Chlorhexidine(BAN, Rinn)
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Antiseptics and Disinfectants for the Treatment Of
Verstraelen et al. BMC Infectious Diseases 2012, 12:148 http://www.biomedcentral.com/1471-2334/12/148 RESEARCH ARTICLE Open Access Antiseptics and disinfectants for the treatment of bacterial vaginosis: A systematic review Hans Verstraelen1*, Rita Verhelst2, Kristien Roelens1 and Marleen Temmerman1,2 Abstract Background: The study objective was to assess the available data on efficacy and tolerability of antiseptics and disinfectants in treating bacterial vaginosis (BV). Methods: A systematic search was conducted by consulting PubMed (1966-2010), CINAHL (1982-2010), IPA (1970- 2010), and the Cochrane CENTRAL databases. Clinical trials were searched for by the generic names of all antiseptics and disinfectants listed in the Anatomical Therapeutic Chemical (ATC) Classification System under the code D08A. Clinical trials were considered eligible if the efficacy of antiseptics and disinfectants in the treatment of BV was assessed in comparison to placebo or standard antibiotic treatment with metronidazole or clindamycin and if diagnosis of BV relied on standard criteria such as Amsel’s and Nugent’s criteria. Results: A total of 262 articles were found, of which 15 reports on clinical trials were assessed. Of these, four randomised controlled trials (RCTs) were withheld from analysis. Reasons for exclusion were primarily the lack of standard criteria to diagnose BV or to assess cure, and control treatment not involving placebo or standard antibiotic treatment. Risk of bias for the included studies was assessed with the Cochrane Collaboration’s tool for assessing risk of bias. Three studies showed non-inferiority of chlorhexidine and polyhexamethylene biguanide compared to metronidazole or clindamycin. One RCT found that a single vaginal douche with hydrogen peroxide was slightly, though significantly less effective than a single oral dose of metronidazole. -

Quaternary Ammonium Compounds
FACT SHEET: Quaternary Ammonium Compounds Quaternary ammonium compounds, also known as “quats” or “QACs,” include a number of chemicals used as sanitizers and disinfectants, including benzalkonium chloride, benzethonium chloride, cetalkonium chloride, cetrimide, cetrimonium bromide, cetylpyridinium chloride, glycidyl trimethyl, ammonium chloride, and stearalkonium chloride.[i] In general, quats cause toxic effects through all Mutagenicity routes of exposure including inhalation, Some quats have shown to be mutagenic and to ingestion, dermal application, and irrigation of damage animal DNA and DNA in human body cavities. Exposure to diluted solutions may lymphocytes at much lower levels than are result in mild irritation, while concentrated present in cleaning chemicals.[6] solutions are corrosive, causing burns to the skin and mucous Membranes. They can produce Antimicrobial Resistance systemic toxicity and can also cause allergic Genes have been discovered that mediate reactions.[2] resistance to quats. There has been an association of some of these genes with beta lactamase genes, Asthma and Allergies raising concern for a relationship between Of particular interest with regard to use as disinfectant resistance and antibiotic resistance.[7] disinfectants in the COVID-19 pandemic, quats increase the risk for asthma and allergic Reproductive Toxicity sensitization. Evidence from occupational Mice whose cages were cleaned with QACs had exposures shows increased risk of rhinitis and very low fertility rates. [8] Exposure to a common asthma -

ANNEX VI List of Preservatives Allowed for Use in Cosmetic Products
ANNEX VI List of preservatives allowed for use in cosmetic products ANNEX VI LIST OF PRESERVATIVES WHICH COSMETIC PRODUCTS MAY CONTAIN Preamble 1. Preservatives are substances which may be added to cosmetic products for the primary purpose of inhibiting the development of micro-organisms in such products. 2. The substances marked with the symbol (+) may also be added to cosmetic products in concentration other than those laid down in this ANNEX for other purposes apparent from the presentation of the products, e.g. as deodorants in soaps or as anti-dandruff agents in shampoos. 3. Other substances used in the formulation of cosmetic products may also have anti-microbial properties and thus help in the preservation of the products, as, for instance, many essential oils and some alcohols. These substances are not included in the ANNEX. 4. For the purposes of this list - “Salts” is taken to mean: salts of the cations sodium, potassium, calcium, magnesium, ammonium, and ethanolamines; salts of the anions chloride, bromide, sulphate, acetate. - “Esters” is taken to mean: esters of methyl, ethyl, propyl, isopropyl, butyl, isobutyl, phenyl. 5. All finished products containing formaldehyde or substances in this ANNEX and which release formaldehyde must be labelled with the warning “contains formaldehyde” where the concentration of formaldehyde in the finished product exceeds 0.05%. Revised as per August 2015 ASEAN Cosmetic Documents 1 Annex VI – Part 1 – List of preservatives allowed for use in cosmetic products ANNEX VI – PART 1 LIST OF PRESERVATIVES ALLOWED Reference Substance Maximum authorized Limitations and Conditions of use and Number concentration requirements warnings which must be printed on the label a b c d e 1 Benzoic acid (CAS No. -

Quaternary Ammonium Compounds
FT-619502 Quaternary ammonium compounds Products Description Name : BenzyldimethylhexadecylAmmoniu m Chloride Catalog Number : 619502, 100g 16-BAC ; Cetalkonium chloride; N-hexadecyl-N,N-dimethyl Benzenemethaminium, Chloride; cloruro de Structure : CAS: [122-18-9]; cetalconio; Chlorure de cetalkonium Benzyldimethylhexadecylammonium chloride; Benzyl n-hexadecyl Molecular Weight : MW= 396.1 dimethylammonium chloride; Cetol; Benzyldimethylhexadecylammonium chloride, hydrate; N-Hexadecyl- N,N-dimethylbenzenemethanaminium chloride; Alkyl C-16 Properties: Freely soluble benzyldimethyl ammonium chloride; pH : 6.0 - 8.5 (10% sol.) Corrosive Avoid shock and frictions S26-45-60-61 Storage: Room temperature (Z) Technical and Scientific Information General description and applications Quaternary ammonium compounds are any of a group of ammonium salts in which organic radicals have been substituted for all four hydrogens of the original ammonium cation. They has a central nitrogen atom which is joined to four organic radicals and one acid radical. The organic radicals may be alkyl, aryl, or aralkyl, and the nitrogen can be part of a ring system. They are prepared by treatment of an amine with an alkylating agent. They show a variety of physical, chemical, and biological properties and most compounds are soluble in water and strong electrolytes. Such compounds include: Benzalkonium Chloride ( CAS RN: 8001-54-5) Benzethonium Chloride CAS 121-54-0 Cetalkonium Chloride( CAS 122-18-9) Cetrimide ( CAS 8044-71-1) Cetrimonium Bromide ( CAS 57-09-0) Cetylpyridinium -

Of 8 August 2014 Amending Annexes III, V and VI to Regulation (EC) No 1223/2009 of the European Parliament and the Council on Cosmetic Products
9.8.2014 EN Official Journal of the European Union L 238/3 COMMISSION REGULATION (EU) No 866/2014 of 8 August 2014 amending Annexes III, V and VI to Regulation (EC) No 1223/2009 of the European Parliament and the Council on cosmetic products (Text with EEA relevance) THE EUROPEAN COMMISSION, Having regard to the Treaty on the Functioning of the European Union, Having regard to Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products (1), and in particular Article 31(2) thereof, Whereas: (1) The substances identified by the denominations alkyl (C12-22) trimethyl ammonium bromide and chloride are regu lated as preservatives under entry 44 of Annex V to Regulation (EC) No 1223/2009 with a maximum concentra tion of 0,1 %. (2) The Scientific Committee on Consumer Products (‘SCCP’), subsequently replaced by the Scientific Committee on Consumer Safety (‘SCCS’) pursuant to Commission Decision 2008/721/EC (2), evaluated the safety of alkyl (C16, C18, C22) trimethylammonium chloride (cetrimonium chloride, steartrimonium chloride and behentrimonium chloride) for other uses than as preservatives in 2005, 2007 and 2009. (3) The SCCS concluded in its opinion of 8 December 2009 (3) that, apart from the fact that quaternary ammonium derivative formulations have the potential to be skin irritants, especially when combinations of the concerned compounds are used, the use of cetrimonium chloride, steartrimonium chloride and behentrimonium chloride does not pose a risk to the health of the consumer in concentrations below certain limits, which are explicitly set out in the SCCS opinion. -
![Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set](https://docslib.b-cdn.net/cover/8870/ehealth-dsi-ehdsi-v2-2-2-or-ehealth-dsi-master-value-set-1028870.webp)
Ehealth DSI [Ehdsi V2.2.2-OR] Ehealth DSI – Master Value Set
MTC eHealth DSI [eHDSI v2.2.2-OR] eHealth DSI – Master Value Set Catalogue Responsible : eHDSI Solution Provider PublishDate : Wed Nov 08 16:16:10 CET 2017 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 1 of 490 MTC Table of Contents epSOSActiveIngredient 4 epSOSAdministrativeGender 148 epSOSAdverseEventType 149 epSOSAllergenNoDrugs 150 epSOSBloodGroup 155 epSOSBloodPressure 156 epSOSCodeNoMedication 157 epSOSCodeProb 158 epSOSConfidentiality 159 epSOSCountry 160 epSOSDisplayLabel 167 epSOSDocumentCode 170 epSOSDoseForm 171 epSOSHealthcareProfessionalRoles 184 epSOSIllnessesandDisorders 186 epSOSLanguage 448 epSOSMedicalDevices 458 epSOSNullFavor 461 epSOSPackage 462 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 2 of 490 MTC epSOSPersonalRelationship 464 epSOSPregnancyInformation 466 epSOSProcedures 467 epSOSReactionAllergy 470 epSOSResolutionOutcome 472 epSOSRoleClass 473 epSOSRouteofAdministration 474 epSOSSections 477 epSOSSeverity 478 epSOSSocialHistory 479 epSOSStatusCode 480 epSOSSubstitutionCode 481 epSOSTelecomAddress 482 epSOSTimingEvent 483 epSOSUnits 484 epSOSUnknownInformation 487 epSOSVaccine 488 © eHealth DSI eHDSI Solution Provider v2.2.2-OR Wed Nov 08 16:16:10 CET 2017 Page 3 of 490 MTC epSOSActiveIngredient epSOSActiveIngredient Value Set ID 1.3.6.1.4.1.12559.11.10.1.3.1.42.24 TRANSLATIONS Code System ID Code System Version Concept Code Description (FSN) 2.16.840.1.113883.6.73 2017-01 A ALIMENTARY TRACT AND METABOLISM 2.16.840.1.113883.6.73 2017-01 -

List Item Cetrimide: Summary Report
The European Agency for the Evaluation of Medicinal Products Veterinary Medicines Evaluation Unit EMEA/MRL/073/96-FINAL March 1996 COMMITTEE FOR VETERINARY MEDICINAL PRODUCTS CETRIMIDE SUMMARY REPORT 1. Cetrimide (CAS 8044-71-1) is a quaternary ammonium antiseptic and surfactant. It consists mostly of trimethyltetradecylammonium bromide (CAS 1119-97-7; also known as tetradonium bromide) with smaller amounts of dodecyltrimethylammonium bromide (CAS 1119-94-4; also known as DTAB) and hexadecyltrimethylammonium bromide (CAS 57-09-0). The European Pharmacopoeia requires that cetrimide should contain at least 96% but no more than 101.0% alkyltrimethylammonium bromides. 2. Historically, the name cetrimide was used for a material consisting of predominantly hexadecyltrimethylammonium bromide, (CAS 57-09-0; also known as cetrimonium bromide, CTAB, and cetyltrimethylammonium bromide), together with smaller amounts of analogous alkyltrimethylammonium bromides. Some of the safety studies were carried out using this "cetrimide" rather than the cetrimide currently specified in the Europeam Pharmacopoeia. 3. In veterinary medicine, cetrimide is used as a topical antiseptic at concentrations of up to 2%. It is also used as an excipient in an injectable antibiotic formulation intended for use in cattle, sheep and pigs. When used as excipient, the concentration of cetrimide in the formulation is around 0.25 mg/ml, resulting in a dose of approximately 0.01 mg/kg bw of cetrimide in the target species. 4. In aqueous solution, cetrimide dissociates to a biologically-active cation and an inactive anion. Cetrimide is inactive towards bacterial spores, it is effective against some viruses and has variable anti-fungal activity. The cation is also responsible for the surfactant activity. -

Federal Communications Commission
FEDERAL REGISTER VOLUME 34 • NUM BER 189 Thursday, October 2,1969 • Washington, D.C. Pages 15331-15405 Agencies in this issue— The President Agency for International Development Atomic Energy Commission Civil Aeronautics Board Consumer and Marketing Service Customs Bureau Emergency Preparedness Office Federal Aviation Administration Federal Communications Commission Federal Home Loälh Bank Board Federal Maritime Commission Federal Power Commission Federal Trade Commission Fish and Wildlife Service Food and Drug Administration General Services Administration Indian Aifairs Bureau Internal Revenue Service Interstate Commerce Commission Land Management Bureau Public Health Service Securities and Exchange Commission _ _ Small Business Administration Treasury Department Detailed list of Contents appears inside. MICROFILM EDITION FEDERAL REGISTER 35mm MICROFILM Complete Set 19 3 6 -6 7,16 7 Rolls $1,162 \ Vol. Year Price Vol. Year Price Vol. Year Price 1 1936 $8 12 1947 $26 23 1958 $36 2 1937 10 13 1948 27 24 1959 40 3 1938 9 14 1949 22 25 1960 49 4 1939 14 15 1950 26 26 1961 46 5 1940 15 16 1951 43 27 1962 50 6 1941 20 17 1952 35 28 1963 49 7 1942 35 18 1953 32 29 1964 57 8 1943 52 19 1954 39 30 1965 58 9 1944 42 20 1955 36 31 1966 61 10 1945 43 21 1956 38 32 1967 64 11 1946 42 22 1957 38 Order Microfilm Edition from Publications Sales Branch National Archives and Records Service Washington, D.C. 20408 rr<nr<n ■ w n m C T m Published daily, Tuesday through Saturday (no publication on Sundays, Mondays» rrllrll/ll jMllFd1 K r III NI I* K on the day aiter an official Federal holiday), by the Office of the Federal Register, Na ■ ArchivesA«.Msa> nnrtand RaanrHsRecords Service.Service, General Services Administration.Administration, Washington, D.C. -

Vr Meds Ex01 3B 0825S Coding Manual Supplement Page 1
vr_meds_ex01_3b_0825s Coding Manual Supplement MEDNAME OTHER_CODE ATC_CODE SYSTEM THER_GP PHRM_GP CHEM_GP SODIUM FLUORIDE A12CD01 A01AA01 A A01 A01A A01AA SODIUM MONOFLUOROPHOSPHATE A12CD02 A01AA02 A A01 A01A A01AA HYDROGEN PEROXIDE D08AX01 A01AB02 A A01 A01A A01AB HYDROGEN PEROXIDE S02AA06 A01AB02 A A01 A01A A01AB CHLORHEXIDINE B05CA02 A01AB03 A A01 A01A A01AB CHLORHEXIDINE D08AC02 A01AB03 A A01 A01A A01AB CHLORHEXIDINE D09AA12 A01AB03 A A01 A01A A01AB CHLORHEXIDINE R02AA05 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S01AX09 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S02AA09 A01AB03 A A01 A01A A01AB CHLORHEXIDINE S03AA04 A01AB03 A A01 A01A A01AB AMPHOTERICIN B A07AA07 A01AB04 A A01 A01A A01AB AMPHOTERICIN B G01AA03 A01AB04 A A01 A01A A01AB AMPHOTERICIN B J02AA01 A01AB04 A A01 A01A A01AB POLYNOXYLIN D01AE05 A01AB05 A A01 A01A A01AB OXYQUINOLINE D08AH03 A01AB07 A A01 A01A A01AB OXYQUINOLINE G01AC30 A01AB07 A A01 A01A A01AB OXYQUINOLINE R02AA14 A01AB07 A A01 A01A A01AB NEOMYCIN A07AA01 A01AB08 A A01 A01A A01AB NEOMYCIN B05CA09 A01AB08 A A01 A01A A01AB NEOMYCIN D06AX04 A01AB08 A A01 A01A A01AB NEOMYCIN J01GB05 A01AB08 A A01 A01A A01AB NEOMYCIN R02AB01 A01AB08 A A01 A01A A01AB NEOMYCIN S01AA03 A01AB08 A A01 A01A A01AB NEOMYCIN S02AA07 A01AB08 A A01 A01A A01AB NEOMYCIN S03AA01 A01AB08 A A01 A01A A01AB MICONAZOLE A07AC01 A01AB09 A A01 A01A A01AB MICONAZOLE D01AC02 A01AB09 A A01 A01A A01AB MICONAZOLE G01AF04 A01AB09 A A01 A01A A01AB MICONAZOLE J02AB01 A01AB09 A A01 A01A A01AB MICONAZOLE S02AA13 A01AB09 A A01 A01A A01AB NATAMYCIN A07AA03 A01AB10 A A01 -

Patch Test Products and Reference Manual 2015
PATCH TEST PRODUCTS & REFERENCE MANUAL MANUAL REFERENCE & PRODUCTS TEST PATCH 4 World Leader in Patch Testing 2015 CHEMOTECHNIQUE DIAGNOSTICS CHEMOTECHNIQUE PATCH TEST PRODUCTS & REFERENCE MANUAL 2015 MODEMGATAN 9 | SE-235 39 VELLINGE |SWEDEN PHONE +46 40 466 077 | FAX +46 40 466 700 WWW.CHEMOTECHNIQUE.SE [email protected] | [email protected] ...for the diagnosis of contact allergy 2015 The complete range of products for Patch Testing A1 Foreword by Bo Niklasson, CEO First of all I would like to thank all our faithful customers for your support during the past year and also welcome our new distributors that have been appointed during the year. Chemotechnique Diagnostic’s 34 years of continuous growth and development has been the result of our belief in building strong and long term business relationships with our global network of distributors, combined with the ongoing support and contributions of our product-user base of physicians. Our commitment is to continue serving dermatology in future years... maintaining our leadership position. During the past year we have invested in new state of the art equipment in both the analytical and production sector and developed a closed loop system in the production of haptens. When looking at our website you will note that we have made it possible also for patients to freely access our database of extensive information regarding the haptens and being able to print information sheets. We have also changed the site to be adapted to mobile platforms such as mobile phones, iPads and similar devices. There will as always be additions and amendments in our range and these are found at the end of the Patch Test Products & Reference Manual. -
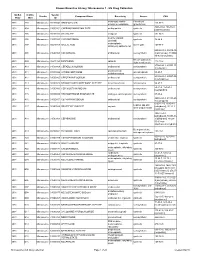
Known Bioactive Library: Microsource 1 - US Drug Collection
Known Bioactive Library: Microsource 1 - US Drug Collection ICCB-L ICCB-L Vendor Vendor Compound Name Bioactivity Source CAS Plate Well ID antifungal, inhibits Penicillium 2091 A03 Microsource 00200046 GRISEOFULVIN 126-07-8 mitosis in metaphase griseofulvum 3505-38-2, 486-16-8 2091 A04 Microsource 01500161 CARBINOXAMINE MALEATE antihistaminic synthetic [carbinoxamine] 2091 A05 Microsource 00200331 SALSALATE analgesic synthetic 552-94-3 muscle relaxant 2091 A06 Microsource 01500162 CARISOPRODOL synthetic 78-44-4 (skeletal) antineoplastic, 2091 A07 Microsource 00210369 GALLIC ACID insect galls 149-91-7 astringent, antibacterial 66592-87-8, 50370-12- 2091 A08 Microsource 01500163 CEFADROXIL antibacterial semisynthetic 2 [anhydrous], 119922- 89-9 [hemihydrate] Rheum palmatum, 2091 A09 Microsource 00211468 DANTHRON cathartic 117-10-2 Xyris semifuscata 27164-46-1, 25953-19- 2091 A10 Microsource 01500164 CEFAZOLIN SODIUM antibacterial semisynthetic 9 [cefazolin] glucocorticoid, 2091 A11 Microsource 00300024 HYDROCORTISONE adrenal glands 50-23-7 antiinflammatory 64485-93-4, 63527-52- 2091 A12 Microsource 01500165 CEFOTAXIME SODIUM antibacterial semisynthetic 6 [cefotaxime] 2091 A13 Microsource 00300029 DESOXYCORTICOSTERONE ACETATE mineralocorticoid adrenocortex 56-47-3 58-71-9, 153-61-7 2091 A14 Microsource 01500166 CEPHALOTHIN SODIUM antibacterial semisynthetic [cephalothin] 2091 A15 Microsource 00300034 TESTOSTERONE PROPIONATE androgen, antineoplastic semisynthetic 57-85-2 24356-60-3, 21593-23- 2091 A16 Microsource 01500167 CEPHAPIRIN SODIUM -

Improved Pentamethine Cyanine Nanosensors for Optoacoustic Imaging of Pancreatic Cancer Matthew D
www.nature.com/scientificreports OPEN Improved pentamethine cyanine nanosensors for optoacoustic imaging of pancreatic cancer Matthew D. Laramie1,9, Benjamin L. Fouts2,3,9, William M. MacCuaig3,4, Emmanuel Buabeng1,5, Meredith A. Jones4, Priyabrata Mukherjee6, Bahareh Behkam7, Lacey R. McNally2,3,4,8* & Maged Henary1,5* Optoacoustic imaging is a new biomedical imaging technology with clear benefts over traditional optical imaging and ultrasound. While the imaging technology has improved since its initial development, the creation of dedicated contrast agents for optoacoustic imaging has been stagnant. Current exploration of contrast agents has been limited to standard commercial dyes that have already been established in optical imaging applications. While some of these compounds have demonstrated utility in optoacoustic imaging, they are far from optimal and there is a need for contrast agents with tailored optoacoustic properties. The synthesis, encapsulation within tumor targeting silica nanoparticles and applications in in vivo tumor imaging of optoacoustic contrast agents are reported. Diagnosis and treatment of pancreatic cancer remains daunting. Pancreatic tumors generally respond poorly to chemotherapy, and resection is the only truly curative option1–7. However, because pancreatic malignancies typically occur without symptoms, diagnosis is not usually made until the cancer has spread to other organs, at which point complete surgical resection is impossible 3,8,9. Due to the combined difculties of diagnosis and treatment, the prognosis for pancreatic cancer remains grim; 80–85% of patients will be untreatable at the time of diagnosis, and the overall 5-year survival rate is 4%10. Numerous approaches to improve the detection of pancreatic lesions have been attempted, but little success has been achieved.