Bee Diseases and Enemies; Control and Preventive Measures Introduction
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Acoustic Communication in the Nocturnal Lepidoptera
Chapter 6 Acoustic Communication in the Nocturnal Lepidoptera Michael D. Greenfield Abstract Pair formation in moths typically involves pheromones, but some pyra- loid and noctuoid species use sound in mating communication. The signals are generally ultrasound, broadcast by males, and function in courtship. Long-range advertisement songs also occur which exhibit high convergence with commu- nication in other acoustic species such as orthopterans and anurans. Tympanal hearing with sensitivity to ultrasound in the context of bat avoidance behavior is widespread in the Lepidoptera, and phylogenetic inference indicates that such perception preceded the evolution of song. This sequence suggests that male song originated via the sensory bias mechanism, but the trajectory by which ances- tral defensive behavior in females—negative responses to bat echolocation sig- nals—may have evolved toward positive responses to male song remains unclear. Analyses of various species offer some insight to this improbable transition, and to the general process by which signals may evolve via the sensory bias mechanism. 6.1 Introduction The acoustic world of Lepidoptera remained for humans largely unknown, and this for good reason: It takes place mostly in the middle- to high-ultrasound fre- quency range, well beyond our sensitivity range. Thus, the discovery and detailed study of acoustically communicating moths came about only with the use of electronic instruments sensitive to these sound frequencies. Such equipment was invented following the 1930s, and instruments that could be readily applied in the field were only available since the 1980s. But the application of such equipment M. D. Greenfield (*) Institut de recherche sur la biologie de l’insecte (IRBI), CNRS UMR 7261, Parc de Grandmont, Université François Rabelais de Tours, 37200 Tours, France e-mail: [email protected] B. -

Managing Maladies: Parasites, Pests & Pesticides
MANAGING MALADIES: PARASITES, PESTS & PESTICIDES W. Brian Kreowski Agricultural Inspector Today’s Objectives: • Briefly explain the Virginia Apiary Inspection program • Go over the main insects and mites that might harm your bees • Discuss animals that can potentially harm your hive(s) and their contents • Make you aware of the hazards of pesticides to your investment in bees Apiary Inspection Program • Inspector’s duties: • Examine bees for disease, etc. • Prohibit movement or sale of diseased hives • Destroy hives when necessary Apiary Inspection Program • Beekeepers role: • Provide movable frames • Tightly close dead hives • Notify State Apiarist of diseased bees • Notify State Apiarist of possible Africanized Honeybee Apiary Inspection Program: Regulations – No honey in candy for queen cages – Queen rearing and queen mating apiary inspection – Inspection certificate for packages – Inspection prior to bringing hives into Commonwealth – Certificate for sale of comb, hives, used equipment with comb or appliances Parasites: Varroa Mite • Varroa destructor Anderson & Trueman • External parasite of adults… Parasites: Varroa Mite • …and immatures Parasites: Varroa Mite Parasites: Varroa Mite - Biology • Female mites crawl into brood cells (especially drone cells) before they are capped • Feed on bee food first, then on the prepupa • Lay the first eggs about 60 hours after cell capping, subsequent eggs are laid at 30 hour intervals • Lay about 4 to 6 eggs total Parasites: Varroa Mite - Biology • Immature mites develop on the pupal bee, requiring -

Beekeepers and Bee Wax Moth
Faculty of Natural Resources and Agricultural Sciences Beekeepers and Bee Wax Moth – What’s the Buzz About? A study about the attitude towards beekeeping and wax moth in Kenya Biodlare och bivaxmal – vad är allt surrande om? – En studie om attityden mot biodling och bivaxmal i Kenya Ida Johansson Independent project in Biology • 15 hec Agriculture program - plant and soil sciences Department of Ecology Uppsala 2019 Beekeepers and Bee Wax Moth – What’s the Buzz About? A study about the attitude towards beekeeping and wax moth in Kenya Biodlare och bivaxmal – vad är allt surrande om? – En studie om attityden mot biodling och bivaxmal i Kenya Ida Johansson Supervisor: Helena Bylund, SLU, Department of Ecology Assistant supervisor: Jamleck Muturi, University of Embu, Department of Biological Sciences Examiner: Mattias Jonsson, SLU, Department of Ecology Credits: 15 hec Level: G2E Course title: Independent project in Biology Course code: EX0894 Programme/education: Agriculture Program – Plant and Soil Sciences Course coordinating department: Department of Aquatic Sciences and Assessment Place of publication: Uppsala Year of publication: 2019 Cover picture: Ida Johansson Online publication: https://stud.epsilon.slu.se Keywords: Pollination, wax moth, Kenya, bees, beekeeping Sveriges lantbruksuniversitet Swedish University of Agricultural Sciences Faculty of natural resources and Agricultural Sciences Department of Ecology Abstract The bee wax moth (Galleria mellonella) is a troublesome pest of the honeybees that can cause substantial financial losses that can be devastating for the beekeepers. The moth is a universal pest but thrives in a warmer climate, the insect is nocturnal and feed on pollen, wax and other impurities. The eggs are laid in crevice’s and other small spaces where the bees cannot reach them. -

Moth Hearing and Sound Communication
J Comp Physiol A (2015) 201:111–121 DOI 10.1007/s00359-014-0945-8 REVIEW Moth hearing and sound communication Ryo Nakano · Takuma Takanashi · Annemarie Surlykke Received: 17 July 2014 / Revised: 13 September 2014 / Accepted: 15 September 2014 / Published online: 27 September 2014 © Springer-Verlag Berlin Heidelberg 2014 Abstract Active echolocation enables bats to orient notion of moths predominantly being silent. Sexual sound and hunt the night sky for insects. As a counter-measure communication in moths may apply to many eared moths, against the severe predation pressure many nocturnal perhaps even a majority. The low intensities and high fre- insects have evolved ears sensitive to ultrasonic bat calls. quencies explain that this was overlooked, revealing a bias In moths bat-detection was the principal purpose of hear- towards what humans can sense, when studying (acoustic) ing, as evidenced by comparable hearing physiology with communication in animals. best sensitivity in the bat echolocation range, 20–60 kHz, across moths in spite of diverse ear morphology. Some Keywords Co-evolution · Sensory exploitation · eared moths subsequently developed sound-producing Ultrasound · Echolocating bats · Predator-prey organs to warn/startle/jam attacking bats and/or to com- municate intraspecifically with sound. Not only the sounds for interaction with bats, but also mating signals are within Introduction the frequency range where bats echolocate, indicating that sound communication developed after hearing by “sensory Insect hearing and sound communication is a fascinating exploitation”. Recent findings on moth sound communica- subject, where the combination of many classical studies tion reveal that close-range (~ a few cm) communication and recent progress using new technological and molecular with low-intensity ultrasounds “whispered” by males dur- methods provide an unsurpassed system for studying and ing courtship is not uncommon, contrary to the general understanding the evolution of acoustic communication, intraspecific as well as between predator and prey. -
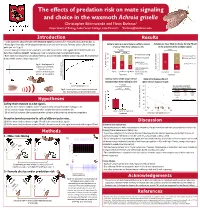
Results Methods Introduction Christopher Edomwande and Flavia Barbosa1 Hypotheses Discussion References Acknowledgements
The effects of predation risk on mate signaling and choice in the waxmoth Achroia grisella Christopher Edomwande and Flavia Barbosa1 Department of Biology, Lake Forest College, Lake Forest IL [email protected] Introduction Results • Male waxmoths attract females with ultrasonic signals consisting of an incessant pulse pair train (fig. 1). • Males signal from leks, which receptive females orient and walk towards. Females prefer calls with faster Solitary males are more likely to exhibit a silence Females are more likely to choose the low PR call pulse‐pair rate1 (fig. 2). response than those calling in a lek. in the presence of the predator signal. • Several mating behaviors make waxmoths vulnerable to predators: male signals attract both females and **p<0.001 *p=0.03 bats, thus creating a tradeoff. Females are more vulnerable when moving towards males. *p=0.014 p=0.06 20 Continuous callingSeries2 12 • Both sexes may respond to bat calls by freezing, and calling males exhibit a silence response. More attractive 2 Chose low PR call males exhibit shorter silence responses2‐4 . 15 8 Silence responseSeries1 9 8 Chose high PR call 13 10 Cochran Q test: 6 13 17 2 Fig. 1 – Oscillogram of a X2 = 15.1667, p<0.001. McNemar test: X = 4.5 5 11 3 typical male call. Males Number of males 5 6 Pairwise comparisons: typically produce 0 McNemar test, Number of females 0 between 70 to 110 pulse Silence Low PR lek High PR lek Bonferroni adjusted No predator Predator pairs s‐1, at 70–130 kHz. Treatment alpha = 0.016. -

Parasites of the Honeybee
Parasites of the Honeybee This publication was produced by Dr. Mary F. Coffey Teagasc, Crops Research Centre, Oak Park, Carlow co-financed by The Department of Agriculture, Fisheries and Food November 2007 Table of Contents Introduction……………………………………………………………………….………….…….1 Bee origin and classification………………..……………………………………………………..2 The honeybee colony……….…………………………………..……………………......................4 Irish floral sources of pollen and nectar……………………………………...…………….…….11 Pests and diseases of the honeybee………………………………………………………….……12 Pests……………………………………….………………………………………...…………....12 Varroa Mite……………………………………………………………………………....12 Tracheal Mite…………………………………………………………………………….25 Bee-Louse (Braula Coeca)………………………………………………...………..........28 Wax Moths (Achroia Grisella & Galleria Mellonella) ………………………………….29 Tropilaelaps Mite…………………………………………………...……………………31 Small Hive Beetle (SHB)……………………………………………………...…….……33 Rodents (Mice and Rats)…………………………………………………………………36 Wasps and Bumblebees…………………………………………………………………..37 Adult bee diseases……………………….………………………………………….................….38 Nosema…………………………………………………………………………………...38 Brood diseases.………………………………………...……………………….…………..….…40 American Foulbrood………………………………………………………..……………40 European Foulbrood………………………...…………………………………………...43 Chalkbrood ……………………………..…………………….………………………….45 Stonebrood ……………………………………………………………………………….47 Viral diseases…………………………………….…………………………….……...………….47 Sacbrood Virus (SBV)……...…………………………………………………………….47 Deformed Wing Virus (DWV)……………..…………………………………………………….49 Kashmir Virus (KBV)……………………………………………………………………………..50 -

Potential of Some Volatile Oils and Chemicals Against Lesser Wax Moth, Acheroia Gresilla F. (Lepidoptera: Pyralidae)
& Herpeto gy lo lo gy o : h C it u n r r r e O n , t y Entomology, Ornithology & R g e o l s o e a Abou El-Ela, Entomol Ornithol Herpetol 2014, 3:3 m r o c t h n E Herpetology: Current Research DOI: 10.4172/2161-0983.1000129 ISSN: 2161-0983 Research Article Open Access Potential of Some Volatile Oils and Chemicals against Lesser Wax Moth, Acheroia gresilla F. (Lepidoptera: Pyralidae) Adel A Abou El-Ela* Zoology Department, Faculty of Science, Fayoum University, Egypt *Corresponding author: Adel A. Abou El-Ela, Zoology Department, Faculty of Science, Fayoum University, Egypt, Tel: 00201017787225; E-mail: [email protected] Rec date: Mar 10, 2014; Acc date: May 24, 2014; Pub date: May 27, 2014 Copyright: © 2014 Abou El-Ela AA. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Abstract The oils of six plants viz.; clove; Eugenia aromatic., basil; Ocimum basillicum L., thyme; Thymus vulgaris L., blue gum; Eucalyptus spp., spearmint; Minth viridis L., Lemongrass oil; Cymbopogon citrates Hort. And six aromatic fragments namely; methyl salicylate, eugenol, menthol, thymol, camphor and naphthalene were tested against Acheroia gresilla F. Paradicholorbenzene, acetic and formic acids were used as check and the LC50s were determined against the last larval instar. Results revealed that methyl salicylate, formic acid, clove and basil oils were highly effective. Keywords: Plant volatile oils; Methyl salicylate; Formic acid; Clove eggs and a black ribbon cloth was hanged inside each cage to serve as and Acherioia gresilla oviposition site. -

Lesser Wax Moth Achroia Grisella Fabricius (Insecta: Lepidoptera: Pyralidae)1 Ashley A
EENY637 Lesser Wax Moth Achroia grisella Fabricius (Insecta: Lepidoptera: Pyralidae)1 Ashley A. Egelie, Ashley N. Mortensen, Lynn Barber, Jessica Sullivan, and James D. Ellis2 Introduction Distribution Achroia grisella Fabricius, the lesser wax moth (Figure 1), Lesser wax moths are cosmopolitan in distribution, and is considered a pest of unoccupied honey bee, Apis spp., are present nearly anywhere honey bees are managed. The hives and stored hive materials. However, wax moths can lesser wax moth is more successful in warmer tropical and be considered beneficial insects in wild/feral colonies subtropical climates and cannot survive long periods of because they destroy combs that remain after a colony of freezing temperatures. However, the lesser wax moth can bees dies or abandons the nest site. These abandoned combs survive at higher latitudes and lower temperatures than can potentially harbor pathogens and/or pesticide residues that the greater wax moth. have been left behind by the previous colony. Wax moths consume the abandoned combs, thereby minimizing the Description risk for exposure to future inhabitants of the cavity. Lesser wax moths have a similar life history to that of greater wax Eggs moths, Galleria mellonella L. Adult females typically lay eggs in protected crevices near a food source. The eggs are creamy white and spherical (Figure 2). The time it takes for eggs to hatch is variable, with warmer temperatures leading to faster development for all life stages. Eggs typically take five to eight days to hatch. Larvae The larvae have narrow, white bodies with a brown head and pronotal shield (Figure 3). Larval development can take between one and five months, at an average of six to seven weeks at 29° to 32°C. -

Bat Predation and the Evolution of Leks in Acoustic Moths
Bat predation and the evolution of leks in acoustic moths Sylvain Alem, Klemen Koselj, Björn M. Siemers & Michael D. Greenfield Behavioral Ecology and Sociobiology ISSN 0340-5443 Behav Ecol Sociobiol DOI 10.1007/s00265-011-1219- x 1 23 Your article is protected by copyright and all rights are held exclusively by Springer- Verlag. This e-offprint is for personal use only and shall not be self-archived in electronic repositories. If you wish to self-archive your work, please use the accepted author’s version for posting to your own website or your institution’s repository. You may further deposit the accepted author’s version on a funder’s repository at a funder’s request, provided it is not made publicly available until 12 months after publication. 1 23 Author's personal copy Behav Ecol Sociobiol DOI 10.1007/s00265-011-1219-x ORIGINAL PAPER Bat predation and the evolution of leks in acoustic moths Sylvain Alem & Klemen Koselj & Björn M. Siemers & Michael D. Greenfield Received: 19 January 2011 /Revised: 21 March 2011 /Accepted: 6 June 2011 # Springer-Verlag 2011 Abstract Theories of lek evolution generally invoke attraction and reduced predation risk. Our results support enhanced mating success experienced by males signalling the idea that multiple factors operating simultaneously may in aggregations. Reduced predation has also been acknowl- maintain lekking behaviour. edged as a potential factor driving lek formation, but its role is more ambiguous. Although lekking is a complex Keywords Acoustic communication . Mate choice . Sexual behaviour, few empirical studies have investigated the role selection . Signal evolution . Ultrasonic signals of both claims. -

Contributions of Deaf People to Entomology: a Hidden Legacy
TAR Terrestrial Arthropod Reviews 5 (2012) 223–268 brill.com/tar Contributions of deaf people to entomology: A hidden legacy Harry G. Lang1 and Jorge A. Santiago-Blay2 1Professor Emeritus, Rochester Institute of Technology, National Technical Institute for the Deaf, Rochester, New York 14623, USA email: [email protected] 2Department of Paleobiology, National Museum of Natural History, 10th and Constitution Avenue, Washington, District of Columbia 20560, USA e-mail: [email protected] Received on April 24, 2012. Accepted on June 15, 2012. Final version received on June 26, 2012 Summary Despite communication challenges, deaf and hard-of-hearing individuals made many new discoveries during the emergence of entomology as a scientific discipline. In the 18th century, Switzerland’s naturalist Charles Bonnet, a preformationist, investigated parthenogenesis, a discovery that laid the groundwork for many scientists to examine conception, embryonic development, and the true, non-preformationist nature of heredity. In the 19th century, insect collectors, such as Arthur Doncaster and James Platt-Barrett in England, as well as Johann Jacob Bremi-Wolf in Switzerland, developed specialized knowledge in sev- eral insect orders, particularly the Lepidoptera. In contrast, the contributions to entomology of Fielding Bradford Meek and Leo Lesquereux in the United States stemmed from their paleontological studies, while the work of Simon S. Rathvon and Henry William Ravenel in economic entomology and botany, respectively, was derived from their strong interests in plants. These and other contributors found ways to overcome the isolation imposed upon them by deafness and, as a group, deaf and hard-of-hearing scien- tists established a legacy in entomology that has not been previously explored. -

Insect Pests and Their Control - Wax Moth - an Apiary Problem
Journal of the Department of Agriculture, Western Australia, Series 3 Volume 7 Number 2 March-April, 1958 Article 12 3-1958 Insect pests and their control - Wax moth - An apiary problem G. D. Rimes Follow this and additional works at: https://researchlibrary.agric.wa.gov.au/journal_agriculture3 Recommended Citation Rimes, G. D. (1958) "Insect pests and their control - Wax moth - An apiary problem," Journal of the Department of Agriculture, Western Australia, Series 3: Vol. 7 : No. 2 , Article 12. Available at: https://researchlibrary.agric.wa.gov.au/journal_agriculture3/vol7/iss2/12 This article is brought to you for free and open access by Research Library. It has been accepted for inclusion in Journal of the Department of Agriculture, Western Australia, Series 3 by an authorized administrator of Research Library. For more information, please contact [email protected]. WAX MOTH —An apiary problem HE damage to honeycomb caused by wax moth is a world-wide problem, the exten T sive distribution of the pest being due to the transport of infected material which has accompanied the expansion of bee-keeping. Although wax moth can be reared on a variety of materials not obtainable in the beehive, their association with the honey-bee is of such antiquity, that honeycomb in one form or another now forms its only natural food supply. Wax moth is a pest to most bee-keepers eggs, barely visible to the eye, are laid, at various times of the year, whether in some reports placing the number as high weak colonies or in stored honey combs. -

An Introduction to the Disorders of the Honey Bee
An introduction to the disorders of the honey bee John Carr Disorders of the Honey Bee – Apis mellifera An introduction Contents The contents are hyperlinked to the relevant page. Please follow the computer’s guide. Introduction Terms used in agriculture 4 Evolution of the honey bee –Apis mellifera 5 Life cycle of the honey bee – Apis mellifera 6 Anatomy of the honey bee – Apis mellifera 8 Anatomy of the hive 17 Properties of Honey 25 Communication between honey bees – an introduction 26 Disorders of Bees Reportable disorders of Bees 28 Disorders of the Brood American Foul Brood 29 Chalkbrood 31 Chilled Brood 32 European Foul Brood 33 Sacbrood Virus 34 Stonebrood 35 Brood conditions others: 36 Bald brood Drone laying queen Frame issues Laying workers Queen half-moon syndrome Disorders of the adult and larvae Nosema 38 Septicaemia 40 Tracheal Mite 41 Tropilaelaps 43 Varroa 47 Viruses of bees 52 Acute bee paralysis virus Bee Virus X Bee Virus Y Black Queen Cell Virus Chronic bee paralysis virus Cloudy Wing Virus Deformed Wing Virus Filamentous Virus Invertebrate Iridescent Virus Israel Acute Paralysis Virus KaKugo virus Kashmir Bee Virus Sacbrood Virus 2 Slow paralysis Virus Varroa-destructor virus -1 Colony Collapse Disorder - CCD 55 Bee Pests 56 Arthropods 57 Ants Beetles others Death’s Head Hawk Moth Earwigs Hornets Pollen mites Small and large hive beetles Spiders The bee louse Wasps Wax Moths Others 60 Birds – Wood peckers and Bee Catchers Honey badgers, badgers and raccoons Mice and shrews Sheep, horses, cows and deer Poisoning poisoning 61 Clinical examination of bees and their hives Biosecurity considerations 63 Clinical examination of a hive 64 Indicators of general health 67 Postmortem examination of honey bees 68 Enhacing hive health 71 Killing bees 75 Further reading 76 Notes 77 Index 78 3 Bee Terms When you are unfamiliar with the world of bee keeping some of the common terms may seem a little daunting.