Volume VI, Technical Aspects of Early Warning and Verification
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Adverse Effects of Organophosphorus Pesticides on the Liver: a Brief Summary of Four Decades of Research
Karami-Mohajeri S, et al. Adverse effects of OPs on the liver: a brief research summary Arh Hig Rada Toksikol 2017;68:261-275 261 Review DOI: 10.1515/aiht-2017-68-2989 Adverse effects of organophosphorus pesticides on the liver: a brief summary of four decades of research Somayyeh Karami-Mohajeri1,2, Ahmad Ahmadipour2, Hamid-Reza Rahimi1,2, and Mohammad Abdollahi3,4 Pharmaceutics Research Center, Institute of Neuropharmacology1, Department of Toxicology and Pharmacology, Faculty of Pharmacy2, Kerman University of Medical Sciences, Kerman, Pharmaceutical Sciences Research Center3, Department of Toxicology and Pharmacology4, Faculty of Pharmacy, Tehran University of Medical Sciences, Tehran, Iran [Received in May 2017; Similarity Check in May 2017; Accepted in December 2017] Organophosphorus pesticides (OPs) are widely used volatile pesticides that have harmful effects on the liver in acute and chronic exposures. This review article summarises and discusses a wide collection of studies published over the last 40 years reporting on the effects of OPs on the liver, in an attempt to propose general mechanisms of OP hepatotoxicity and possible treatment. Several key biological processes have been reported as involved in OP-induced hepatotoxicity such as disturbances in the antioxidant defence system, oxidative stress, apoptosis, and mitochondrial and microsomal metabolism. Most studies show that antioxidants can attenuate oxidative stress and the consequent changes in liver function. However, few studies have examined the relationship between OP structures and the severity and mechanism of their action. We hope that future in vitro, in vivo, and clinical trials will answer the remaining questions about the mechanisms of OP hepatotoxicity and its management. -

Effect of Chlorpyrifos Oxon on M2 Muscarinic Acetylcholine Receptor Trafficking”
EFFECT OF CHLORPYRIFOS OXON ON M2 MUSCARINIC ACETYLCHOLINE RECEPTOR REGULATION BY ELMAR MABUNGA UDARBE Doctor of Veterinary Medicine University of the Philippines Los Baños College, Laguna, Philippines 1999 Submitted to the Faculty of the Graduate College of Oklahoma State University in partial fulfillment of the requirements for the Degree of MASTER OF SCIENCE July, 2004 EFFECT OF CHLORPYRIFOS OXON ON M2 MUSCARINIC ACETYLCHOLINE RECEPTOR REGULATION Thesis Approved: DR. CAREY N. POPE Thesis Advisor DR. CYRIL C. CLARKE DR. CHARLOTTE C. OWNBY DR. DORIS K. PATNEAU DR. AL CARLOZI Dean of Graduate College ii ACKNOWLEDGMENTS My sincerest gratitude goes to my major advisor, Dr. Carey N. Pope for the intelligent supervision, for providing inspiration to do this work. I am also thankful to my committee members, Dr. Cyril Clarke, Dr. Charlotte Ownby and Dr. Doris Patneau for helpful comments on the content and form of this manuscript. I am indebted to the Fulbright-Philippine Agriculture Scholarship Program (FPASP) and the Philippine American Education Foundation (PAEF) whose exchange program deepened my understanding of the U.S. culture and its people and allowed me to promote mutual understanding between the U.S. and the Philippines. I am grateful to the University of the Philippines in Mindanao (UPMINDANAO) for supporting my pursuit for graduate studies, the National Institute of Environmental Health Sciences (NIEHS), Oklahoma State University Board of Regents and Dr. Sidney Ewing, Wendell H. and Nellie G. Krull Endowed professor for the financial assistance. I am also thankful to the following: Ms. Sharon Baker for doing the preliminary work on the project; Dr. -

Approach to Site-Selective Protein Modification JUSTIN M
A “Tag-and-Modify” Approach to Site-Selective Protein Modification JUSTIN M. CHALKER, GONC-ALO J. L. BERNARDES, AND BENJAMIN G. DAVIS* Department of Chemistry, University of Oxford, Chemistry Research Laboratory, 12 Mansfield Road, Oxford OX1 3TA, United Kingdom RECEIVED ON FEBRUARY 27, 2011 CONSPECTUS ovalent modification can expand a protein's functional capacity. Fluorescent or radioactive labeling, for instance, allows C imaging of a protein in real time. Labeling with an affinity probe enables isolation of target proteins and other interacting molecules. At the other end of this functional spectrum, protein structures can be naturally altered by enzymatic action. ProteinÀprotein interactions, genetic regulation, and a range of cellular processes are under the purview of these post- translational modifications. The ability of protein chemists to install these covalent additions selectively has been critical for elucidating their roles in biology. Frequently the transformations must be applied in a site-specific manner, which demands the most selective chemistry. In this Account, we discuss the development and application of such chemistry in our laboratory. A centerpiece of our strategy is a “tag-and-modify” approach, which entails sequential installation of a uniquely reactive chemical group into the protein (the “tag”) and the selective or specific modification of this group. The chemical tag can be a natural or unnatural amino acid residue. Of the natural residues, cysteine is the most widely used as a tag. Early work in our program focused on selective disulfide formation in the synthesis of glycoproteins. For certain applications, the susceptibility of disulfides to reduction was a limitation and prompted the development of several methods for the synthesis of more stable thioether modifications. -

Chemical Name Federal P Code CAS Registry Number Acutely
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extremely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extremely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extremely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extremely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extremely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extremely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extremely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extremely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extremely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extremely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extremely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Ncomms12703.Pdf
ARTICLE Received 17 Feb 2016 | Accepted 26 Jul 2016 | Published 2 Sep 2016 DOI: 10.1038/ncomms12703 OPEN Chemoselective synthesis and analysis of naturally occurring phosphorylated cysteine peptides Jordi Bertran-Vicente1, Martin Penkert1,2, Olaia Nieto-Garcia1,2, Jean-Marc Jeckelmann3, Peter Schmieder1, Eberhard Krause1 & Christian P. R. Hackenberger1,2 In contrast to protein O-phosphorylation, studying the function of the less frequent N- and S-phosphorylation events have lagged behind because they have chemical features that prevent their manipulation through standard synthetic and analytical methods. Here we report on the development of a chemoselective synthetic method to phosphorylate Cys side-chains in unprotected peptides. This approach makes use of a reaction between nucleophilic phosphites and electrophilic disulfides accessible by standard methods. We achieve the stereochemically defined phosphorylation of a Cys residue and verify the modification using electron-transfer higher-energy dissociation (EThcD) mass spectrometry. To demonstrate the use of the approach in resolving biological questions, we identify an endogenous Cys phosphorylation site in IICBGlc, which is known to be involved in the carbohydrate uptake from the bacterial phosphotransferase system (PTS). This new chemical and analytical approach finally allows further investigating the functions and significance of Cys phosphorylation in a wide range of crucial cellular processes. 1 Leibniz-Institut fu¨r Molekulare Pharmakologie (FMP), Robert-Ro¨ssle-Str. 10, D-13125 Berlin, Germany. 2 Humboldt-Universita¨t zu Berlin, Institut fu¨r Chemie, Brook-Taylor-Str. 2, D-12489 Berlin, Germany. 3 Institute of Biochemistry and Molecular Medicine, University of Bern, 3012 Bern, Switzerland. Correspondence and requests for materials should be addressed to C.P.R.H. -
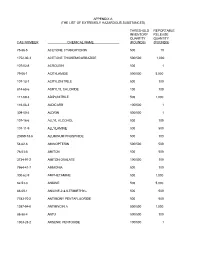
The List of Extremely Hazardous Substances)
APPENDIX A (THE LIST OF EXTREMELY HAZARDOUS SUBSTANCES) THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 75-86-5 ACETONE CYANOHYDRIN 500 10 1752-30-3 ACETONE THIOSEMICARBAZIDE 500/500 1,000 107-02-8 ACROLEIN 500 1 79-06-1 ACRYLAMIDE 500/500 5,000 107-13-1 ACRYLONITRILE 500 100 814-68-6 ACRYLYL CHLORIDE 100 100 111-69-3 ADIPONITRILE 500 1,000 116-06-3 ALDICARB 100/500 1 309-00-2 ALDRIN 500/500 1 107-18-6 ALLYL ALCOHOL 500 100 107-11-9 ALLYLAMINE 500 500 20859-73-8 ALUMINUM PHOSPHIDE 500 100 54-62-6 AMINOPTERIN 500/500 500 78-53-5 AMITON 500 500 3734-97-2 AMITON OXALATE 100/500 100 7664-41-7 AMMONIA 500 100 300-62-9 AMPHETAMINE 500 1,000 62-53-3 ANILINE 500 5,000 88-05-1 ANILINE,2,4,6-TRIMETHYL- 500 500 7783-70-2 ANTIMONY PENTAFLUORIDE 500 500 1397-94-0 ANTIMYCIN A 500/500 1,000 86-88-4 ANTU 500/500 100 1303-28-2 ARSENIC PENTOXIDE 100/500 1 THRESHOLD REPORTABLE INVENTORY RELEASE QUANTITY QUANTITY CAS NUMBER CHEMICAL NAME (POUNDS) (POUNDS) 1327-53-3 ARSENOUS OXIDE 100/500 1 7784-34-1 ARSENOUS TRICHLORIDE 500 1 7784-42-1 ARSINE 100 100 2642-71-9 AZINPHOS-ETHYL 100/500 100 86-50-0 AZINPHOS-METHYL 10/500 1 98-87-3 BENZAL CHLORIDE 500 5,000 98-16-8 BENZENAMINE, 3-(TRIFLUOROMETHYL)- 500 500 100-14-1 BENZENE, 1-(CHLOROMETHYL)-4-NITRO- 500/500 500 98-05-5 BENZENEARSONIC ACID 10/500 10 3615-21-2 BENZIMIDAZOLE, 4,5-DICHLORO-2-(TRI- 500/500 500 FLUOROMETHYL)- 98-07-7 BENZOTRICHLORIDE 100 10 100-44-7 BENZYL CHLORIDE 500 100 140-29-4 BENZYL CYANIDE 500 500 15271-41-7 BICYCLO[2.2.1]HEPTANE-2-CARBONITRILE,5- -

744 Hydrolysis of Chiral Organophosphorus Compounds By
[Frontiers in Bioscience, Landmark, 26, 744-770, Jan 1, 2021] Hydrolysis of chiral organophosphorus compounds by phosphotriesterases and mammalian paraoxonase-1 Antonio Monroy-Noyola1, Damianys Almenares-Lopez2, Eugenio Vilanova Gisbert3 1Laboratorio de Neuroproteccion, Facultad de Farmacia, Universidad Autonoma del Estado de Morelos, Morelos, Mexico, 2Division de Ciencias Basicas e Ingenierias, Universidad Popular de la Chontalpa, H. Cardenas, Tabasco, Mexico, 3Instituto de Bioingenieria, Universidad Miguel Hernandez, Elche, Alicante, Spain TABLE OF CONTENTS 1. Abstract 2. Introduction 2.1. Organophosphorus compounds (OPs) and their toxicity 2.2. Metabolism and treatment of OP intoxication 2.3. Chiral OPs 3. Stereoselective hydrolysis 3.1. Stereoselective hydrolysis determines the toxicity of chiral compounds 3.2. Hydrolysis of nerve agents by PTEs 3.2.1. Hydrolysis of V-type agents 3.3. PON1, a protein restricted in its ability to hydrolyze chiral OPs 3.4. Toxicity and stereoselective hydrolysis of OPs in animal tissues 3.4.1. The calcium-dependent stereoselective activity of OPs associated with PON1 3.4.2. Stereoselective hydrolysis commercial OPs pesticides by alloforms of PON1 Q192R 3.4.3. PON1, an enzyme that stereoselectively hydrolyzes OP nerve agents 3.4.4. PON1 recombinants and stereoselective hydrolysis of OP nerve agents 3.5. The activity of PTEs in birds 4. Conclusions 5. Acknowledgments 6. References 1. ABSTRACT Some organophosphorus compounds interaction of the racemic OPs with these B- (OPs), which are used in the manufacturing of esterases (AChE and NTE) and such interactions insecticides and nerve agents, are racemic mixtures have been studied in vivo, ex vivo and in vitro, using with at least one chiral center with a phosphorus stereoselective hydrolysis by A-esterases or atom. -

Organophosphate Insecticides
CHAPTER 4 HIGHLIGHTS Organophosphate Insecticides Acts through phosphorylation of the acetylcholinesterase enzyme Since the removal of organochlorine insecticides from use, organophosphate at nerve endings insecticides have become the most widely used insecticides available today. More Absorbed by inhalation, than forty of them are currently registered for use and all run the risk of acute ingestion, and skin and subacute toxicity. Organophosphates are used in agriculture, in the home, penetration in gardens, and in veterinary practice. All apparently share a common mecha- Muscarinic, nicotinic & CNS nism of cholinesterase inhibition and can cause similar symptoms. Because they effects share this mechanism, exposure to the same organophosphate by multiple routes or to multiple organophosphates by multiple routes can lead to serious additive Signs and Symptoms: toxicity. It is important to understand, however, that there is a wide range of Headache, hypersecretion, toxicity in these agents and wide variation in cutaneous absorption, making muscle twitching, nausea, specific identification and management quite important. diarrhea Respiratory depression, seizures, loss of consciousness Toxicology Miosis is often a helpful Organophosphates poison insects and mammals primarily by phosphory- diagnostic sign lation of the acetylcholinesterase enzyme (AChE) at nerve endings. The result is a loss of available AChE so that the effector organ becomes overstimulated by Treatment: the excess acetylcholine (ACh, the impulse-transmitting substance) in the nerve Clear airway, improve tissue ending. The enzyme is critical to normal control of nerve impulse transmission oxygenation from nerve fibers to smooth and skeletal muscle cells, glandular cells, and Administer atropine sulfate autonomic ganglia, as well as within the central nervous system (CNS). -

Asymmetric Synthesis of Organophosphates and Their
ASYMMETRIC SYNTHESIS OF ORGANOPHOSPHATES AND THEIR DERIVATIVES Thesis Submitted to The College of Arts and Sciences of the UNIVERSITY OF DAYTON In Partial Fulfillment of the Requirements for The Degree of Master of Science in Chemistry By Batool J. Murtadha Dayton, Ohio May 2020 ASYMMETRIC SYNTHESIS OF ORGANOPHOSPHATES AND THEIR DERIVATIVES Name: Murtadha, Batool J. APPROVED BY: __________________________________ Jeremy Erb, Ph.D. Research Advisor Assistant Professor Department of Chemistry University of Dayton ___________________________________ Vladimir Benin, Ph.D. Professor of Chemistry Department of Chemistry University of Dayton ___________________________________ Justin C. Biffinger, Ph.D. Committee Member Assistant Professor Department of Chemistry University of Dayton ii © Copyright by Batool J. Murtadha All rights reserved 2020 iii ABSTRACT ASYMMETRIC SYNTHESIS OF ORGANOPHOSPHATES AND THEIR DERIVATIVES Name: Murtadha, Batool J. University of Dayton Advisor: Dr. Jeremy Erb Organophosphorus compounds (OPs) are widely used in the agricultural industry especially in the pesticide market. Phosphates play a huge role as biological compounds in the form of energy carrier compounds like ATP, and medicine as antivirals. OPs have become increasingly important as evidenced by the publication of new methods devoted to their uses and synthesis. These well-established studies lay the basis for industrial organic derivatives of phosphorus preparations. The current work explored methods of synthesizing chiral organophosphate triesters. We experimented with different processes roughly divided into either an electrophilic or nucleophilic strategy using chiral Lewis acids, organocatalysts (HyperBTM), activating agents, and chiral auxiliaries with the goal of control stereoselectivity. These methods were explored through the use of different starting materials like POCl3, triethyl phosphate, methyl phosphordichloradate, and PSCl3. -

Acutely / Extremely Hazardous Waste List
Acutely / Extremely Hazardous Waste List Federal P CAS Registry Acutely / Extremely Chemical Name Code Number Hazardous 4,7-Methano-1H-indene, 1,4,5,6,7,8,8-heptachloro-3a,4,7,7a-tetrahydro- P059 76-44-8 Acutely Hazardous 6,9-Methano-2,4,3-benzodioxathiepin, 6,7,8,9,10,10- hexachloro-1,5,5a,6,9,9a-hexahydro-, 3-oxide P050 115-29-7 Acutely Hazardous Methanimidamide, N,N-dimethyl-N'-[2-methyl-4-[[(methylamino)carbonyl]oxy]phenyl]- P197 17702-57-7 Acutely Hazardous 1-(o-Chlorophenyl)thiourea P026 5344-82-1 Acutely Hazardous 1-(o-Chlorophenyl)thiourea 5344-82-1 Extemely Hazardous 1,1,1-Trichloro-2, -bis(p-methoxyphenyl)ethane Extemely Hazardous 1,1a,2,2,3,3a,4,5,5,5a,5b,6-Dodecachlorooctahydro-1,3,4-metheno-1H-cyclobuta (cd) pentalene, Dechlorane Extemely Hazardous 1,1a,3,3a,4,5,5,5a,5b,6-Decachloro--octahydro-1,2,4-metheno-2H-cyclobuta (cd) pentalen-2- one, chlorecone Extemely Hazardous 1,1-Dimethylhydrazine 57-14-7 Extemely Hazardous 1,2,3,4,10,10-Hexachloro-6,7-epoxy-1,4,4,4a,5,6,7,8,8a-octahydro-1,4-endo-endo-5,8- dimethanonaph-thalene Extemely Hazardous 1,2,3-Propanetriol, trinitrate P081 55-63-0 Acutely Hazardous 1,2,3-Propanetriol, trinitrate 55-63-0 Extemely Hazardous 1,2,4,5,6,7,8,8-Octachloro-4,7-methano-3a,4,7,7a-tetra- hydro- indane Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]- 51-43-4 Extemely Hazardous 1,2-Benzenediol, 4-[1-hydroxy-2-(methylamino)ethyl]-, P042 51-43-4 Acutely Hazardous 1,2-Dibromo-3-chloropropane 96-12-8 Extemely Hazardous 1,2-Propylenimine P067 75-55-8 Acutely Hazardous 1,2-Propylenimine 75-55-8 Extemely Hazardous 1,3,4,5,6,7,8,8-Octachloro-1,3,3a,4,7,7a-hexahydro-4,7-methanoisobenzofuran Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime 26419-73-8 Extemely Hazardous 1,3-Dithiolane-2-carboxaldehyde, 2,4-dimethyl-, O- [(methylamino)-carbonyl]oxime. -

Environmental Health Criteria 63 ORGANOPHOSPHORUS
Environmental Health Criteria 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION Please note that the layout and pagination of this web version are not identical with the printed version. Organophophorus insecticides: a general introduction (EHC 63, 1986) INTERNATIONAL PROGRAMME ON CHEMICAL SAFETY ENVIRONMENTAL HEALTH CRITERIA 63 ORGANOPHOSPHORUS INSECTICIDES: A GENERAL INTRODUCTION This report contains the collective views of an international group of experts and does not necessarily represent the decisions or the stated policy of the United Nations Environment Programme, the International Labour Organisation, or the World Health Organization. Published under the joint sponsorship of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization World Health Orgnization Geneva, 1986 The International Programme on Chemical Safety (IPCS) is a joint venture of the United Nations Environment Programme, the International Labour Organisation, and the World Health Organization. The main objective of the IPCS is to carry out and disseminate evaluations of the effects of chemicals on human health and the quality of the environment. Supporting activities include the development of epidemiological, experimental laboratory, and risk-assessment methods that could produce internationally comparable results, and the development of manpower in the field of toxicology. Other activities carried out by the IPCS include the development of know-how for coping with chemical accidents, coordination -
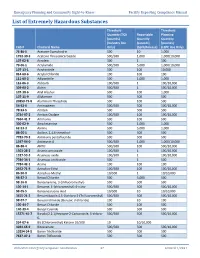
List of Extremely Hazardous Substances
Emergency Planning and Community Right-to-Know Facility Reporting Compliance Manual List of Extremely Hazardous Substances Threshold Threshold Quantity (TQ) Reportable Planning (pounds) Quantity Quantity (Industry Use (pounds) (pounds) CAS # Chemical Name Only) (Spill/Release) (LEPC Use Only) 75-86-5 Acetone Cyanohydrin 500 10 1,000 1752-30-3 Acetone Thiosemicarbazide 500/500 1,000 1,000/10,000 107-02-8 Acrolein 500 1 500 79-06-1 Acrylamide 500/500 5,000 1,000/10,000 107-13-1 Acrylonitrile 500 100 10,000 814-68-6 Acrylyl Chloride 100 100 100 111-69-3 Adiponitrile 500 1,000 1,000 116-06-3 Aldicarb 100/500 1 100/10,000 309-00-2 Aldrin 500/500 1 500/10,000 107-18-6 Allyl Alcohol 500 100 1,000 107-11-9 Allylamine 500 500 500 20859-73-8 Aluminum Phosphide 500 100 500 54-62-6 Aminopterin 500/500 500 500/10,000 78-53-5 Amiton 500 500 500 3734-97-2 Amiton Oxalate 100/500 100 100/10,000 7664-41-7 Ammonia 500 100 500 300-62-9 Amphetamine 500 1,000 1,000 62-53-3 Aniline 500 5,000 1,000 88-05-1 Aniline, 2,4,6-trimethyl- 500 500 500 7783-70-2 Antimony pentafluoride 500 500 500 1397-94-0 Antimycin A 500/500 1,000 1,000/10,000 86-88-4 ANTU 500/500 100 500/10,000 1303-28-2 Arsenic pentoxide 100/500 1 100/10,000 1327-53-3 Arsenous oxide 100/500 1 100/10,000 7784-34-1 Arsenous trichloride 500 1 500 7784-42-1 Arsine 100 100 100 2642-71-9 Azinphos-Ethyl 100/500 100 100/10,000 86-50-0 Azinphos-Methyl 10/500 1 10/10,000 98-87-3 Benzal Chloride 500 5,000 500 98-16-8 Benzenamine, 3-(trifluoromethyl)- 500 500 500 100-14-1 Benzene, 1-(chloromethyl)-4-nitro- 500/500