Imaging of the Central Skull Base
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Middle Cranial Fossa Sphenoidal Region Dural Arteriovenous Fistulas: Anatomic and Treatment Considerations
ORIGINAL RESEARCH INTERVENTIONAL Middle Cranial Fossa Sphenoidal Region Dural Arteriovenous Fistulas: Anatomic and Treatment Considerations Z.-S. Shi, J. Ziegler, L. Feng, N.R. Gonzalez, S. Tateshima, R. Jahan, N.A. Martin, F. Vin˜uela, and G.R. Duckwiler ABSTRACT BACKGROUND AND PURPOSE: DAVFs rarely involve the sphenoid wings and middle cranial fossa. We characterize the angiographic findings, treatment, and outcome of DAVFs within the sphenoid wings. MATERIALS AND METHODS: We reviewed the clinical and radiologic data of 11 patients with DAVFs within the sphenoid wing that were treated with an endovascular or with a combined endovascular and surgical approach. RESULTS: Nine patients presented with ocular symptoms and 1 patient had a temporal parenchymal hematoma. Angiograms showed that 5 DAVFs were located on the lesser wing of sphenoid bone, whereas the other 6 were on the greater wing of the sphenoid bone. Multiple branches of the ICA and ECA supplied the lesions in 7 patients. Four patients had cortical venous reflux and 7 patients had varices. Eight patients were treated with transarterial embolization using liquid embolic agents, while 3 patients were treated with transvenous embo- lization with coils or in combination with Onyx. Surgical disconnection of the cortical veins was performed in 2 patients with incompletely occluded DAVFs. Anatomic cure was achieved in all patients. Eight patients had angiographic and clinical follow-up and none had recurrence of their lesions. CONCLUSIONS: DAVFs may occur within the dura of the sphenoid wings and may often have a presentation similar to cavernous sinus DAVFs, but because of potential associations with the cerebral venous system, may pose a risk for intracranial hemorrhage. -

MR Imaging of the Orbital Apex
J Korean Radiol Soc 2000;4 :26 9-0 6 1 6 MR Imaging of the Orbital Apex: An a to m y and Pat h o l o g y 1 Ho Kyu Lee, M.D., Chang Jin Kim, M.D.2, Hyosook Ahn, M.D.3, Ji Hoon Shin, M.D., Choong Gon Choi, M.D., Dae Chul Suh, M.D. The apex of the orbit is basically formed by the optic canal, the superior orbital fis- su r e , and their contents. Space-occupying lesions in this area can result in clinical d- eficits caused by compression of the optic nerve or extraocular muscles. Even vas c u l a r changes in the cavernous sinus can produce a direct mass effect and affect the orbit ap e x. When pathologic changes in this region is suspected, contrast-enhanced MR imaging with fat saturation is very useful. According to the anatomic regions from which the lesions arise, they can be classi- fied as belonging to one of five groups; lesions of the optic nerve-sheath complex, of the conal and intraconal spaces, of the extraconal space and bony orbit, of the cav- ernous sinus or diffuse. The characteristic MR findings of various orbital lesions will be described in this paper. Index words : Orbit, diseases Orbit, MR The apex of the orbit is a complex region which con- tains many nerves, vessels, soft tissues, and bony struc- Anatomy of the orbital apex tures such as the superior orbital fissure and the optic canal (1-3), and is likely to be involved in various dis- The orbital apex region consists of the optic nerve- eases (3). -

Gross Anatomy Assignment Name: Olorunfemi Peace Toluwalase Matric No: 17/Mhs01/257 Dept: Mbbs Course: Gross Anatomy of Head and Neck
GROSS ANATOMY ASSIGNMENT NAME: OLORUNFEMI PEACE TOLUWALASE MATRIC NO: 17/MHS01/257 DEPT: MBBS COURSE: GROSS ANATOMY OF HEAD AND NECK QUESTION 1 Write an essay on the carvernous sinus. The cavernous sinuses are one of several drainage pathways for the brain that sits in the middle. In addition to receiving venous drainage from the brain, it also receives tributaries from parts of the face. STRUCTURE ➢ The cavernous sinuses are 1 cm wide cavities that extend a distance of 2 cm from the most posterior aspect of the orbit to the petrous part of the temporal bone. ➢ They are bilaterally paired collections of venous plexuses that sit on either side of the sphenoid bone. ➢ Although they are not truly trabeculated cavities like the corpora cavernosa of the penis, the numerous plexuses, however, give the cavities their characteristic sponge-like appearance. ➢ The cavernous sinus is roofed by an inner layer of dura matter that continues with the diaphragma sellae that covers the superior part of the pituitary gland. The roof of the sinus also has several other attachments. ➢ Anteriorly, it attaches to the anterior and middle clinoid processes, posteriorly it attaches to the tentorium (at its attachment to the posterior clinoid process). Part of the periosteum of the greater wing of the sphenoid bone forms the floor of the sinus. ➢ The body of the sphenoid acts as the medial wall of the sinus while the lateral wall is formed from the visceral part of the dura mater. CONTENTS The cavernous sinus contains the internal carotid artery and several cranial nerves. Abducens nerve (CN VI) traverses the sinus lateral to the internal carotid artery. -

Septation of the Sphenoid Sinus and Its Clinical Significance
1793 International Journal of Collaborative Research on Internal Medicine & Public Health Septation of the Sphenoid Sinus and its Clinical Significance Eldan Kapur 1* , Adnan Kapidžić 2, Amela Kulenović 1, Lana Sarajlić 2, Adis Šahinović 2, Maida Šahinović 3 1 Department of anatomy, Medical faculty, University of Sarajevo, Čekaluša 90, 71000 Sarajevo, Bosnia and Herzegovina 2 Clinic for otorhinolaryngology, Clinical centre University of Sarajevo, Bolnička 25, 71000 Sarajevo, Bosnia and Herzegovina 3 Department of histology and embriology, Medical faculty, University of Sarajevo, Čekaluša 90, 71000 Sarajevo, Bosnia and Herzegovina * Corresponding Author: Eldan Kapur, MD, PhD Department of anatomy, Medical faculty, University of Sarajevo, Bosnia and Herzegovina Email: [email protected] Phone: 033 66 55 49; 033 22 64 78 (ext. 136) Abstract Introduction: Sphenoid sinus is located in the body of sphenoid, closed with a thin plate of bone tissue that separates it from the important structures such as the optic nerve, optic chiasm, cavernous sinus, pituitary gland, and internal carotid artery. It is divided by one or more vertical septa that are often asymmetric. Because of its location and the relationships with important neurovascular and glandular structures, sphenoid sinus represents a great diagnostic and therapeutic challenge. Aim: The aim of this study was to assess the septation of the sphenoid sinus and relationship between the number and position of septa and internal carotid artery in the adult BH population. Participants and Methods: A retrospective study of the CT analysis of the paranasal sinuses in 200 patients (104 male, 96 female) were performed using Siemens Somatom Art with the following parameters: 130 mAs: 120 kV, Slice: 3 mm. -
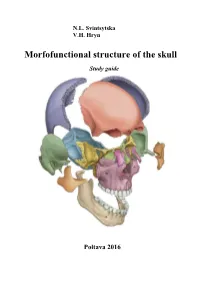
Morfofunctional Structure of the Skull
N.L. Svintsytska V.H. Hryn Morfofunctional structure of the skull Study guide Poltava 2016 Ministry of Public Health of Ukraine Public Institution «Central Methodological Office for Higher Medical Education of MPH of Ukraine» Higher State Educational Establishment of Ukraine «Ukranian Medical Stomatological Academy» N.L. Svintsytska, V.H. Hryn Morfofunctional structure of the skull Study guide Poltava 2016 2 LBC 28.706 UDC 611.714/716 S 24 «Recommended by the Ministry of Health of Ukraine as textbook for English- speaking students of higher educational institutions of the MPH of Ukraine» (minutes of the meeting of the Commission for the organization of training and methodical literature for the persons enrolled in higher medical (pharmaceutical) educational establishments of postgraduate education MPH of Ukraine, from 02.06.2016 №2). Letter of the MPH of Ukraine of 11.07.2016 № 08.01-30/17321 Composed by: N.L. Svintsytska, Associate Professor at the Department of Human Anatomy of Higher State Educational Establishment of Ukraine «Ukrainian Medical Stomatological Academy», PhD in Medicine, Associate Professor V.H. Hryn, Associate Professor at the Department of Human Anatomy of Higher State Educational Establishment of Ukraine «Ukrainian Medical Stomatological Academy», PhD in Medicine, Associate Professor This textbook is intended for undergraduate, postgraduate students and continuing education of health care professionals in a variety of clinical disciplines (medicine, pediatrics, dentistry) as it includes the basic concepts of human anatomy of the skull in adults and newborns. Rewiewed by: O.M. Slobodian, Head of the Department of Anatomy, Topographic Anatomy and Operative Surgery of Higher State Educational Establishment of Ukraine «Bukovinian State Medical University», Doctor of Medical Sciences, Professor M.V. -
The Orbit Is Composed Anteri
DAVID L. PARVER, MD The University of Texas Southwestern Medical Center, Dallas Theability to successfully assess and treat The Orbit physical ailments requires an understanding of the anatomy involved in the injury or The eye itself lies within a protective shell trauma. When dealing with injuries and called the bony orbits. These bony cavities are trauma associated with the eye, it is neces- located on each side of the root of the nose. sary to have a work- Each orbit is structured like a pear with the ing knowledge of optic nerve, the nerve that carries visual im- basic ocular anatomy pulses from the retina to the brain, represent- so that an accurate ing the stem of the orbtt (Duke-Elder, 1976). Understa eye also diagnosis can be Seven bones make up the bony orbit: frontal, achieved and treat- zygomatic, maxillary, ethmoidal, sphenoid, ment can be imple- lacrimal, and palatine (Figures 1 and 2). in a bony " mented. The roof of the orbit is composed anteri- . .. The upcoming ar- orly of the orbital plate of the frontal bone ticles in this special and posteriorly by the lesser wing of the sphe- Each portion of the 01 I noid bone. The lateral wall is separated from .r. theme section the nervc an eye will deal specifically 2 with recognizing ocular illness, disease, and injuries, and will also address the incidence of sports related eye injuries and trauma. This paper covers the ba- sics of eye anatomy, focusing on the eye globe and its surrounding struc- tures. Once one gains an understand- ing of the normal anatomy of the eye, it will be easier to recognize trauma, injury, or illness. -

Clinical Importance of the Middle Meningeal Artery
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Jagiellonian Univeristy Repository FOLIA MEDICA CRACOVIENSIA 41 Vol. LIII, 1, 2013: 41–46 PL ISSN 0015-5616 Przemysław Chmielewski1, Janusz skrzat1, Jerzy waloCha1 CLINICAL IMPORTANCE OF THE MIDDLE MENINGEAL ARTERY Abstract: Middle meningeal artery (MMA)is an important branch which supplies among others cranial dura mater. It directly attaches to the cranial bones (is incorporated into periosteal layer of dura mater), favors common injuries in course of head trauma. This review describes available data on the MMA considering its varability, or treats specific diseases or injuries where the course of MMA may have clinical impact. Key words: Middle meningeal artery (MMA), aneurysm of the middle meningeal artery, epidural he- matoma, anatomical variation of MMA. TOPOGRAPHY OF THE MIDDLE MENINGEAL ARTERY AND ITS BRANCHES Middle meningeal artery (MMA) [1] is most commonly the strongest branch of maxillary artery (from external carotid artery) [2]. It supplies blood to cranial dura mater, and through the numerous perforating branches it nourishes also periosteum of the inner aspect of cranial bones. It enters the middle cranial fossa through the foramen spinosum, and courses between the dura mater and the inner aspect of the vault of the skull. Next it divides into two terminal branches — frontal (anterior) which supplies blood to bones forming anterior cranial fossa and the anterior part of the middle cranial fossa; parietal branch (posterior), which runs more horizontally toward the back and supplies posterior part of the middle cranial fossa and supratentorial part of the posterior cranial fossa. -

Partial Closure of Right Superior Orbital Fissure with Narrow Optic Foramen
eISSN 1308-4038 International Journal of Anatomical Variations (2010) 3: 188–190 Case Report Partial closure of right superior orbital fissure with narrow optic foramen Published online November 26th, 2010 © http://www.ijav.org Shankreppa Doddappa DESAI ABSTRACT Sunkeswari SREEPADMA Superior orbital fissure is situated between the greater and lesser wings of sphenoid, with the optic strut at its superomedial margin. It lies between the roof and lateral wall of the orbit. The superior orbital fissure is divided by the common tendinous origin of the recti muscles. Compression of the neurovascular structures due to variations in the superior orbital fissure may result in signs and symptoms due to involvement of cranial nerves III, IV, V1, and VI. We report here a variation of the superior orbital fissure. Superior orbital fissure was partly closed by a thin plate of bone on the right side, and on the same side there was a narrow optic foramen. It is essential to know such variations to understand the underlying cause for the clinical conditions and operate in those areas. © IJAV. 2010; 3: 188–190. Department of Anatomy, Blde University, Shri B. M. Patil Medical College, Bijapur, Karnataka State, INDIA. Dr. S. D. Desai Professor & Head Department of Anatomy Blde University Shri B. M. Patil Medical College Bijapur-586103, Karnataka, INDIA. +91 8352 262770 ext.:2211 [email protected] Received April 7th, 2010; accepted October 31st, 2010 Key words [superior orbital fissure] [neurological deficits] [superior orbital fissure syndrome] [optic canal] [optic foramen] Introduction in shape and measured 0.9 cm vertically and 1.4 cm Superior orbital fissure is an oblique cleft and connects transversely (Figure 4). -

Evisceration, Enucleation and Exenteration
CHAPTER 10 EVISCERATION, ENUCLEATION AND EXENTERATION This chapter describes three operations that either remove the contents of the eye (evisceration), the eye itself (enucleation) or the whole orbital contents (exenteration). Each operation has specific indications which are important to understand. In many cultures the removal of an eye, even if blind, is resisted. If an eye is very painful or grossly disfigured an operation will be accepted more readily. However, if the eye looks normal the patient or their family may be very reluctant to accept its removal. Therefore tact, compassion and patience are needed when recommending these operations. ENUCLEATION AND EVISCERATION There are several reasons why either of these destructive operations may be necessary: 1. Malignant tumours in the eye. In the case of a malignant tumour or suspected malignant tumour the eye should be removed by enucleation and not evisceration.There are two important intraocular tumours, retinoblastoma and melanoma and for both of them the basic treatment is enucleation. Retinoblastoma is a relatively common tumour in early childhood. At first the growth is confined to the eye. Enucleation must be carried out at this stage and will probably save the child’s life. It is vital not to delay or postpone surgery. If a child under 6 has a blind eye and the possibility of a tumour cannot be ruled out, it is best to remove the eye. Always examine the other eye very carefully under anaesthetic as well. It may contain an early retinoblastoma which could be treatable and still save the eye. Retinoblastoma spreads along the optic nerve to the brain. -

98796-Anatomy of the Orbit
Anatomy of the orbit Prof. Pia C Sundgren MD, PhD Department of Diagnostic Radiology, Clinical Sciences, Lund University, Sweden Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Lay-out • brief overview of the basic anatomy of the orbit and its structures • the orbit is a complicated structure due to its embryological composition • high number of entities, and diseases due to its composition of ectoderm, surface ectoderm and mesoderm Recommend you to read for more details Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 3 x 3 Imaging technique 3 layers: - neuroectoderm (retina, iris, optic nerve) - surface ectoderm (lens) • CT and / or MR - mesoderm (vascular structures, sclera, choroid) •IOM plane 3 spaces: - pre-septal •thin slices extraconal - post-septal • axial and coronal projections intraconal • CT: soft tissue and bone windows 3 motor nerves: - occulomotor (III) • MR: T1 pre and post, T2, STIR, fat suppression, DWI (?) - trochlear (IV) - abducens (VI) Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Superior orbital fissure • cranial nerves (CN) III, IV, and VI • lacrimal nerve • frontal nerve • nasociliary nerve • orbital branch of middle meningeal artery • recurrent branch of lacrimal artery • superior orbital vein • superior ophthalmic vein Lund University / Faculty of Medicine / Inst. Clinical Sciences / Radiology / ECNR Dubrovnik / Oct 2018 Lund University / Faculty of Medicine / Inst. -

Morphology of the Foramen Magnum in Young Eastern European Adults
Folia Morphol. Vol. 71, No. 4, pp. 205–216 Copyright © 2012 Via Medica O R I G I N A L A R T I C L E ISSN 0015–5659 www.fm.viamedica.pl Morphology of the foramen magnum in young Eastern European adults F. Burdan1, 2, J. Szumiło3, J. Walocha4, L. Klepacz5, B. Madej1, W. Dworzański1, R. Klepacz3, A. Dworzańska1, E. Czekajska-Chehab6, A. Drop6 1Department of Human Anatomy, Medical University of Lublin, Lublin, Poland 2St. John’s Cancer Centre, Lublin, Poland 3Department of Clinical Pathomorphology, Medical University of Lublin, Lublin, Poland 4Department of Anatomy, Collegium Medicum, Jagiellonian University, Krakow, Poland 5Department of Psychiatry and Behavioural Sciences, Behavioural Health Centre, New York Medical College, Valhalla NY, USA 6Department of General Radiology and Nuclear Medicine, Medical University of Lublin, Lublin, Poland [Received 21 July 2012; Accepted 7 September 2012] Background: The foramen magnum is an important anatomical opening in the base of the skull through which the posterior cranial fossa communicates with the vertebral canal. It is also related to a number of pathological condi- tions including Chiari malformations, various tumours, and occipital dysplasias. The aim of the study was to evaluate the morphology of the foramen magnum in adult individuals in relation to sex. Material and methods: The morphology of the foramen magnum was evalu- ated using 3D computer tomography images in 313 individuals (142 male, 171 female) aged 20–30 years. Results: The mean values of the foramen length (37.06 ± 3.07 vs. 35.47 ± ± 2.60 mm), breadth (32.98 ± 2.78 vs. 30.95 ± 2.71 mm) and area (877.40 ± ± 131.64 vs. -

Investigating the Various Shapes of Sella Turcica in Nigerian Children Using Lateral Skull Radiographs
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article Investigating the Various Shapes of Sella Turcica in Nigerian Children Using Lateral Skull Radiographs Bello A., Usman J.D. Department of Anatomy, Faculty of Basic Medical Sciences, College of Health Sciences, Usmanu Danfodiyo University, Sokoto, Nigeria. Corresponding Author: Bello A ABSTRACT The knowledge of the normal radiographic anatomy of the sella turcica and sella point is of great importance to clinicians in enabling them quickly recognize, investigate or evaluate any deviation from normal as well as any pathological situation related to the pituitary gland. This study investigated the various shapes of the sella turcica in children. A total of 250 lateral skull radiographs taken in the Department of Radiology, Usmanu Danfodiyo University Teaching Hospital (UDUTH), Sokoto from January 2013 to December 2014 were retrieved for the purpose of this study. Radiographs were mounted on the viewing box and variants of the anatomical shapes of the sella turcica were studied and classified. Of the 162 radiographs used in this study, 114 (70.4%) sella turcica were round shaped while 48 (29.6%) oval in shaped. This observed difference was statistically significant (p<0.001). Meanwhile, the floor of the sella turcica of the study participants showed a concave outline in 130 (80%) of the children and flat outline in 32 (20%) of the children. Sexual dimorphism was seen in this study with respect to shape of sella turcica. Round shaped sella turcica was predominant in Nigerian children used in this study. The prevalence and the relative frequencies of the normal variants of the anatomical shapes of the sella turcica of male Nigerian children differ significantly from those of their female counterparts.