Receptor 3 Adenosine a Activation of Murine Lung Mast Cells By
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Adenosine Receptors and Cancer
Adenosine Receptors and Cancer P. Fishman, S. Bar-Yehuda, M. Synowitz, J.D. Powell, K.N. Klotz, S. Gessi, and P.A. Borea Contents 1 Introduction..................................................................................... 402 2A1 Adenosine Receptor........................................................................ 402 3A2A Adenosine Receptor...................................................................... 406 3.1 The A2AAR:ProtectorofHostTissue,ProtectorofTumors......................... 406 3.2 TumorsEvadetheImmuneSystembyInhibitingImmuneCellFunction........... 406 3.3 The A2AAR Negatively Regulates Immune Responses............................... 407 3.4 AdenosineProtectsTumorsfromImmuneDestruction............................... 408 3.5 A2AAR Antagonism as a Means of Enhancing Immunotherapy..................... 410 4A2B Adenosine Receptors..................................................................... 410 5A3 Adenosine Receptor........................................................................ 414 5.1 Overexpression of the A3AR in Tumor Versus Normal Adjacent Tissues........... 415 5.2 InVitroStudies......................................................................... 417 5.3 InVivoStudies.......................................................................... 419 5.4 Mechanisms of Action for the Anticancer Activity of the A3AR.................... 424 6 Anticancer Activity of A3AR Antagonists.................................................... 429 7 SummaryandConclusions................................................................... -

A3 Adenosine Receptor As a Target for Cancer Therapy
Anti-Cancer Drugs 2002, 13, pp. 437-443 Review paper A3 adenosine receptor as a target for cancer therapy Pnina Fishman,1,2 Sara Bar-Yehuda,1 Lea Madi2 and Ilan Cohn2 1Laboratory of Clinical and Tumor Immunology,The Felsenstein Medical Research Center,Tel-Aviv University, Rabin Medical Center, Petach Tikva 49100, Israel. 2Can-Fite BioPharma, Kiryat-Matalon, Petach Tikva 49160, Israel. Targeting the A3 adenosine receptor (A3AR) by adenosine or a tumor metastases are extremely rare in muscle tissue, synthetic agonist to this receptor (IB-MECA and Cl-IB-MECA) not withstanding the fact that it constitutes about results in a di¡erential e¡ect on tumor and on normal cells. Both the adenosine and the agonists inhibit the growth of various tumor 65–70% of lean body mass. This observation led to cell types such as melanoma, colon or prostate carcinoma and research that has its purpose to decipher the lymphoma. This e¡ect is speci¢c and is exerted on tumor cells physiological basis for this intruiging phenomenon. only. Moreover, exposure of peripheral blood mononuclear cells It was found that muscle cells secrete small mole- to adenosine or the agonists leads to the induction of granulocyte cules (MF) which inhibit growth of tumor cells.1,2 colony stimulating factor (G-CSF) production.When given orally to mice, the agonists suppress the growth of melanoma, colon and This growth inhibitory effect was observed on a prostate carcinoma in these animals, while inducing a myelopro- broad range of different tumor cell lines in vitro, tective e¡ect via the induction of G-CSF production.The de-regu- such as melanoma, carcinoma, leukomia and lym- lation of theWnt signaling pathway was found to be involved in the phoma. -

Biased Signaling of G Protein Coupled Receptors (Gpcrs): Molecular Determinants of GPCR/Transducer Selectivity and Therapeutic Potential
Pharmacology & Therapeutics 200 (2019) 148–178 Contents lists available at ScienceDirect Pharmacology & Therapeutics journal homepage: www.elsevier.com/locate/pharmthera Biased signaling of G protein coupled receptors (GPCRs): Molecular determinants of GPCR/transducer selectivity and therapeutic potential Mohammad Seyedabadi a,b, Mohammad Hossein Ghahremani c, Paul R. Albert d,⁎ a Department of Pharmacology, School of Medicine, Bushehr University of Medical Sciences, Iran b Education Development Center, Bushehr University of Medical Sciences, Iran c Department of Toxicology–Pharmacology, School of Pharmacy, Tehran University of Medical Sciences, Iran d Ottawa Hospital Research Institute, Neuroscience, University of Ottawa, Canada article info abstract Available online 8 May 2019 G protein coupled receptors (GPCRs) convey signals across membranes via interaction with G proteins. Origi- nally, an individual GPCR was thought to signal through one G protein family, comprising cognate G proteins Keywords: that mediate canonical receptor signaling. However, several deviations from canonical signaling pathways for GPCR GPCRs have been described. It is now clear that GPCRs can engage with multiple G proteins and the line between Gprotein cognate and non-cognate signaling is increasingly blurred. Furthermore, GPCRs couple to non-G protein trans- β-arrestin ducers, including β-arrestins or other scaffold proteins, to initiate additional signaling cascades. Selectivity Biased Signaling Receptor/transducer selectivity is dictated by agonist-induced receptor conformations as well as by collateral fac- Therapeutic Potential tors. In particular, ligands stabilize distinct receptor conformations to preferentially activate certain pathways, designated ‘biased signaling’. In this regard, receptor sequence alignment and mutagenesis have helped to iden- tify key receptor domains for receptor/transducer specificity. -

Adenosine, an Endogenous Distress Signal, Modulates Tissue Damage and Repair
Cell Death and Differentiation (2007) 14, 1315–1323 & 2007 Nature Publishing Group All rights reserved 1350-9047/07 $30.00 www.nature.com/cdd Review Adenosine, an endogenous distress signal, modulates tissue damage and repair BB Fredholm*,1 Adenosine is formed inside cells or on their surface, mostly by breakdown of adenine nucleotides. The formation of adenosine increases in different conditions of stress and distress. Adenosine acts on four G-protein coupled receptors: two of them, A1 and A3, are primarily coupled to Gi family G proteins; and two of them, A2A and A2B, are mostly coupled to Gs like G proteins. These receptors are antagonized by xanthines including caffeine. Via these receptors it affects many cells and organs, usually having a cytoprotective function. Joel Linden1 recently grouped these protective effects into four general modes of action: increased oxygen supply/demand ratio, preconditioning, anti-inflammatory effects and stimulation of angiogenesis. This review will briefly summarize what is known and what is not in this regard. It is argued that drugs targeting adenosine receptors might be useful adjuncts in many therapeutic approaches. Cell Death and Differentiation (2007) 14, 1315–1323; doi:10.1038/sj.cdd.4402132; published online 30 March 2007 Adenosine Formation and Adenosine Levels out of the cells by means of efficient equilibrative transporters. There are inhibitors for these transporters, including the drug Adenosine is always present both within and outside cells, dipyridamole. These inhibitors will increase extracellular since it is at a crossroads between different metabolic adenosine if it derives from extracellular breakdown of pathways. Levels of adenosine in cells and tissue fluids is adenine nucleotides, but decrease it if adenosine is formed in the nanomolar range under physiological conditions intracellularly. -

Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Amy Sue Bogard University of Tennessee Health Science Center
University of Tennessee Health Science Center UTHSC Digital Commons Theses and Dissertations (ETD) College of Graduate Health Sciences 12-2013 Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Amy Sue Bogard University of Tennessee Health Science Center Follow this and additional works at: https://dc.uthsc.edu/dissertations Part of the Medical Cell Biology Commons, and the Medical Molecular Biology Commons Recommended Citation Bogard, Amy Sue , "Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells" (2013). Theses and Dissertations (ETD). Paper 330. http://dx.doi.org/10.21007/etd.cghs.2013.0029. This Dissertation is brought to you for free and open access by the College of Graduate Health Sciences at UTHSC Digital Commons. It has been accepted for inclusion in Theses and Dissertations (ETD) by an authorized administrator of UTHSC Digital Commons. For more information, please contact [email protected]. Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells Document Type Dissertation Degree Name Doctor of Philosophy (PhD) Program Biomedical Sciences Track Molecular Therapeutics and Cell Signaling Research Advisor Rennolds Ostrom, Ph.D. Committee Elizabeth Fitzpatrick, Ph.D. Edwards Park, Ph.D. Steven Tavalin, Ph.D. Christopher Waters, Ph.D. DOI 10.21007/etd.cghs.2013.0029 Comments Six month embargo expired June 2014 This dissertation is available at UTHSC Digital Commons: https://dc.uthsc.edu/dissertations/330 Adenylyl Cyclase 2 Selectively Regulates IL-6 Expression in Human Bronchial Smooth Muscle Cells A Dissertation Presented for The Graduate Studies Council The University of Tennessee Health Science Center In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy From The University of Tennessee By Amy Sue Bogard December 2013 Copyright © 2013 by Amy Sue Bogard. -

Adenosine Receptor Containing Oligomers: Their Role in the Control of Dopamine and Glutamate Neurotransmission in the Brain☆
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Biochimica et Biophysica Acta 1808 (2011) 1245–1255 Contents lists available at ScienceDirect Biochimica et Biophysica Acta journal homepage: www.elsevier.com/locate/bbamem Review Adenosine receptor containing oligomers: Their role in the control of dopamine and glutamate neurotransmission in the brain☆ Francisco Ciruela a,⁎, Maricel Gómez-Soler a, Diego Guidolin b, Dasiel O. Borroto-Escuela c, Luigi F. Agnati d, Kjell Fuxe c, Víctor Fernández-Dueñas a a Unitat de Farmacologia, Departament de Patologia i Terapèutica Experimental, Facultat de Medicina, Universitat de Barcelona, 08097 L'Hospitalet de Llobregat, Spain b Department of Human Anatomy and Physiology, University of Padova, Padova, Italy c Department of Neuroscience, Karolinska Institutet, Stockholm, Sweden d IRCCS San Camillo, Lido Venezia, Italy article info abstract Article history: While the G protein-coupled receptor (GPCR) oligomerization has been questioned during the last fifteen years, Received 22 November 2010 the existence of a multi-receptor complex involving direct receptor–receptor interactions, called receptor Received in revised form 2 February 2011 oligomers, begins to be widely accepted. Eventually, it has been postulated that oligomers constitute a distinct Accepted 5 February 2011 functional form of the GPCRs with essential receptorial features. Also, it has been proven, under certain Available online 18 February 2011 circumstances, that the GPCR oligomerization phenomenon is crucial for the receptor biosynthesis, maturation, trafficking, plasma membrane diffusion, and pharmacology and signalling. Adenosine receptors are GPCRs that Keywords: G protein-coupled receptors mediate the physiological functions of adenosine and indeed these receptors do also oligomerize. -

Receptor Signaling Through Osteoclast-Associated Monocyte
Downloaded from http://www.jimmunol.org/ by guest on September 29, 2021 is online at: average * The Journal of Immunology The Journal of Immunology , 20 of which you can access for free at: 2015; 194:3169-3179; Prepublished online 27 from submission to initial decision 4 weeks from acceptance to publication February 2015; doi: 10.4049/jimmunol.1402800 http://www.jimmunol.org/content/194/7/3169 Collagen Induces Maturation of Human Monocyte-Derived Dendritic Cells by Signaling through Osteoclast-Associated Receptor Heidi S. Schultz, Louise M. Nitze, Louise H. Zeuthen, Pernille Keller, Albrecht Gruhler, Jesper Pass, Jianhe Chen, Li Guo, Andrew J. Fleetwood, John A. Hamilton, Martin W. Berchtold and Svetlana Panina J Immunol cites 43 articles Submit online. Every submission reviewed by practicing scientists ? is published twice each month by Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Author Choice option Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts http://jimmunol.org/subscription Freely available online through http://www.jimmunol.org/content/suppl/2015/02/27/jimmunol.140280 0.DCSupplemental This article http://www.jimmunol.org/content/194/7/3169.full#ref-list-1 Information about subscribing to The JI No Triage! Fast Publication! Rapid Reviews! 30 days* Why • • • Material References Permissions Email Alerts Subscription Author Choice Supplementary The Journal of Immunology The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2015 by The American Association of Immunologists, Inc. All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. -
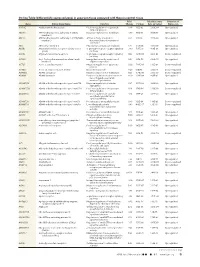
Differentially Expressed Genes in Aneurysm Tissue Compared With
On-line Table: Differentially expressed genes in aneurysm tissue compared with those in control tissue Fold False Discovery Direction of Gene Entrez Gene Name Function Change P Value Rate (q Value) Expression AADAC Arylacetamide deacetylase Positive regulation of triglyceride 4.46 1.33E-05 2.60E-04 Up-regulated catabolic process ABCA6 ATP-binding cassette, subfamily A (ABC1), Integral component of membrane 3.79 9.15E-14 8.88E-12 Up-regulated member 6 ABCC3 ATP-binding cassette, subfamily C (CFTR/MRP), ATPase activity, coupled to 6.63 1.21E-10 7.33E-09 Up-regulated member 3 transmembrane movement of substances ABI3 ABI family, member 3 Peptidyl-tyrosine phosphorylation 6.47 2.47E-05 4.56E-04 Up-regulated ACKR1 Atypical chemokine receptor 1 (Duffy blood G-protein–coupled receptor signaling 3.80 7.95E-10 4.18E-08 Up-regulated group) pathway ACKR2 Atypical chemokine receptor 2 G-protein–coupled receptor signaling 0.42 3.29E-04 4.41E-03 Down-regulated pathway ACSM1 Acyl-CoA synthetase medium-chain family Energy derivation by oxidation of 9.87 1.70E-08 6.52E-07 Up-regulated member 1 organic compounds ACTC1 Actin, ␣, cardiac muscle 1 Negative regulation of apoptotic 0.30 7.96E-06 1.65E-04 Down-regulated process ACTG2 Actin, ␥2, smooth muscle, enteric Blood microparticle 0.29 1.61E-16 2.36E-14 Down-regulated ADAM33 ADAM domain 33 Integral component of membrane 0.23 9.74E-09 3.95E-07 Down-regulated ADAM8 ADAM domain 8 Positive regulation of tumor necrosis 4.69 2.93E-04 4.01E-03 Up-regulated factor (ligand) superfamily member 11 production ADAMTS18 -

A3R Agonists
Last updated on May 14, 2020 Cognitive Vitality Reports® are reports written by neuroscientists at the Alzheimer’s Drug Discovery Foundation (ADDF). These scientific reports include analysis of drugs, drugs-in- development, drug targets, supplements, nutraceuticals, food/drink, non-pharmacologic interventions, and risk factors. Neuroscientists evaluate the potential benefit (or harm) for brain health, as well as for age-related health concerns that can affect brain health (e.g., cardiovascular diseases, cancers, diabetes/metabolic syndrome). In addition, these reports include evaluation of safety data, from clinical trials if available, and from preclinical models. A3R Agonists Evidence Summary Protects tissues against hypoxia and other metabolic stressors. Likely offers clinical benefits for inflammatory and fibrotic diseases and cancer. Clinically tested agonists have a good safety profile. Neuroprotective Benefit: Blood-brain barrier penetrant A3R agonists may protect against metabolic stress-related neurodegeneration by mitigating inflammation and excitotoxicity. Aging and related health concerns: Clinical studies show benefits in diseases characterized by inflammation and fibrosis including arthritis and fatty liver disease. Preclinical studies show cytoprotection during ischemia and mitigation of neuropathic pain. Safety: Clinically tested A3R agonists are well-tolerated and have a good safety profile. Very high doses may elevate heart rate. 1 Last updated on May 14, 2020 Availability: In clinical trials Dose: Not established CF101 -

A Non-Imaging High Throughput Approach to Chemical Library Screening at the Unmodified Adenosine-A3 Receptor in Living Cells
ORIGINAL RESEARCH published: 13 December 2017 doi: 10.3389/fphar.2017.00908 A Non-imaging High Throughput Approach to Chemical Library Screening at the Unmodified Adenosine-A3 Receptor in Living Cells Maria Augusta Arruda 1, 2, 3†, Leigh A. Stoddart 1, 2†, Karolina Gherbi 1, 2, Stephen J. Briddon 1, 2*, Barrie Kellam 4 and Stephen J. Hill 1, 2* 1 Division of Physiology, Pharmacology and Neuroscience, School of Life Sciences, Medical School, Queen’s Medical Centre, University of Nottingham, Nottingham, United Kingdom, 2 Centre of Membrane Proteins and Receptors, University of Birmingham and University of Nottingham, The Midlands, United Kingdom, 3 Vice-Diretoria de Ensino, Pesquisa e Inovacao, Farmanguinhos, Fiocruz, Rio de Janeiro, Brazil, 4 Centre for Biomolecular Sciences, School of Pharmacy, University of Nottingham, Nottingham, United Kingdom Edited by: Kenneth A. Jacobson, National Institutes of Health (NIH), Recent advances in fluorescent ligand technology have enabled the study of G United States protein-coupled receptors in their native environment without the need for genetic Reviewed by: Vittoria Colotta, modification such as addition of N-terminal fluorescent or bioluminescent tags. Here, University of Florence, Italy we have used a non-imaging plate reader (PHERAstar FS) to monitor the binding Eddy Sotelo, of fluorescent ligands to the human adenosine-A receptor (A AR; CA200645 and Universidade de Santiago de 3 3 Compostela, Spain AV039), stably expressed in CHO-K1 cells. To verify that this method was suitable *Correspondence: for the study of other GPCRs, assays at the human adenosine-A1 receptor, and β1 Stephen J. Briddon and β2 adrenoceptors (β1AR and β2AR; BODIPY-TMR-CGP-12177) were also carried [email protected] Stephen J. -

Supp Fig 1 Categorization of the Genes Regulated in the Adult but Not
Supp Fig 1 Categorization of the genes regulated in the adult but not the neonate dorsal horn following peripheral nerve injury by ‘Function and Disease’ within the Ingenuity Pathway Analysis (IPA) software system. Bars represent the LOD value (-log (p-value)) of the likelihood of an association between the Function and Disease gene groups with the genes in the Ad0.05/Ne0.9 list (right-tailed Fisher's Exact Test); Only functional categories with LOD scores of above 10 shown. Immune related gene groups marked. Supp Fig 2 A; Mechanical sensitivity as assayed by von Frey stimulation does not vary when assayed on the ipsilateral hindpaw relative to the contralateral paw of the same animal as a function of time subsequent to SNI injury in T cell deficient Nude mice. B; Mechanical sensitivity (von Frey) is significantly reduced on the ipsilateral hindpaw relative to the contralateral paw of the same animal over time subsequent to SNI injury in B cell deficient mice. All data are mean ± SEM, one-way ANOVA, Students t-test *P < 0.05, n=6 animals per group. Supp Fig 3 CD4 (A) and CD8 (B) expression in the dorsal horn of the spinal cord of neonatal and adult rats over time post SNI relative to age matched controls and uninjured animals respectively, detected by Q-PCR. Data are expressed as mean fold ±SEM, one- way ANOVA, *P < 0.05 Student’s t-test, n=4 per group. Supp Table 1 Genes identified by microarray analysis to be regulated in the neonatal dorsal horn 7 days post SNI relative to age match control sham animals. -

Treatment of Chronic Neuropathic Pain
Narrative Review Treatment of chronic neuropathic pain: purine receptor modulation Kenneth A. Jacobsona, Luigino Antonio Giancottib, Filomena Laurob, Fatma Muftib, Daniela Salveminib,* 06/11/2020 on 3U6Qur4PG8Cp3fhvloi8qBpY7fujG3dPKasYMD+lWJpib/J9/jG1M3P5/SdPwJzQZopI9kkCkONW3pMktyxp4tovfFowndLubZk7zJkoatOlb7XXAf6/mu3JhfuJ6/VZKl/sopHhTqbQ4RCp2fMF2A== by https://journals.lww.com/pain from Downloaded Downloaded Abstract from https://journals.lww.com/pain Extracellular nucleosides and nucleotides have widespread functions in responding to physiological stress. The “purinome” encompasses 4 G-protein-coupled receptors (GPCRs) for adenosine, 8 GPCRs activated by nucleotides, 7 adenosine 59-triphosphate-gated P2X ion channels, as well as the associated enzymes and transporters that regulate native agonist levels. Purinergic signaling modulators, such as receptor agonists and antagonists, have potential for treating chronic pain. Adenosine and its analogues potently suppress nociception in preclinical models by activating A1 and/or A3 adenosine receptors (ARs), but safely harnessing this pathway to clinically treat pain has not by 3U6Qur4PG8Cp3fhvloi8qBpY7fujG3dPKasYMD+lWJpib/J9/jG1M3P5/SdPwJzQZopI9kkCkONW3pMktyxp4tovfFowndLubZk7zJkoatOlb7XXAf6/mu3JhfuJ6/VZKl/sopHhTqbQ4RCp2fMF2A== been achieved. Both A2AAR agonists and antagonists are efficacious in pain models. Highly selective A3AR agonists offer a novel approach to treat chronic pain. We have explored the structure activity relationship of nucleoside derivatives at this subtype using a computational structure-based approach. Novel A3AR agonists for pain control containing a bicyclic ring system (bicyclo [3.1.0] hexane) in place of ribose were designed and screened using an in vivo phenotypic model, which reflected both pharmacokinetic and pharmacodynamic parameters. High specificity (.10,000-fold selective for A3AR) was achieved with the aid of receptor homology models based on related GPCR structures. These A3AR agonists are well tolerated in vivo and highly efficacious in models of chronic neuropathic pain.