombin antibodies type M and G (aPT-IgM and aPT-IgG). -

ER Guide to Bleeding Disorders
Bleeding disorders ER guide to bleeding disorders 1 Table of contents 4 General Guidelines 4–5 national Hemophilia Foundation guidelines 5–10 Treatment options 10 HemopHilia a Name:__________________________________________________________________________________________________ 10–11 national Hemophilia Foundation guidelines Address:________________________________________________________________________________________________ 12 dosage chart Phone:__________________________________________________________________________________________________ 14–15 Treatment products 16 HemopHilia B In case of emergency, contact: ______________________________________________________________________________ 16 national Hemophilia Foundation guidelines Relation to patient:________________________________________________________________________________________ 17 dosage chart 18 Treatment products 19 HemopHilia a or B with inHiBiTors Diagnosis: Hemophilia A: Mild Moderate Severe 20 national Hemophilia Foundation guidelines Inhibitors Inhibitors Bethesda units (if known) ____________________________________ 21 Treatment products Hemophilia B: Mild Moderate Severe 22–23 Von willeBrand disease Inhibitors Inhibitors Bethesda units (if known) ____________________________________ 23–24 national Hemophilia Foundation guidelines von Willebrand disease: Type 1 Type 2 Type 3 Platelet type 25 Treatment products 27 Bibliography Preferred product:_________________________________________________________________________________________ Dose for life-threatening -

Phenotypic Correction of Factor IX Deficiency in Skin Fibroblasts
Proc. Nati. Acad. Sci. USA Vol. 87, pp. 5173-5177, July 1990 Genetics Phenotypic correction of factor IX deficiency in skin fibroblasts of hemophilic dogs (molecular cloning/hemophilia B/retroviral vectors/endothelial cells/gene therapy) J. H. AXELROD*, M. S. READt, K. M. BRINKHOUSt, AND 1. M. VERMA*t *Molecular Biology and Virology Laboratory, Salk Institute, Post Office Box 85800, San Diego, CA 92138; and tDepartment of Pathology and Center for Thrombosis and Hemostasis, University of North Carolina, Chapel Hill, NC 27599 Contributed by K. M. Brinkhous, April 24, 1990 ABSTRACT Primary skin fibroblasts from hemophilic endothelial cells as potential targets for gene transfer in the dogs were transduced by recombinant retrovirus (LNCdF9L) treatment of hemophilia B. containing a canine factor IX cDNA. High levels of biologically active canine factor IX (1.0 ,ug per 106 cells per 24 hr) were secreted in the medium. The level of factor IX produced MATERIALS AND METHODS increased substantially if the cells were stimulated by basic Construction and Isolation of a Canine Factor IX cDNA. A fibroblast growth factor during infection. Additionally, we also canine liver cDNA library was constructed using total cel- report that endothelial cells transduced by this virus can lular poly(A)+ RNA isolated from mongrel dog liver. cDNA produce high levels ofbiologically active factor IX. We propose was prepared using a cDNA synthesis kit (Pharmacia) and that skin fibroblasts and endothelial cells from hemophilia B ligated in the vector AZAP (Stratagene), which was packaged dogs may serve as potential venues for the development and with Gigapack Plus (Stratagene) according to the manufac- testing of models for treatment of hemophilia B by retrovirally turer's instructions. -

Factor IX Complex (Human - Bebulin, Profilnine) Reference Number: ERX.SPMN.201 Effective Date: 01/17 Coding Implications Last Review Date: Revision Log
Clinical Policy: Factor IX complex (Human - Bebulin, Profilnine) Reference Number: ERX.SPMN.201 Effective Date: 01/17 Coding Implications Last Review Date: Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Policy/Criteria It is the policy of health plans affiliated with Envolve Pharmacy SolutionsTM that factor IX complex (Bebulin®, Profilnine®) is medically necessary when the following criteria are met: I. Initial Approval Criteria A. Hemophilia B (must meet all): 1. Prescribed by or in consultation with a hematologist; 2. Diagnosis of hemophilia B; 3. Agent will used for prevention/control of bleeding episodes. Approval duration: 3 months B. Other diagnoses/indications: Refer to ERX.SPMN.16 - Global Biopharm Policy. II. Continued Approval A. Hemophilia B (must meet all): 1. Currently receiving medication via health plan benefit or member has previously met all initial approval criteria. Approval duration: 3 months B. Other diagnoses/indications (must meet 1 or 2): 1. Currently receiving medication via health plan benefit and documentation supports positive response to therapy; or 2. Refer to ERX.SPMN.16 - Global Biopharm Policy. Background Description/Mechanism of Action: Factor IX complex replaces deficient clotting factors including factor X. Hemophilia B, or Christmas disease, is an X-linked recessively inherited disorder of blood coagulation characterized by insufficient or abnormal synthesis of the clotting protein factor IX. Factor IX is a vitamin K-dependent coagulation factor which is synthesized in the liver. Factor IX is activated by factor XIa in the intrinsic coagulation pathway. Activated factor IX (IXa), in combination with factor VII: C, activates factor X to Xa, resulting ultimately in the conversion of prothrombin to thrombin and the formation of a fibrin clot.