Who Drug Information
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-
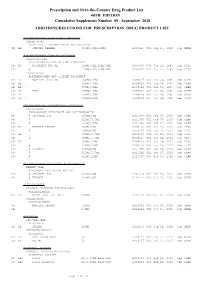
Additions and Deletions to the Drug Product List
Prescription and Over-the-Counter Drug Product List 40TH EDITION Cumulative Supplement Number 09 : September 2020 ADDITIONS/DELETIONS FOR PRESCRIPTION DRUG PRODUCT LIST ACETAMINOPHEN; BUTALBITAL; CAFFEINE TABLET;ORAL BUTALBITAL, ACETAMINOPHEN AND CAFFEINE >A> AA STRIDES PHARMA 325MG;50MG;40MG A 203647 001 Sep 21, 2020 Sep NEWA ACETAMINOPHEN; CODEINE PHOSPHATE SOLUTION;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >D> AA WOCKHARDT BIO AG 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC >A> @ 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC TABLET;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >A> AA NOSTRUM LABS INC 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >A> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >A> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN >D> AA TEVA 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >D> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >D> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN ACETAMINOPHEN; HYDROCODONE BITARTRATE TABLET;ORAL HYDROCODONE BITARTRATE AND ACETAMINOPHEN >A> @ CEROVENE INC 325MG;5MG A 211690 001 Feb 07, 2020 Sep CAHN >A> @ 325MG;7.5MG A 211690 002 Feb 07, 2020 Sep CAHN >A> @ 325MG;10MG A 211690 003 Feb 07, 2020 Sep CAHN >D> AA VINTAGE PHARMS 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >A> @ 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >D> AA 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >A> @ 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >D> AA 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >A> @ 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >D> @ XIROMED 325MG;5MG A 211690 -

Enzymes Handling/Processing
Enzymes Handling/Processing 1 Identification of Petitioned Substance 2 3 This Technical Report addresses enzymes used in used in food processing (handling), which are 4 traditionally derived from various biological sources that include microorganisms (i.e., fungi and 5 bacteria), plants, and animals. Approximately 19 enzyme types are used in organic food processing, from 6 at least 72 different sources (e.g., strains of bacteria) (ETA, 2004). In this Technical Report, information is 7 provided about animal, microbial, and plant-derived enzymes generally, and more detailed information 8 is presented for at least one model enzyme in each group. 9 10 Enzymes Derived from Animal Sources: 11 Commonly used animal-derived enzymes include animal lipase, bovine liver catalase, egg white 12 lysozyme, pancreatin, pepsin, rennet, and trypsin. The model enzyme is rennet. Additional details are 13 also provided for egg white lysozyme. 14 15 Chemical Name: Trade Name: 16 Rennet (animal-derived) Rennet 17 18 Other Names: CAS Number: 19 Bovine rennet 9001-98-3 20 Rennin 25 21 Chymosin 26 Other Codes: 22 Prorennin 27 Enzyme Commission number: 3.4.23.4 23 Rennase 28 24 29 30 31 Chemical Name: CAS Number: 32 Peptidoglycan N-acetylmuramoylhydrolase 9001-63-2 33 34 Other Name: Other Codes: 35 Muramidase Enzyme Commission number: 3.2.1.17 36 37 Trade Name: 38 Egg white lysozyme 39 40 Enzymes Derived from Plant Sources: 41 Commonly used plant-derived enzymes include bromelain, papain, chinitase, plant-derived phytases, and 42 ficin. The model enzyme is bromelain. -

Telaprevir (Incivek)
© Hepatitis C Online PDF created September 25, 2021, 4:19 pm Telaprevir (Incivek) Discontinued. This treatment has been discontinued. Table of Contents Telaprevir Incivek Summary Drug Summary Adverse Effects Class and Mechanism Manufacturer for United States FDA Status Indications Dosing Clinical Use Cost and Medication Access Resistance Key Drug Interactions Full Prescribing Information Figures Drug Summary Although telaprevir was a promising direct-acting antiviral agent that had impact in the hepatitis C treatment field during 2011 to 2013, it was subsequently replaced by newer direct-acting antiviral agents that were more effective, better tolerated, and more convenient. Based on the dwindling role of telaprevir after newer direct-acting antiviral agents were approved, Vertex pharmaceuticals discontinued the sales and distribution of telaprevir in the United States in October 2014. Telaprevir does have some current importance since persons who previously failed a telaprevir-based regimen may have developed resistant associated variants, which could potentially impact subsequent therapy. Adverse Effects The most significant adverse effects reported in the main registration trials and in post-marketing experience were rash, anorectal complaints, and anemia. When comparing triple therapy of telaprevir, peginterferon, and ribavirin with dual therapy of peginterferon and ribavirin alone significant differences were noted with rash (56% versus 34%), anemia (36% versus 17%), and anorectal complaints that include anorectal discomfort, anal pruritus, and hemorrhoids (29% versus 7%). In most cases, the rash that develops is eczematous or maculopapular in character and mild to moderate in severity; the rash is typically manageable with good skin care and topical emollients or corticosteroids. In some instances, however, telaprevir has caused serious skin Page 1/5 rashes, including Steven's Johnson Syndrome (SJS), Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS), and Toxic Epidermal Necrolysis (TEN). -

Cardiac Drug Therapy
Series Editor: Christopher P. Cannon ebook M. Gabriel Khan Cardiac Drug Therapy Contemporary Cardiology 8th Edition Disclaimer: This book is meant only for academic reference purpose only We acknowledge the Author and the Publisher www.pubrica.com +91-9884350006 [email protected] CONTEMPORARY CARDIOLOGY CHRISTOPHER P. C ANNON, MD SERIES EDITOR More information about this series at http://www.springer.com/series/7677 M. Gabriel Khan Cardiac Drug Therapy 8th Edition M. Gabriel Khan, MD, FRCPC, FRCP (London), FACC University of Ottawa The Ottawa Hospital Ottawa , ON , Canada ISSN 2196-8969 ISSN 2196-8977 (electronic) ISBN 978-1-61779-961-7 ISBN 978-1-61779-962-4 (eBook) DOI 10.1007/978-1-61779-962-4 Springer Totowa Heidelberg New York Dordrecht London Library of Congress Control Number: 2014952818 © Springer Science+Business Media New York 2015 This work is subject to copyright. All rights are reserved by the Publisher, whether the whole or part of the material is concerned, specifi cally the rights of translation, reprinting, reuse of illustrations, recitation, broadcasting, reproduction on microfi lms or in any other physical way, and transmission or information storage and retrieval, electronic adaptation, computer software, or by similar or dissimilar methodology now known or hereafter developed. Exempted from this legal reservation are brief excerpts in connection with reviews or scholarly analysis or material supplied specifi cally for the purpose of being entered and executed on a computer system, for exclusive use by the purchaser of the work. Duplication of this publication or parts thereof is permitted only under the provisions of the Copyright Law of the Publisher’s location, in its current version, and permission for use must always be obtained from Springer. -

Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix
United States International Trade Commission Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix USITC Publication 4208 December 2010 U.S. International Trade Commission COMMISSIONERS Deanna Tanner Okun, Chairman Irving A. Williamson, Vice Chairman Charlotte R. Lane Daniel R. Pearson Shara L. Aranoff Dean A. Pinkert Address all communications to Secretary to the Commission United States International Trade Commission Washington, DC 20436 U.S. International Trade Commission Washington, DC 20436 www.usitc.gov Modifications to the Harmonized Tariff Schedule of the United States to Implement Changes to the Pharmaceutical Appendix Publication 4208 December 2010 (This page is intentionally blank) Pursuant to the letter of request from the United States Trade Representative of December 15, 2010, set forth at the end of this publication, and pursuant to section 1207(a) of the Omnibus Trade and Competitiveness Act, the United States International Trade Commission is publishing the following modifications to the Harmonized Tariff Schedule of the United States (HTS) to implement changes to the Pharmaceutical Appendix, effective on January 1, 2011. Table 1 International Nonproprietary Name (INN) products proposed for addition to the Pharmaceutical Appendix to the Harmonized Tariff Schedule INN CAS Number Abagovomab 792921-10-9 Aclidinium Bromide 320345-99-1 Aderbasib 791828-58-5 Adipiplon 840486-93-3 Adoprazine 222551-17-9 Afimoxifene 68392-35-8 Aflibercept 862111-32-8 Agatolimod -

Comparison of the Anti-Inflammatory Effects of Cilomilast, Budesonide
Ratcliffe and Dougall BMC Pharmacology and Toxicology 2012, 13:15 http://www.biomedcentral.com/2050-6511/13/15 RESEARCH ARTICLE Open Access Comparison of the anti-inflammatory effects of Cilomilast, Budesonide and a p38 Mitogen activated protein kinase inhibitor in COPD lung tissue macrophages Marianne Jennifer Ratcliffe1* and Iain Gordon Dougall2 Abstract Chronic Obstructive Pulmonary Disease (COPD) is a disease characterized by a largely irreversible airflow obstruction and a persistent, excessive inflammatory response. Alveolar macrophages (AMs) are increased in the lungs of COPD patients, and act as orchestrators of the inflammatory response, releasing a range of mediators to coordinate recruitment and activation of leukocytes. Attempts to treat the inflammatory component of COPD with anti-inflammatory drugs such as steroids has met with limited success. In this study, we compared the ability of the phosphodiesterase IV (PDEIV) inhibitor Cilomilast, the steroid Budesonide, and the p38 mitogen activated protein kinase inhibitor BIRB-796 to inhibit tumour necrosis factor alpha (TNFα) and interleukin 6 (IL-6) releases from AMs isolated from COPD lung transplant tissue. All studies were carried out with appropriate ethical approval and written, informed consent was obtained from each subject. Cilomilast had little effect on cytokine release from AMs. There was considerable variability in the responsiveness of AMs to Budesonide, with a subset of AMs responding poorly to Budesonide. BIRB-796 inhibited TNFα release from all AM donors, including those that responded poorly to steroids. Treatment with BIRB-796 and Budesonide together gave an additive decrease in TNFa release. These results suggest that a p38 inhibitor may provide advantages over existing anti-inflammatory treatments for COPD, either as an add-on to existing therapy, or to treat patients who respond poorly to steroids. -

Telaprevir for HIV/Hepatitis C Virus–Coinfected Patients Failing
HIV/AIDS MAJOR ARTICLE Telaprevir for HIV/Hepatitis C Virus–Coinfected Patients Failing Treatment With Pegylated Interferon/Ribavirin (ANRS HC26 TelapreVIH): An Open-Label, Single-Arm, Phase 2 Trial Downloaded from https://academic.oup.com/cid/article/59/12/1768/2895305 by guest on 01 October 2021 Laurent Cotte,1 Joséphine Braun,2 Caroline Lascoux-Combe,3 Corine Vincent,2 Marc-Antoine Valantin,4 Philippe Sogni,5 Karine Lacombe,6 Didier Neau,7 Hugues Aumaitre,8 Dominique Batisse,9 Pierre de Truchis,10 Anne Gervais,11 Christian Michelet,12 Philippe Morlat,13 Daniel Vittecoq,14 Isabelle Rosa,15 Inga Bertucci,16 Stéphane Chevaliez,17 Jean-Pierre Aboulker,2 and Jean-Michel Molina3; for the French National Agency for Research on AIDS and Viral Hepatitis (ANRS) HC26 Study Groupa 1Hospices Civils de Lyon, Croix-Rousse Hospital, and INSERM U1052, 2INSERM SC10-US019, Villejuif, 3Assistance Publique–Hôpitaux de Paris (AP-HP), Saint-Louis Hospital, University of Paris VII Denis Diderot, and Sorbonne Paris-Cité, INSERM U941, 4AP-HP, Pitié-Salpêtrière Hospital, and UMR-S 943, INSERM, 5AP-HP, Cochin Hospital and Paris Descartes University, INSERM U-1016, 6AP-HP, Saint-Antoine Hospital, Sorbonne Universités, UPMC University Paris 06, UMR-S1136, 7Pellegrin University Hospital, Bordeaux, 8Saint-Jean Hospital, Perpignan, 9AP-HP, Georges Pompidou European Hospital, Paris, 10AP-HP, Raymond Poincaré Hospital, Garches, 11AP-HP, Bichat-Claude Bernard Hospital, Paris, 12Pontchaillou University Hospital, Rennes, 13Saint-André University Hospital, Bordeaux, 14AP-HP, Bicètre Hospital, Le Kremlin-Bicètre, 15Créteil Hospital, 16French National Agency for Research on AIDS and Viral Hepatitis, Paris, and 17AP-HP, Henri Mondor Hospital, Créteil, France (See the Editorial Commentary by Rockstroh on pages 1777–8.) Background. -

Use of Plackett-Burman Design for Rapid Screening of Diverse Raw Pectin Sources for Cold-Active Polygalacturonase and Amylase Production by Geotrichum Sp
Int.J.Curr.Microbiol.App.Sci (2015) 4(6): 821-827 ISSN: 2319-7706 Volume 4 Number 6 (2015) pp. 821-827 http://www.ijcmas.com Original Research Article Use of Plackett-Burman Design for rapid Screening of Diverse Raw Pectin Sources for Cold-Active Polygalacturonase and Amylase Production by Geotrichum sp K. Divya and P. Naga Padma* Bhavan s Vivekananda College of Science, Humanities and Commerce, Secunderabad 94, India *Corresponding author A B S T R A C T Cold active polygalacturonases and amylases play significant role in extraction and clarification of fruit juices at industrial level. An optimized production medium with low cost substrates would be very useful for commercial production of these enzymes. The present study was done to screen low cost pectin and starch K e y w o r d s substrates for cold active enzymes production. The different pectin and starch sources screened were fruit and vegetables peels like citrus, pineapple, apple, Amylase, banana, mango, guava, carrot, beetroot, bottle gourd, ridge gourd and potato. For Cold-active efficient screening of the best sources a statistical design like Plackett-Burman was enzyme, used as in this design n variables can be studied in just n-1 experiments only. A Geotrichum sps, twelve experimental design Plackett-Burman was used as the best sources can be Poly shortlisted in consideration with their interactive effects. The pectinolytic and galacturonase, amylolytic yeast isolate was identified as Geotrichum sps and was used for the Plackett- present study. Cold-active pectinase and amylase enzyme activity was assayed by Burman, dinitrosalicylic acid (DNS) method. -

Guanidine-Containing Polyhydroxyl Macrolides: Chemistry, Biology, and Structure-Activity Relationship
molecules Review Guanidine-Containing Polyhydroxyl Macrolides: Chemistry, Biology, and Structure-Activity Relationship Xiaoyuan Song 1, Ganjun Yuan 1,* , Peibo Li 2 and Sheng Cao 1 1 College of Bioscience and Bioengineering, Jiangxi Agricultural University, Nanchang 330045, China; [email protected] (X.S.); [email protected] (S.C.) 2 School of Life Sciences, Sun Yat-sen University, 135 Xingang Road, Guangzhou 510275, China; [email protected] * Correspondence: [email protected]; Tel.: +86-0791-83813459 Academic Editor: Jesus Simal-Gandara Received: 7 October 2019; Accepted: 29 October 2019; Published: 30 October 2019 Abstract: Antimicrobial resistance has been seriously threatening human health, and discovering new antimicrobial agents from the natural resource is still an important pathway among various strategies to prevent resistance. Guanidine-containing polyhydroxyl macrolides, containing a polyhydroxyl lactone ring and a guanidyl side chain, can be produced by many actinomycetes and have been proved to possess many bioactivities, especially broad-spectrum antibacterial and antifungal activities. To explore the potential of these compounds to be developed into new antimicrobial agents, a review on their structural diversities, spectroscopic characterizations, bioactivities, acute toxicities, antimicrobial mechanisms, and the structure-activity relationship was first performed based on the summaries and analyses of related publications from 1959 to 2019. A total of 63 guanidine-containing polyhydroxyl macrolides were reported, including -

Pectinase: a Useful Tool in Fruit Processing Industries
Mini Review Nutri Food Sci Int J - Volume 5 Issue 5 March 2018 Copyright © All rights are reserved by Jyoti Singh Jadaun DOI: 10.19080/NFSIJ.2018.05.555673 Pectinase: A Useful Tool in Fruit Processing Industries Heena Verma1, Lokesh K Narnoliya2 and Jyoti Singh Jadaun3* 1Department of Microbiology, Panjab university, India 2Department of Biotechnology (DBT), Center of Innovative and Applied Bioprocessing (CIAB), India 3Dyanand Girls Post Graduate College, India Submission: February 03, 2018; Published: March 07, 2018 *Corresponding author: Jyoti Singh Jadaun, Dyanand Girls Post Graduate College, 13/394, Parwati Bagla Rd, Kanpur, Uttar Pradesh 208001, India, Email: Abstract Owing to increase in the demand of fruit juices and fruit products, it became an indispensable need for the fruit processing industries to improve the quality of the fruit juices in a cost effective manner. Enzymes, being the highly efficient biocatalysts, are used at different steps in ofthe juice. process Visual of juiceacceptance production. of the Pectinases juice by the are consumers used for theneed clarification better clarity of the and juice improved by breaking colour thethat polysaccharide remain stable evenpectin during structure cold presentstorage inof the cellproduct. wall of plants into galacturonic acid monomers. Pectin structure breakage facilitates the filtration process and it increases the total yield Keywords: Abbreviations: Biocatalyst; PG: Polygalcturonase; Pectinase; Amylase; PE: Pectin Cellulase; Esterase; Pectin; PL: Starch;Pectin Lyase; Galacturonic -

Efficacy of Amiodarone in the Treatment of Ventricular
EFFICACY OF AMIODARONE IN THE TREATMENT OF VENTRICULAR ARRHYTHMIAS IN PATIENTS WITH CORONARY ARTERY DISEASE IN BANGLADESH 1 2 3 1 4 5 ADHIKARY DK , SAHA SK , MAHMOOD M , SALIM A , JOARDER AI , SINGHA CK , RAHMAN 6 7 8 9 10 11 MW , AHASAN MH , KHALED MFI , BANERJEE SK , MAHBUBA F , AHMED A Abstract Background: Ventricular arrhythmias (VA) are among the most feared complications of coronary artery disease (CAD) and one of the major contributors of death in CAD patients. Antiarrhythmic drug (AAD) therapy is required for recurrent significant VA in the absence of need for further revascularization. But all AADs do not have the same efficacy against life threatening VA and supraventricular arrhythmias (SVAs). Methodology: All (50) patients admitted in the department of Cardiology, BSMMU with ventricular arrhythmias with CAD fulfilling the inclusion and exclusion criteria were included in the study. Informed written consent was taken from each patient before enrollment. Detailed history was taken and relevant physical examinations were done. Loading dose followed by maintenance dose of amiodarone was given and recorded. Relevant lab investigations were performed and recorded in predesigned semi-structured data collection sheet. Symptomatic improvement was assessed, relevant physical examination was done and lab investigations were performed at 1, 3 and 6 month follow up. After editing data analysis was carried out by using the Statistical Package for Social Science (SPSS) version 23.0 windows software. Results: The mean age was found 57.7±8.0 years with a range of 45 to 78 years. Almost two third (62.0%) patients were male and 19(38.0%) patients were female. -

PHARMACEUTICAL APPENDIX to the TARIFF SCHEDULE 2 Table 1
Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE HARMONIZED TARIFF SCHEDULE Harmonized Tariff Schedule of the United States (2020) Revision 19 Annotated for Statistical Reporting Purposes PHARMACEUTICAL APPENDIX TO THE TARIFF SCHEDULE 2 Table 1. This table enumerates products described by International Non-proprietary Names INN which shall be entered free of duty under general note 13 to the tariff schedule. The Chemical Abstracts Service CAS registry numbers also set forth in this table are included to assist in the identification of the products concerned. For purposes of the tariff schedule, any references to a product enumerated in this table includes such product by whatever name known.