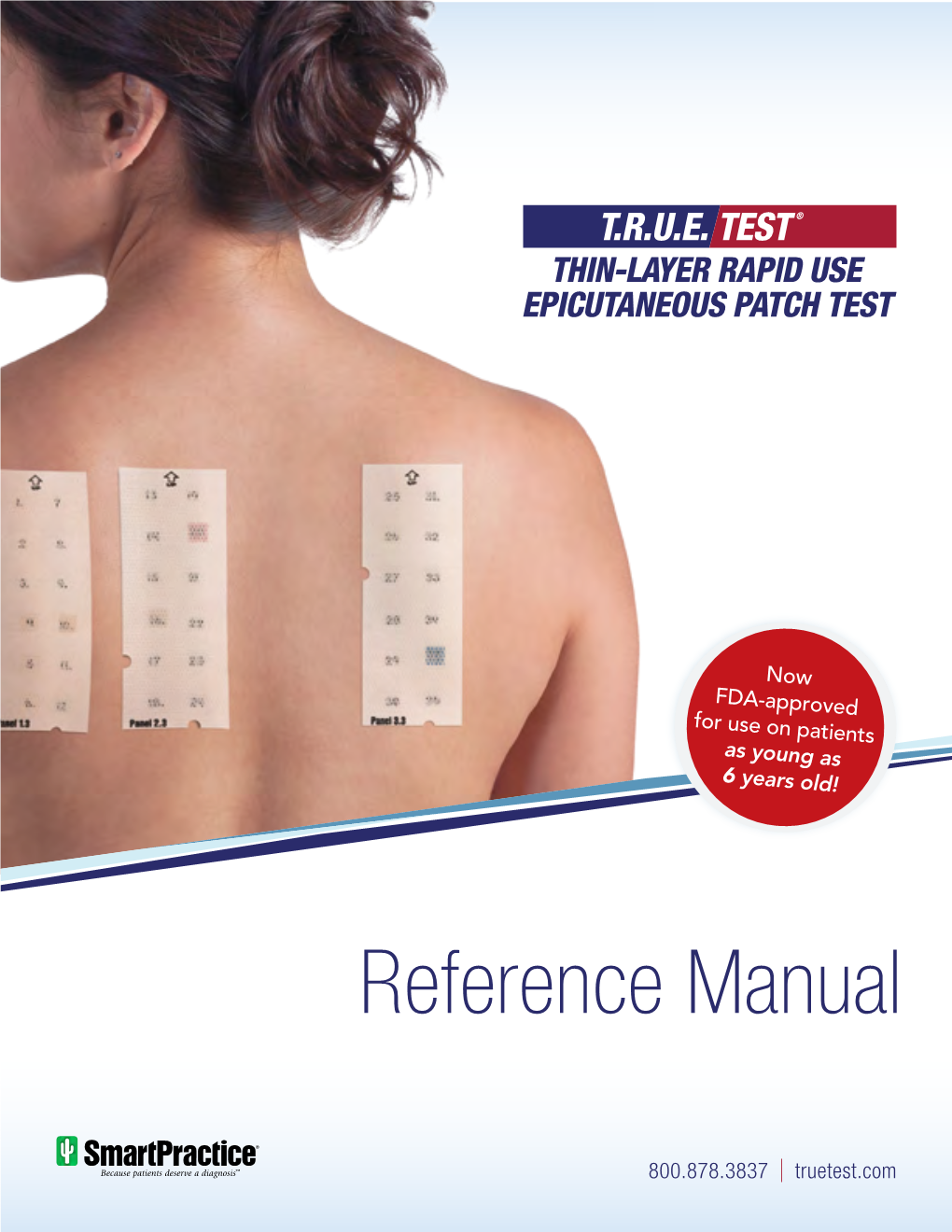
T.R.U.E. TEST Reference Manual
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Fcer11/CD23 Receptor Distribution in Patch Test Reactions to Aeroallergens in Atopic Dermatitis
FceRll/CD23 Receptor Distribution in Patch Test Reactions to Aeroallergens in Atopic Dermatitis Colin C. Buckley, Carol Ivison, Leonard W. Poulter, and Malcolm H.A. Rustin Departments of Dermatology and Immunology (LWP), The Royal Free Hospital and School of Medicine, London, U.K. There is increasing evidence that exposure to organic aller The numbers of Langer hans cells were reduced in the epider gens may induce or exacerbate lesional skin in patients with mis :and increased in the dermis in patch test reactions and atopic dermatitis. In this study, patients with atopic dermati lesional skin compared to their controls. Double staining tis were patch tested to 11 common organic allergens and to reve:aled a change in the distribution of CD23 antigen. In control chambers containing 0.4% phenol and 50% glycerin patch test control and non-Iesional biopsies many macro in 0.9% saline. In biopsies from positive patch test reactions, phages and only a few Langerhans cells within the dermal patch test control skin, lesional eczematous and non-lesional infiltrates expressed this antigen. In patch test reaction and skin from atopic individuals, and normal skin from non lesional skin samples, however, the proportion of CD23+ atopic volunteers, the presence and distribution of macro dermal Langerhans cells had increased compared to macro phages (RFD7+), dendritic cells (RFD1 +), and Langerhans phages. Furthermore, in these latter samples an increased cells, and the expression of the low-affinity receptor for IgE proportion of dermal CD 1+ cells -

NDPSC Record of Reasons, Meeting 37, Feb 2003
National Drugs and Poisons Schedule Committee Record of the Reasons 37th Meeting 25-26 February 2003 The Record of the Reasons contains the basis of scheduling decisions and other outcomes arising from the meeting. Please note that the Record of the Reasons includes extracts from the NDPSC minutes which have been edited to remove confidential information. National Drugs and Poisons Schedule Committee Record of the Reasons Meeting 37 - February 2003 i TABLE OF CON TEN TS GLOSSARY.........................................................................................................................IV 2. PROPOSED CHANGES/ADDITIONS TO PARTS 1 TO 3 AND PART 5 OF THE STANDARD FOR THE UNIFORM SCHEDULING OF DRUGS AND POISONS. ........................9 2.1 SUSDP, PART 1 ..........................................................................................................9 2.2 SUSDP, PART 2 ..........................................................................................................9 2.3 SUSDP, PART 3 ..........................................................................................................9 2.3.1 Organotin Compounds...........................................................................................9 2.4.1 BORON TRIFLUORIDE, BIFLUORIDES AND HYDROSILICOFLUORIC ACID – APPENDIX F PART 3 ...9 AGRICULTURAL/VETERINARY, INDUSTRIAL AND DOMESTIC CHEMICALS.................. 11 3. MATTERS ARISING FROM THE MINUTES OF THE PREVIOUS MEETING (CONSIDERATION OF POST-MEETING SUBMISSIONS UNDER 42ZCZ)............................. -

International Survey on Skin Patch Test Procedures, Attitudes and Interpretation Luciana K
Tanno et al. World Allergy Organization Journal (2016) 9:8 DOI 10.1186/s40413-016-0098-z ORIGINAL RESEARCH Open Access International survey on skin patch test procedures, attitudes and interpretation Luciana K. Tanno1*, Razvigor Darlenski2, Silvia Sánchez-Garcia3, Matteo Bonini4, Andrea Vereda5, Pavel Kolkhir6, Dario Antolin-Amerigo7, Vesselin Dimov8, Claudia Gallego-Corella9, Juan Carlos Aldave Becerra10, Alexander Diaz11, Virginia Bellido Linares12, Leonor Villa13, Lanny J. Rosenwasser14, Mario Sanchez-Borges15, Ignacio Ansotegui16, Ruby Pawankar17, Thomas Bieber18 and on behalf of the WAO Junior Members Group Abstract Background: Skin patch test is the gold standard method in diagnosing contact allergy. Although used for more than 100 years, the patch test procedure is performed with variability around the world. A number of factors can influence the test results, namely the quality of reagents used, the timing of the application, the patch test series (allergens/haptens) that have been used for testing, the appropriate interpretation of the skin reactions or the evaluation of the patient’s benefit. Methods: We performed an Internet –based survey with 38 questions covering the educational background of respondents, patch test methods and interpretation. The questionnaire was distributed among all representatives of national member societies of the World Allergy Organization (WAO), and the WAO Junior Members Group. Results: One hundred sixty-nine completed surveys were received from 47 countries. The majority of participants had more than 5 years of clinical practice (61 %) and routinely carried out patch tests (70 %). Both allergists and dermatologists were responsible for carrying out the patch tests. We could observe the use of many different guidelines regardless the geographical distribution. -

Allergy and Dermatology Patch Test Clinic (ADPT)
4 Skin Patch Tests – Allergy and Dermatology Patch Test Clinic (ADPT) Questions the doctor or nurse will ask you during your first visit. Your answers can help us with collecting your medical information: • When did your symptoms begin? • Have your symptoms changed over time? • What at home treatments have you used? • How did those treatments work? • What, if anything, appears to make your symptoms worse? • Do allergies run in your family? Skin Patch Tests Plan your schedule Allergy and Dermatology Patch Test Clinic (ADPT) Patches are applied on Monday, and then you return on Wednesday for patch removal and Friday for final reading. On Wednesday wear an old or dark coloured shirt. We mark the back after the patches are removed and the ink Why have skin patch testing? may rub off on your clothing. The cause of an allergy is usually found based on: Day Length of visit During the visit Special care • Your symptoms, a rash is very common. Monday 30 to 60 minutes Discussion about the No water or sweat on • A history of contact with substances that cause the allergy. history of your the patches for 2 days. These substances are called allergens. rash/symptoms. Physical exam. When the cause of an allergy is not clear, a skin patch test may be done to Patches applied. help find the cause of your allergy. Skin patch tests are done to see if a Wednesday Short visit Patches removed. Wear an old or dark certain substance is causing an allergic skin reaction. 10 to 15 minutes shirt to this visit. -

Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017
Q UO N T FA R U T A F E BERMUDA PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 BR 111 / 2017 The Minister responsible for health, in exercise of the power conferred by section 48A(1) of the Pharmacy and Poisons Act 1979, makes the following Order: Citation 1 This Order may be cited as the Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017. Repeals and replaces the Third and Fourth Schedule of the Pharmacy and Poisons Act 1979 2 The Third and Fourth Schedules to the Pharmacy and Poisons Act 1979 are repealed and replaced with— “THIRD SCHEDULE (Sections 25(6); 27(1))) DRUGS OBTAINABLE ONLY ON PRESCRIPTION EXCEPT WHERE SPECIFIED IN THE FOURTH SCHEDULE (PART I AND PART II) Note: The following annotations used in this Schedule have the following meanings: md (maximum dose) i.e. the maximum quantity of the substance contained in the amount of a medicinal product which is recommended to be taken or administered at any one time. 1 PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 mdd (maximum daily dose) i.e. the maximum quantity of the substance that is contained in the amount of a medicinal product which is recommended to be taken or administered in any period of 24 hours. mg milligram ms (maximum strength) i.e. either or, if so specified, both of the following: (a) the maximum quantity of the substance by weight or volume that is contained in the dosage unit of a medicinal product; or (b) the maximum percentage of the substance contained in a medicinal product calculated in terms of w/w, w/v, v/w, or v/v, as appropriate. -

Diazolidinyl Urea (Germall® II)
the art and science of smart patch testingTM AP87: Diazolidinyl urea (Germall® II) Patient Information Your patch test result indicates that you have a contact allergy to diazolidinyl urea. This contact allergy may cause your skin to react when it is exposed to this substance although it may take several days for the symptoms to appear. Typical symptoms include redness, swelling, itching, and fluid-filled blisters. Where is diazolidinyl urea found? Diazolidinyl urea is found in cosmetics, skin care products, shampoos, bubble baths, baby wipes, and household detergents. It is used as a preservative and acts as a formaldehyde releaser. How can you avoid contact with diazolidinyl urea? Avoid products that list any of the following names in the ingredients: Diazolidinyl urea N-(Hydroxymethyl)-N-(1,3- EINECS 278-928-2 dihydroxymethyl-2,5-dioxo-4- Germall 11 imidazolidinyl)- Imidazolidinyl urea 11 N'-(hydroxymethyl) urea N-(1,3-Bis(hydroxymethyl)-2,5- N-(Hydroxymethyl)-N-(1,3- dioxo-4-imidazolidinyl)-N,N'- dihydroxymethyl-2,5-dioxo-4- bis(hydroxymethyl)urea imidazolidinyl)- N'-(hydroxymethyl)urea What are some products that may contain diazolidinyl urea? Body Washes: Herbal Essence Body Wash (Dry Skin and Normal) Herbal Essence Fruit Fusions Moisturizing Body Wash Vitabath Bath & Shower Gelee Moisturizing Bath Gel, Original Spring Green Vitabath Bath & Shower Gelee, Moisturizing Bath Gel, Plus for Dry Skin Vitabath Bath & Shower Gelee Moisturizing Bath/Shower Gel, Plus Dry Skin Cleansers: pHisoderm Antibacterial Skin Cleanser Cosmetics: -

Allergy Testing
Effective February 26, 2018 Policy Replaced by LCD L33417 Name of Blue Advantage Policy: Allergy Testing Policy #: 071 Latest Review Date: February 2017 Category: Laboratory/Medical Policy Grade: D Background: Blue Advantage medical policy does not conflict with Local Coverage Determinations (LCDs), Local Medical Review Policies (LMRPs) or National Coverage Determinations (NCDs) or with coverage provisions in Medicare manuals, instructions or operational policy letters. In order to be covered by Blue Advantage the service shall be reasonable and necessary under Title XVIII of the Social Security Act, Section 1862(a)(1)(A). The service is considered reasonable and necessary if it is determined that the service is: 1. Safe and effective; 2. Not experimental or investigational*; 3. Appropriate, including duration and frequency that is considered appropriate for the service, in terms of whether it is: • Furnished in accordance with accepted standards of medical practice for the diagnosis or treatment of the patient’s condition or to improve the function of a malformed body member; • Furnished in a setting appropriate to the patient’s medical needs and condition; • Ordered and furnished by qualified personnel; • One that meets, but does not exceed, the patient’s medical need; and • At least as beneficial as an existing and available medically appropriate alternative. *Routine costs of qualifying clinical trial services with dates of service on or after September 19, 2000 which meet the requirements of the Clinical Trials NCD are considered reasonable and necessary by Medicare. Providers should bill Original Medicare for covered services that are related to clinical trials that meet Medicare requirements (Refer to Medicare National Coverage Determinations Manual, Chapter 1, Section 310 and Medicare Claims Processing Manual Chapter 32, Sections 69.0-69.11). -

Dermatology Patch Allergy Testing Post Service - Information Request Form
Dermatology Patch Allergy Testing Post Service - Information Request Form Blue Cross NC will review associated claim(s) for services rendered on the patient listed below. In order to determine benefits are available for the reported condition, please answer the questions below. If you would prefer to send medical records, relating to the condition for the dates listed you may do so. In this case, all answers must be supported by documentation in the patient's medical record. Please submit the completed form to Blue Cross NC per the Medical Record Submission instructions found on the bcbsnc.com provider site (https://www.bcbsnc.com/assets/providers/public/pdfs/submissions/how_to_submit_provider_initiated_medical_records.pdf) or if requested by Blue Cross NC via a bar-coded coversheet, please fax the form/medical records to the number noted on the bar-coded cover sheet within 7-10 days to facilitate the claim payment. This form must be filled out by the patient's physician or their designee which may be any of the following: Physician Assistant (PA), Nurse Practitioner (NP), Registered Nurse (RN), or Licensed Practical Nurse (LPN). Note: Credentials must be provided with signature or the form will be returned. PROVIDER INFORMATION Requesting Provider Information Place of Service Provider Name Facility Name Provider ID Facility ID PATIENT INFORMATION Patient Name:_____________________________ Patient DOB :_____________ Patient Account Number______________ Patient ID:______________ CLAIM INFORMATION Date(s) of Service_____________ CPT_________ Diagnosis_______ Dermatology Patch Allergy Testing - Information Request Form Page 1 of 3 CLINICAL INFORMATION Did the patient have direct skin testing (for immediate hypersensitivity) by: Percutaneous or epicutaneous (scratch, prick, or puncture)? ________________ Intradermal testing? ___________________________________ Inhalant allergy evaluation? _________________ Did the patient have patch (application) testing (most commonly used: T.R.U.E. -

Allergy Skin and Challenge Testing
Corporate Medical Policy Allergy Skin and Challenge Testing File Name: allergy_skin_and_challenge_testing Origination: 7/1979 Last CAP Review: 11/2020 Next CAP Review: 11/2021 Last Review: 11/2020 Description of Procedure or Service Management of the allergic patient may include identifying the offending agent by means of allergy testing. Allergy testing can be broadly grouped into in vivo and in vitro methodologies: • In vivo testing - includes allergy skin testing such as the scratch, puncture or prick test (epicutaneous), intradermal test (intracutaneous) and patch test, and food and bronchial challenges. • In vitro testing - includes various techniques to test the blood for the presence of specific IgE antibodies to a particular antigen. Once the agent is identified, treatment is provided by avoidance, medication or immunotherapy (allergy shots). Allergic or hypersensitivity disorders may be manifested by generalized systemic reactions as well as by localized reactions in any organ system of the body. The reactions may be acute, subacute, or chronic, immediate or delayed, and may be caused by numerous offending agents: pollen, molds, dust mites, animal dander, stinging insect venoms, foods, drugs, etc. This policy only addresses in vivo (skin and challenge) testing; for vitro testing, see policy titled, Allergen Testing. Related Policies: Allergy Immunotherapy Desensitization Allergen Testing AHS – G2031 Maximum Units of Service Idi ***Note: This Medical Policy is complex and technical. For questions concerning the technical language and/or specific clinical indications for its use, please consult your physician. Policy BCBSNC will provide coverage for Allergy skin and challenge testing when it is determined to be medically necessary because the medical criteria and guidelines shown below are met. -

Guideline on Diagnostic Procedures for Suspected Hypersensitivity to Beta-Lactam Antibiotics
published by: guidelines Allergo J Int https://doi.org/10.1007/s40629-019-0100-8 Guideline on diagnostic procedures for suspected hypersensitivity to beta-lactam antibiotics Guideline of the German Society for Allergology and Clinical Immunology (DGAKI) in collaboration with the German Society of Allergology (AeDA), German Society for Pediatric Allergology and Environmental Medicine (GPA), the German Contact Dermatitis Research Group (DKG), the Austrian Society for Allergology and Immunology (ÖGAI), and the Paul-Ehrlich Society for Chemotherapy (PEG) Gerda Wurpts · Werner Aberer · Heinrich Dickel · Randolf Brehler · Thilo Jakob · Burkhard Kreft · Vera Mahler · Hans F. Merk · Norbert Mülleneisen · Hagen Ott · Wolfgang Pfützner · Stefani Röseler · Franziska Ruëff · Helmut Sitter · Cord Sunderkötter · Axel Trautmann · Regina Treudler · Bettina Wedi · Margitta Worm · Knut Brockow © The Author(s) 2019 Electronic supplementary material The online version of this article (https://doi.org/10.1007/s40629-019-0100-8) contains supplementary material, which is available to authorized users. Dr. G. Wurpts () · H. F.Merk · S. Röseler V. Ma h l e r Clinic for Dermatology and Allergology, Aachen Paul-Ehrlich Institute, Langen, Germany Comprehensive Allergy Center (ACAC), Uniklinik RWTH Department of Dermatology, University Hospital Erlangen, Aachen, Pauwelsstraße 30, 52074 Aachen, Germany Erlangen, Germany [email protected] N. Mülleneisen W. Aberer Asthma and Allergy Centre, Leverkusen, Germany Department of Dermatology, Graz Medical University, Graz, Austria H. Ott Division of Pediatric Dermatology and Allergology, Auf der H. Dickel Bult Children’s Hospital, Hannover, Germany Department of Dermatology, Venereology and Allergology, St. Josef Hospital, University Hospital of the Ruhr University W. Pfützner Bochum, Bochum, Germany Department of Dermatology and Allergology, University Hospital Gießen und Marburg, Marburg Site, Marburg, R. -

University of Groningen Formaldehyde-Releasers In
University of Groningen Formaldehyde-releasers in cosmetics de Groot, Anton C.; White, Ian R.; Flyvholm, Mari-Ann; Lensen, Gerda; Coenraads, Pieter- Jan Published in: CONTACT DERMATITIS IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 2010 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): de Groot, A. C., White, I. R., Flyvholm, M-A., Lensen, G., & Coenraads, P-J. (2010). Formaldehyde- releasers in cosmetics: relationship to formaldehyde contact allergy Part 1. Characterization, frequency and relevance of sensitization, and frequency of use in cosmetics. CONTACT DERMATITIS, 62(1), 18-31. Copyright Other than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). The publication may also be distributed here under the terms of Article 25fa of the Dutch Copyright Act, indicated by the “Taverne” license. More information can be found on the University of Groningen website: https://www.rug.nl/library/open-access/self-archiving-pure/taverne- amendment. Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from the University of Groningen/UMCG research database (Pure): http://www.rug.nl/research/portal. -

The Dynamics of Technological Innovation: the Case of the Pharmaceutical Industry
Research Policy 30Ž. 2001 535–588 www.elsevier.nlrlocatereconbase The dynamics of technological innovation: the case of the pharmaceutical industry Basil Achilladelis a,), Nicholas Antonakis b a 39, Ionias Street, Kifissia, Athens 14563, Greece b Ministry of DeÕelopment and UniÕersity of Athens, Athens Greece Received 9 April 1999; received in revised form 7 October 1999; accepted 18 January 2000 Abstract This is an empirical and historical study of the dynamics of technological innovationŽ. TI in the pharmaceutical industry from its establishment at the beginning of the 19th century to 1990. It is based on the identification and evaluation of the originality and commercial significance of 1736 product innovationsŽ. new medicines commercialized between 1800 and 1990, and on company economic data for the period 1950–1990. The study is presented in the framework of established macroeconomic theory of technical change. Applying both empirical and historical evidence, the study:Ž. a identifies the technological, social and economic driving forces for TI;Ž. b examines the relation between originality and market performance of medicinal innovations;Ž. c studies the mechanisms of the diffusion of medicinal technologies that led to the formation of five successive generations of drugsŽ long waves.Ž. ; d describes the structural changes forced on the pharmaceutical industry by the introduction and development of each successive generation of drugs;Ž. e provides evidence of the concentration of the innovative segment of the pharmaceutical industry among few large companies, which sustained high levels of growth and R&D expenditures by means of inhouse innovation, technological and therapeutic market specialization, and mergers and acquisitions of companies within and outside the pharmaceutical industry; andŽ.