A Functional C–G Polymorphism in the CYP7B1 Promoter Region and Its Different Distribution in Orientals and Caucasians
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Guideline for CYP2D6 and Tamoxifen Therapy
CPIC GUIDELINES Clinical Pharmacogenetics Implementation Consortium (CPIC) Guideline for CYP2D6 and Tamoxifen Therapy Matthew P. Goetz1, Katrin Sangkuhl2, Henk-Jan Guchelaar3, Matthias Schwab4,5,6, Michael Province7, Michelle Whirl-Carrillo2, W. Fraser Symmans8, Howard L. McLeod9, Mark J. Ratain10, Hitoshi Zembutsu11, Andrea Gaedigk12, Ron H. van Schaik13,14, James N. Ingle1, Kelly E. Caudle15 and Teri E. Klein2 Tamoxifen is biotransformed by CYP2D6 to 4-hydroxytamoxifen gene/CYP2D6; CYP2D6 Allele Definition Table in Ref. 1). and 4-hydroxy N-desmethyl tamoxifen (endoxifen), both with CYP2D6 alleles have been extensively studied in multiple geo- greater antiestrogenic potency than the parent drug. Patients with graphically, racially, and ethnically diverse groups and significant certain CYP2D6 genetic polymorphisms and patients who receive differences in allele frequencies have been observed (CYP2D6 strong CYP2D6 inhibitors exhibit lower endoxifen concentrations Frequency Table1). The most commonly reported alleles are cate- and a higher risk of disease recurrence in some studies of tamoxi- gorized into functional groups as follows: normal function (e.g., fen adjuvant therapy of early breast cancer. We summarize CYP2D6*1 and *2), decreased function (e.g., CYP2D6*9, *10, evidence from the literature and provide therapeutic recommenda- 2,3 tions for tamoxifen based on CYP2D6 genotype. *17, and *41), and no function (e.g., CYP2D6*3, *4, *5, *6). Because CYP2D6 is subject to gene deletions, duplications, or The purpose of this guideline is to provide clinicians information multiplications, many clinical laboratories also report copy num- that will allow the interpretation of clinical CYP2D6 genotype ber variations. CYP2D6*5 represents a gene deletion (no func- tests so that the results can be used to guide prescribing of tamoxi- fen. -

The Role of Vitamin D in Autoimmune Hepatitis
Elmer ress Review J Clin Med Res • 2013;5(6):407-415 The Role of Vitamin D in Autoimmune Hepatitis Khanh vinh quoc Luonga, b, Lan Thi Hoang Nguyena disease is classified into 2 distinct types according to the na- Abstract ture of autoantibodies [1, 2]. Type 1 AIH is characterized by anti-nuclear antibodies and/or smooth muscle antibodies Autoimmune hepatitis is an inflammation of the liver characterized in serum of northern European and American adults. Type 2 by the presence of peri-portal hepatitis, hypergammaglobulinemia, AIH is characterized by antibodies to the liver-kidney mi- and the serum autoantibodies. The disease is classified into 2 dis- crosome type 1 (anti-LKM1) and primarily affects children tinct types according to the nature of auto-antibodies. Disturbances between the ages of 2 and 14 years. Disturbances of the cal- of the calcium-parathyroid hormone-vitamin D axis are frequently cium-parathyroid hormone-vitamin D axis are frequently as- associated with chronic liver disease. Patients with AIH have a high sociated with chronic liver disease [3]. Vitamin D deficiency prevalence of vitamin D deficiency. Genetic studies have provided the opportunity to determine which proteins link vitamin D to AIH is common in non-cholestatic liver disease and correlates pathology, namely, the major histocompatibility complex class II with disease severity [4]. AIH patients have low of vitamin D molecules, vitamin D receptors, toll-like receptors, cytotoxic T levels compared with control group [5]. Many studies have lymphocyte antigen-4, cytochrome P450 CYP2D6, regulatory T shown a significant effect of calcitriol on liver cell physiol- cells (Tregs) and the forkhead/winged helix transcription factor 3. -

Cytochrome P450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid
Comprehensive Summaries of Uppsala Dissertations from the Faculty of Pharmacy 231 _____________________________ _____________________________ Cytochrome P450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid Cloning, Expression and Catalytic Properties of CYP4F8 and CYP4F21 BY JOHAN BYLUND ACTA UNIVERSITATIS UPSALIENSIS UPPSALA 2000 Dissertation for the Degree of Doctor of Philosophy (Faculty of Pharmacy) in Pharmaceutical Pharmacology presented at Uppsala University in 2000 ABSTRACT Bylund, J. 2000. Cytochrome P450 Enzymes in Oxygenation of Prostaglandin Endoperoxides and Arachidonic Acid: Cloning, Expression and Catalytic Properties of CYP4F8 and CYP4F21. Acta Universitatis Upsaliensis. Comprehensive Summaries of Uppsala Dissertations from Faculty of Pharmacy 231 50 pp. Uppsala. ISBN 91-554-4784-8. Cytochrome P450 (P450 or CYP) is an enzyme system involved in the oxygenation of a wide range of endogenous compounds as well as foreign chemicals and drugs. This thesis describes investigations of P450-catalyzed oxygenation of prostaglandins, linoleic and arachidonic acids. The formation of bisallylic hydroxy metabolites of linoleic and arachidonic acids was studied with human recombinant P450s and with human liver microsomes. Several P450 enzymes catalyzed the formation of bisallylic hydroxy metabolites. Inhibition studies and stereochemical analysis of metabolites suggest that the enzyme CYP1A2 may contribute to the biosynthesis of bisallylic hydroxy fatty acid metabolites in adult human liver microsomes. 19R-Hydroxy-PGE and 20-hydroxy-PGE are major components of human and ovine semen, respectively. They are formed in the seminal vesicles, but the mechanism of their biosynthesis is unknown. Reverse transcription-polymerase chain reaction using degenerate primers for mammalian CYP4 family genes, revealed expression of two novel P450 genes in human and ovine seminal vesicles. -

ACAT) in Cholesterol Metabolism: from Its Discovery to Clinical Trials and the Genomics Era
H OH metabolites OH Review Acyl-Coenzyme A: Cholesterol Acyltransferase (ACAT) in Cholesterol Metabolism: From Its Discovery to Clinical Trials and the Genomics Era Qimin Hai and Jonathan D. Smith * Department of Cardiovascular & Metabolic Sciences, Cleveland Clinic, Cleveland, OH 44195, USA; [email protected] * Correspondence: [email protected]; Tel.: +1-216-444-2248 Abstract: The purification and cloning of the acyl-coenzyme A: cholesterol acyltransferase (ACAT) enzymes and the sterol O-acyltransferase (SOAT) genes has opened new areas of interest in cholesterol metabolism given their profound effects on foam cell biology and intestinal lipid absorption. The generation of mouse models deficient in Soat1 or Soat2 confirmed the importance of their gene products on cholesterol esterification and lipoprotein physiology. Although these studies supported clinical trials which used non-selective ACAT inhibitors, these trials did not report benefits, and one showed an increased risk. Early genetic studies have implicated common variants in both genes with human traits, including lipoprotein levels, coronary artery disease, and Alzheimer’s disease; however, modern genome-wide association studies have not replicated these associations. In contrast, the common SOAT1 variants are most reproducibly associated with testosterone levels. Keywords: cholesterol esterification; atherosclerosis; ACAT; SOAT; inhibitors; clinical trial Citation: Hai, Q.; Smith, J.D. Acyl-Coenzyme A: Cholesterol Acyltransferase (ACAT) in Cholesterol Metabolism: From Its 1. Introduction Discovery to Clinical Trials and the The acyl-coenzyme A:cholesterol acyltransferase (ACAT; EC 2.3.1.26) enzyme family Genomics Era. Metabolites 2021, 11, consists of membrane-spanning proteins, which are primarily located in the endoplasmic 543. https://doi.org/10.3390/ reticulum [1]. -

Pgx - Cytochrome P450 2C19 (CYP2C19)
PGx - Cytochrome P450 2C19 (CYP2C19) This assay is used to identify patients who may be at risk for altered metabolism of drugs that are modified by CYP2C19. Cytochrome P450 (CYP) isozyme 2C19 is responsible for phase I metabloism of about 40% of drugs including: clopidogrel, phenytoin, diazepam, R-warfarin, tamoxifen, some antidepressants, proton pump inhibitors, and antimalarials. Testing Method and Background This test utilizes the eSensor® 2C19 Genotyping Test is an in vitro diagnostic for the detection and genotyping a panel of variants involved with enzyme metabolism using isolated genomic DNA. The eSensor® technology uses a solid-phase electrochemical method for determining the genotyping status. The eSensor® 2C19 Genotyping Test is for research use only (RUO). CYP2C19 drug metabolism varies among individuals depending on specific genotype. The CYP2 family has many single- nucleotide polymorphisms (SNPs). CYP2C19 gene is located on chromosome 10q23.33 and is highly polymorphic, which can lead to large individual variation in CYP2C19 enzyme activity and related drug response phenotypes and/or undesired adverse drug events. Specifically, the CYP2C19 gene has more than 34 variant alleles identified, which in turn affects the pharmacokinetics of many drugs. Highlights of PGx - Cytochrome P450 2C19 (CYP2C19) Targeted Region CYP2C19: Detects 11 variants/polymorphisms 681G>A (*2) 636G>A (*3) 1A>G (*4) 1297C>T (*5) 395G>A (*6) 19294T>A (*7) 358T>C (*8) 431G>A (*9) 680C>T (*10) 1228C>T (*13) -806C>T (*17) Ordering Information Get started -

Mining for Oxysterols in Cyp7b1-/- Mouse Brain and Plasma
biomolecules Communication − − Mining for Oxysterols in Cyp7b1 / Mouse Brain and Plasma: Relevance to Spastic Paraplegia Type 5 Anna Meljon 1,2, Peter J. Crick 1, Eylan Yutuc 1 , Joyce L. Yau 3, Jonathan R. Seckl 3, Spyridon Theofilopoulos 1,4 , Ernest Arenas 4, Yuqin Wang 1 and William J. Griffiths 1,* 1 Swansea University Medical School, ILS1 Building, Singleton Park, Swansea SA2 8PP, UK; [email protected] (A.M.); [email protected] (P.J.C.); [email protected] (E.Y.); s.theofi[email protected] (S.T.); [email protected] (Y.W.) 2 Institute for Global Food Security, Queens University Belfast, Stranmillis Road, Belfast BT9 5AG, UK 3 Endocrinology Unit, BHF Centre for Cardiovascular Science, The Queen’s Medical Research Institute, University of Edinburgh, 47 Little France Crescent, Edinburgh EH16 4TJ, UK; [email protected] (J.L.Y.); [email protected] (J.R.S.) 4 Laboratory of Molecular Neurobiology, Department of Medical Biochemistry and Biophysics, Karolinska Institutet, SE-17177 Stockholm, Sweden; [email protected] * Correspondence: w.j.griffi[email protected]; Tel.: +44-1792-295562 Received: 6 March 2019; Accepted: 2 April 2019; Published: 13 April 2019 Abstract: Deficiency in cytochrome P450 (CYP) 7B1, also known as oxysterol 7α-hydroxylase, in humans leads to hereditary spastic paraplegia type 5 (SPG5) and in some cases in infants to liver disease. SPG5 is medically characterized by loss of motor neurons in the corticospinal tract. In an effort to gain a better understanding of the fundamental biochemistry of this disorder, we have extended our previous profiling of the oxysterol content of brain and plasma of Cyp7b1 knockout (-/-) mice to include, amongst other sterols, 25-hydroxylated cholesterol metabolites. -

Polymorphic Human Sulfotransferase 2A1 Mediates the Formation of 25-Hydroxyvitamin
Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2018/01/17/dmd.117.078428.DC1 1521-009X/46/4/367–379$35.00 https://doi.org/10.1124/dmd.117.078428 DRUG METABOLISM AND DISPOSITION Drug Metab Dispos 46:367–379, April 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Polymorphic Human Sulfotransferase 2A1 Mediates the Formation of 25-Hydroxyvitamin D3-3-O-Sulfate, a Major Circulating Vitamin D Metabolite in Humans s Timothy Wong, Zhican Wang, Brian D. Chapron, Mizuki Suzuki, Katrina G. Claw, Chunying Gao, Robert S. Foti, Bhagwat Prasad, Alenka Chapron, Justina Calamia, Amarjit Chaudhry, Erin G. Schuetz, Ronald L. Horst, Qingcheng Mao, Ian H. de Boer, Timothy A. Thornton, and Kenneth E. Thummel Departments of Pharmaceutics (T.W., Z.W., B.D.C., M.S., K.G.C., C.G., B.P., Al.C., J.C., Q.M., K.E.T.), Medicine and Kidney Research Institute (I.H.d.B.), and Biostatistics (T.A.T.), University of Washington, Seattle, Washington; Department of Pharmacokinetics and Drug Metabolism, Amgen Inc., South San Francisco, California (Z.W.); Department of Pharmacokinetics and Drug Metabolism, Amgen Inc., Cambridge, Massachusetts (R.S.F.); St. Jude Children’s Research Hospital, Memphis, Tennessee Downloaded from (Am.C., E.G.S.); and Heartland Assays LLC, Ames, Iowa (R.L.H.) Received September 1, 2017; accepted January 10, 2018 ABSTRACT dmd.aspetjournals.org Metabolism of 25-hydroxyvitamin D3 (25OHD3) plays a central role in with the rates of dehydroepiandrosterone sulfonation. Further analysis regulating the biologic effects of vitamin D in the body. -

Frequencies of Clinically Important CYP2C19 and CYP2D6 Alleles Are Graded Across Europe
European Journal of Human Genetics (2020) 28:88–94 https://doi.org/10.1038/s41431-019-0480-8 ARTICLE Frequencies of clinically important CYP2C19 and CYP2D6 alleles are graded across Europe 1,2 2 1 Jelena Petrović ● Vesna Pešić ● Volker M. Lauschke Received: 15 March 2019 / Revised: 3 July 2019 / Accepted: 17 July 2019 / Published online: 29 July 2019 © The Author(s) 2019. This article is published with open access Abstract CYP2C19 and CYP2D6 are important drug-metabolizing enzymes that are involved in the metabolism of around 30% of all medications. Importantly, the corresponding genes are highly polymorphic and these genetic differences contribute to interindividual and interethnic differences in drug pharmacokinetics, response, and toxicity. In this study we systematically analyzed the frequency distribution of clinically relevant CYP2C19 and CYP2D6 alleles across Europe based on data from 82,791 healthy individuals extracted from 79 original publications and, for the first time, provide allele confidence intervals for the general population. We found that frequencies of CYP2D6 gene duplications showed a clear South-East to North- West gradient ranging from <1% in Sweden and Denmark to 6% in Greece and Turkey. In contrast, an inverse distribution was observed for the loss-of-function alleles CYP2D6*4 and CYP2D6*5. Similarly, frequencies of the inactive CYP2C19*2 allele were graded from North-West to South-East Europe. In important contrast to previous work we found that the increased activity allele CYP2C19*17 was most prevalent in Central Europe (25–33%) with lower prevalence in Mediterranean-South Europeans (11–24%). In summary, we provide a detailed European map of common CYP2C19 and CYP2D6 variants and find that frequencies of the most clinically relevant alleles are geographically graded reflective of Europe’s migratory history. -

Synonymous Single Nucleotide Polymorphisms in Human Cytochrome
DMD Fast Forward. Published on February 9, 2009 as doi:10.1124/dmd.108.026047 DMD #26047 TITLE PAGE: A BIOINFORMATICS APPROACH FOR THE PHENOTYPE PREDICTION OF NON- SYNONYMOUS SINGLE NUCLEOTIDE POLYMORPHISMS IN HUMAN CYTOCHROME P450S LIN-LIN WANG, YONG LI, SHU-FENG ZHOU Department of Nutrition and Food Hygiene, School of Public Health, Peking University, Beijing 100191, P. R. China (LL Wang & Y Li) Discipline of Chinese Medicine, School of Health Sciences, RMIT University, Bundoora, Victoria 3083, Australia (LL Wang & SF Zhou). 1 Copyright 2009 by the American Society for Pharmacology and Experimental Therapeutics. DMD #26047 RUNNING TITLE PAGE: a) Running title: Prediction of phenotype of human CYPs. b) Author for correspondence: A/Prof. Shu-Feng Zhou, MD, PhD Discipline of Chinese Medicine, School of Health Sciences, RMIT University, WHO Collaborating Center for Traditional Medicine, Bundoora, Victoria 3083, Australia. Tel: + 61 3 9925 7794; fax: +61 3 9925 7178. Email: [email protected] c) Number of text pages: 21 Number of tables: 10 Number of figures: 2 Number of references: 40 Number of words in Abstract: 249 Number of words in Introduction: 749 Number of words in Discussion: 1459 d) Non-standard abbreviations: CYP, cytochrome P450; nsSNP, non-synonymous single nucleotide polymorphism. 2 DMD #26047 ABSTRACT Non-synonymous single nucleotide polymorphisms (nsSNPs) in coding regions that can lead to amino acid changes may cause alteration of protein function and account for susceptivity to disease. Identification of deleterious nsSNPs from tolerant nsSNPs is important for characterizing the genetic basis of human disease, assessing individual susceptibility to disease, understanding the pathogenesis of disease, identifying molecular targets for drug treatment and conducting individualized pharmacotherapy. -

Cytochrome P450 2C19 Loss-Of-Function Polymorphism Is Associated with an Increased Treatment-Related Mortality in Patients Undergoing Allogeneic Transplantation
Bone Marrow Transplantation (2007) 40, 659–664 & 2007 Nature Publishing Group All rights reserved 0268-3369/07 $30.00 www.nature.com/bmt ORIGINAL ARTICLE Cytochrome P450 2C19 loss-of-function polymorphism is associated with an increased treatment-related mortality in patients undergoing allogeneic transplantation AH Elmaagacli, M Koldehoff, NK Steckel, R Trenschel, H Ottinger and DW Beelen Department of Bone Marrow Transplantation, University Hospital of Essen, Essen, Germany The polymorphic gene expression of CYP2C19 causes characterized enzymes with a large number of genetic individual variability in drug metabolism and thereby in polymorphisms is cytochrome P450 (CYP) 2C19.1 Pre- pharmacologic and toxicologic responses. We genotyped vious studies on CYP2C19 using probe drugs such as 286 patients and their donors for the CYP2C19 gene who S-mephenytoin showed that individuals could be classified underwent allogeneic transplantation for various diseases into three different groups: poor metabolizers (PMs), and analyzed their outcome. Patients were classified as: intermediate metabolizers (IMs) and extensive metabolizers poor metabolizers (PMs; 3.1%), intermediate metaboli- (EMs). PMs have a genetically determined absence of active zers (IMs; 24.5%) and extensive metabolizers (EMs; enzymes, which is the cause for a slower metabolism of 72.5%). Patients genotyped as PMs had significant higher active drugs and is associated with prolonged side effects.1 hepato- and nephrotoxicities compared to IMs or EMs. If prodrugs are applied, the metabolism in their active form Maximum bilirubin and serum creatinine levels measured is delayed and reduced effectiveness may occur. Among the after transplant were approximately twofold higher than drugs which are metabolized by CYP2C19 enzymes are those of EMs or IMs. -
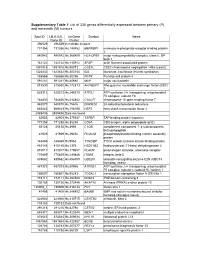
Supplementary Table 1 List of 335 Genes Differentially Expressed Between Primary (P) and Metastatic (M) Tumours
Supplementary Table 1 List of 335 genes differentially expressed between primary (P) and metastatic (M) tumours Spot ID I.M.A.G.E. UniGene Symbol Name Clone ID Cluster 296529 296529 In multiple clusters 731356 731356 Hs.140452 M6PRBP1 mannose-6-phosphate receptor binding protein 1 840942 840942 Hs.368409 HLA-DPB1 major histocompatibility complex, class II, DP beta 1 142122 142122 Hs.115912 AFAP actin filament associated protein 1891918 1891918 Hs.90073 CSE1L CSE1 chromosome segregation 1-like (yeast) 1323432 1323432 Hs.303154 IDS iduronate 2-sulfatase (Hunter syndrome) 788566 788566 Hs.80296 PCP4 Purkinje cell protein 4 591281 591281 Hs.80680 MVP major vault protein 815530 815530 Hs.172813 ARHGEF7 Rho guanine nucleotide exchange factor (GEF) 7 825312 825312 Hs.246310 ATP5J ATP synthase, H+ transporting, mitochondrial F0 complex, subunit F6 784830 784830 Hs.412842 C10orf7 chromosome 10 open reading frame 7 840878 840878 Hs.75616 DHCR24 24-dehydrocholesterol reductase 669443 669443 Hs.158195 HSF2 heat shock transcription factor 2 2485436 2485436 Data not found 82903 82903 Hs.370937 TAPBP TAP binding protein (tapasin) 771258 771258 Hs.85258 CD8A CD8 antigen, alpha polypeptide (p32) 85128 85128 Hs.8986 C1QB complement component 1, q subcomponent, beta polypeptide 41929 41929 Hs.39252 PICALM phosphatidylinositol binding clathrin assembly protein 148469 148469 Hs.9963 TYROBP TYRO protein tyrosine kinase binding protein 415145 415145 Hs.1376 HSD11B2 hydroxysteroid (11-beta) dehydrogenase 2 810017 810017 Hs.179657 PLAUR plasminogen activator, -

Polyclonal Antibody to CYP7B1 (C-Term) - Aff - Purified
OriGene Technologies, Inc. OriGene Technologies GmbH 9620 Medical Center Drive, Ste 200 Schillerstr. 5 Rockville, MD 20850 32052 Herford UNITED STATES GERMANY Phone: +1-888-267-4436 Phone: +49-5221-34606-0 Fax: +1-301-340-8606 Fax: +49-5221-34606-11 [email protected] [email protected] AP20144PU-N Polyclonal Antibody to CYP7B1 (C-term) - Aff - Purified Alternate names: 25-hydroxycholesterol 7-alpha-hydroxylase, Cytochrome P450 7B1, Oxysterol 7-alpha- hydroxylase Quantity: 0.1 mg Concentration: 0.5 mg/ml Background: P450 enzymes constitute a family of monooxygenase enzymes that are involved in the metabolism of a wide array of endogenous and xenobiotic compounds including cholesterol. CYP8B1 moderates the ratio of cholic acid over chenodeoxycholic acid to control the solubility of cholesterol. P450 cholesterol 7-hydroxylase, CYP7A1, is the rate limiting enzyme of bile acid synthesis in the liver, and its expression is mediated by the bile acid receptor FXR. CYP27A1 catalyzes vitamin D3 25-hydroxylation and is localized to the mitochondria in kidney and liver. CYP7B1 (oxysterol 7-α-hydroxylase) functions as an enzyme in the alternate bile acid synthesis pathway. Specifically, CYP7B1 metabolizes 25-and 27-hydroxycholesterol. The gene encoding human CYP7B1 maps to chromosome 8q21.3. Mutations in the CYP7B1 gene may cause a metabolic defect in bile acid synthesis characterized by elevated urinary bile acid excretion, severe cholestasis, cirrhosis and liver synthetic failure. Uniprot ID: O75881 NCBI: NP_004811 GeneID: 9420 Host: Goat Immunogen: Peptide with sequence C-YPDSDVLFRYKVKS, from the C Terminus of the protein sequence according to NP_004811.1. Format: State: Liquid purified IgG fraction.