Memory Part 2: the Role of the Medial Temporal Lobe
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Lesions of Perirhinal and Parahippocampal Cortex That Spare the Amygdala and Hippocampal Formation Produce Severe Memory Impairment
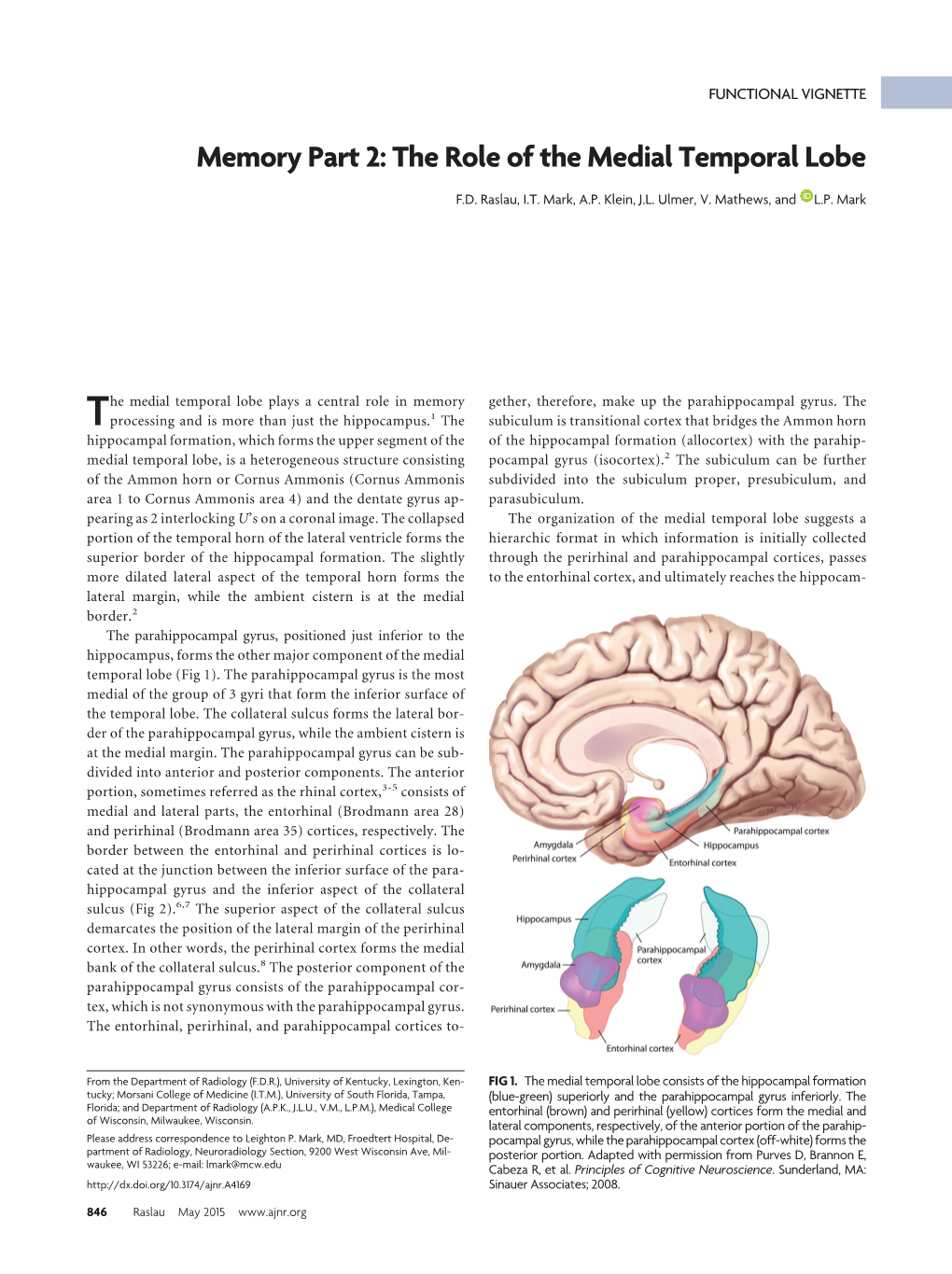
The Journal of Neuroscience, December 1989, 9(12): 4355-4370 Lesions of Perirhinal and Parahippocampal Cortex That Spare the Amygdala and Hippocampal Formation Produce Severe Memory Impairment Stuart Zola-Morgan,’ Larry Ft. Squire,’ David G. Amaral,2 and Wendy A. Suzuki2J Veterans Administration Medical Center, San Diego, California, 92161, and Department of Psychiatry, University of California, San Diego, La Jolla, California 92093, The Salk Institute, San Diego, California 92136, and 3Group in Neurosciences, University of California, San Diego, La Jolla, California 92093 In monkeys, bilateral damage to the medial temporal region Moss, 1984). (In this notation, H refers to the hippocampus, A produces severe memory impairment. This lesion, which in- to the amygdala, and the plus superscript (+) to the cortical cludes the hippocampal formation, amygdala, and adjacent tissue adjacent to each structure.) This lesion appears to con- cortex, including the parahippocampal gyrus (the H+A+ le- stitute an animal model of medial temporal lobe amnesia like sion), appears to constitute an animal model of human me- that exhibited by the well-studied patient H.M. (Scoville and dial temporal lobe amnesia. Reexamination of histological Milner, 1957). material from previously studied monkeys with H+A+ lesions The H+A+ lesion produces greater memory impairment than indicated that the perirhinal cortex had also sustained sig- a lesion limited to the hippocampal formation and parahip- nificant damage. Furthermore, recent neuroanatomical stud- pocampal cortex-the H+ lesion (Mishkin, 1978; Mahut et al., ies show that the perirhinal cortex and the closely associated 1982; Zola-Morgan and Squire, 1985, 1986; Zola-Morgan et al., parahippocampal cortex provide the major source of cortical 1989a). -

Neural Mechanisms of Navigation Involving Interactions of Cortical and Subcortical Structures
J Neurophysiol 119: 2007–2029, 2018. First published February 14, 2018; doi:10.1152/jn.00498.2017. REVIEW Where Are You Going? The Neurobiology of Navigation Neural mechanisms of navigation involving interactions of cortical and subcortical structures James R. Hinman, Holger Dannenberg, Andrew S. Alexander, and Michael E. Hasselmo Center for Systems Neuroscience, Boston University, Boston, Massachusetts Submitted 5 July 2017; accepted in final form 1 February 2018 Hinman JR, Dannenberg H, Alexander AS, Hasselmo ME. Neural mecha- nisms of navigation involving interactions of cortical and subcortical structures. J Neurophysiol 119: 2007–2029, 2018. First published February 14, 2018; doi: 10.1152/jn.00498.2017.—Animals must perform spatial navigation for a range of different behaviors, including selection of trajectories toward goal locations and foraging for food sources. To serve this function, a number of different brain regions play a role in coding different dimensions of sensory input important for spatial behavior, including the entorhinal cortex, the retrosplenial cortex, the hippocampus, and the medial septum. This article will review data concerning the coding of the spatial aspects of animal behavior, including location of the animal within an environment, the speed of movement, the trajectory of movement, the direction of the head in the environment, and the position of barriers and objects both relative to the animal’s head direction (egocentric) and relative to the layout of the environment (allocentric). The mechanisms for coding these important spatial representations are not yet fully understood but could involve mechanisms including integration of self-motion information or coding of location based on the angle of sensory features in the environment. -

Sustained Activities and Retrieval in a Computational Model of Perirhinal
Sustained activities and retrieval in a computational model of perirhinal cortex Julien Vitay and Fred H. Hamker∗ Abstract Perirhinal cortex is involved in object recognition and novelty detection, but also in multimodal in- tegration, reward association and visual working memory. We propose a computational model that focuses on the role of perirhinal cortex in working memory, particularly with respect to sustained activities and memory retrieval. This model describes how different partial informations are inte- grated into assemblies of neurons that represent the identity of an object. Through dopaminergic modulation, the resulting clusters can retrieve the global information with recurrent interactions between neurons. Dopamine leads to sustained activities after stimulus disappearance that form the basis of the involvement of perirhinal cortex in visual working memory processes. The infor- mation carried by a cluster can also be retrieved by a partial thalamic or prefrontal stimulation. Thus, we suggest that areas involved in planning and memory coordination encode a pointer to access the detailed information encoded in associative cortex such as perirhinal cortex. ∗Allgemeine Psychologie, Psychologisches Institut II, Westf. Wilhelms-Universit¨at M¨unster, Germany 1 Introduction Perirhinal cortex (PRh), composed of cortical areas 35 and 36, is located in the ventromedial part of the temporal lobe. It receives its major inputs from areas TE and TEO of inferotemporal cortex, as well as from entorhinal cortex (ERh), parahippocampal cortex, insular cortex and orbitofrontal cortex (Suzuki & Amaral, 1994). As part of the medial temporal lobe system (with hippocampus and ERh), its primary role is considered to be object-recognition memory, as shown by impairements in delayed matching-to-sample (DMS) or delayed nonmatching-to-sample (DNMS) tasks following PRh cooling or removal (Horel, Pytko-Joiner, Voytko, & Salsbury, 1987; Zola-Morgan, Squire, Amaral, & Suzuki, 1989; Meunier, Bachevalier, Mishkin, & Murray, 1993; Buffalo, Reber, & Squire, 1998). -

Neuromodulators and Long-Term Synaptic Plasticity in Learning and Memory: a Steered-Glutamatergic Perspective
brain sciences Review Neuromodulators and Long-Term Synaptic Plasticity in Learning and Memory: A Steered-Glutamatergic Perspective Amjad H. Bazzari * and H. Rheinallt Parri School of Life and Health Sciences, Aston University, Birmingham B4 7ET, UK; [email protected] * Correspondence: [email protected]; Tel.: +44-(0)1212044186 Received: 7 October 2019; Accepted: 29 October 2019; Published: 31 October 2019 Abstract: The molecular pathways underlying the induction and maintenance of long-term synaptic plasticity have been extensively investigated revealing various mechanisms by which neurons control their synaptic strength. The dynamic nature of neuronal connections combined with plasticity-mediated long-lasting structural and functional alterations provide valuable insights into neuronal encoding processes as molecular substrates of not only learning and memory but potentially other sensory, motor and behavioural functions that reflect previous experience. However, one key element receiving little attention in the study of synaptic plasticity is the role of neuromodulators, which are known to orchestrate neuronal activity on brain-wide, network and synaptic scales. We aim to review current evidence on the mechanisms by which certain modulators, namely dopamine, acetylcholine, noradrenaline and serotonin, control synaptic plasticity induction through corresponding metabotropic receptors in a pathway-specific manner. Lastly, we propose that neuromodulators control plasticity outcomes through steering glutamatergic transmission, thereby gating its induction and maintenance. Keywords: neuromodulators; synaptic plasticity; learning; memory; LTP; LTD; GPCR; astrocytes 1. Introduction A huge emphasis has been put into discovering the molecular pathways that govern synaptic plasticity induction since it was first discovered [1], which markedly improved our understanding of the functional aspects of plasticity while introducing a surprisingly tremendous complexity due to numerous mechanisms involved despite sharing common “glutamatergic” mediators [2]. -

Anatomy of the Temporal Lobe
Hindawi Publishing Corporation Epilepsy Research and Treatment Volume 2012, Article ID 176157, 12 pages doi:10.1155/2012/176157 Review Article AnatomyoftheTemporalLobe J. A. Kiernan Department of Anatomy and Cell Biology, The University of Western Ontario, London, ON, Canada N6A 5C1 Correspondence should be addressed to J. A. Kiernan, [email protected] Received 6 October 2011; Accepted 3 December 2011 Academic Editor: Seyed M. Mirsattari Copyright © 2012 J. A. Kiernan. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Only primates have temporal lobes, which are largest in man, accommodating 17% of the cerebral cortex and including areas with auditory, olfactory, vestibular, visual and linguistic functions. The hippocampal formation, on the medial side of the lobe, includes the parahippocampal gyrus, subiculum, hippocampus, dentate gyrus, and associated white matter, notably the fimbria, whose fibres continue into the fornix. The hippocampus is an inrolled gyrus that bulges into the temporal horn of the lateral ventricle. Association fibres connect all parts of the cerebral cortex with the parahippocampal gyrus and subiculum, which in turn project to the dentate gyrus. The largest efferent projection of the subiculum and hippocampus is through the fornix to the hypothalamus. The choroid fissure, alongside the fimbria, separates the temporal lobe from the optic tract, hypothalamus and midbrain. The amygdala comprises several nuclei on the medial aspect of the temporal lobe, mostly anterior the hippocampus and indenting the tip of the temporal horn. The amygdala receives input from the olfactory bulb and from association cortex for other modalities of sensation. -

The Neurobiology of Agrammatic Sentence Comprehension: a Lesion Study
The Neurobiology of Agrammatic Sentence Comprehension: A Lesion Study Corianne Rogalsky1, Arianna N. LaCroix1, Kuan-Hua Chen2,3, Steven W. Anderson2, Hanna Damasio4, Tracy Love5, and Gregory Hickok6 Abstract ■ Broca’s area has long been implicated in sentence compre- average did not exhibit the expected agrammatic compre- hension. Damage to this region is thought to be the central hension pattern—for example, their performance was >80% source of “agrammatic comprehension” in which performance on noncanonical sentences in the sentence–picture matching is substantially worse (and near chance) on sentences with non- task. Patients with ATL damage (n = 18) also did not exhibit canonical word orders compared with canonical word order an agrammatic comprehension pattern. Across our entire sentences (in English). This claim is supported by functional patient sample, the lesions of patients with agrammatic com- neuroimaging studies demonstrating greater activation in prehension patterns in either task had maximal overlap in pos- Broca’s area for noncanonical versus canonical sentences. terior superior temporal and inferior parietal regions. Using However, functional neuroimaging studies also have frequently voxel-based lesion–symptom mapping, we find that lower per- implicated the anterior temporal lobe (ATL) in sentence pro- formances on canonical and noncanonical sentences in each cessing more broadly, and recent lesion–symptom mapping task are both associated with damage to a large left superior studies have implicated the ATL and mid temporal regions in temporal–inferior parietal network including portions of the agrammatic comprehension. This study investigates these ATL, but not Broca’s area. Notably, however, response bias in seemingly conflicting findings in 66 left-hemisphere patients plausibility judgments was significantly associated with damage with chronic focal cerebral damage. -

Connectivity of BA46 Involvement in the Executive Control of Language
Alfredo Ardila, Byron Bernal and Monica Rosselli Psicothema 2016, Vol. 28, No. 1, 26-31 ISSN 0214 - 9915 CODEN PSOTEG Copyright © 2016 Psicothema doi: 10.7334/psicothema2015.174 www.psicothema.com Connectivity of BA46 involvement in the executive control of language Alfredo Ardila1, Byron Bernal2 and Monica Rosselli3 1 Florida International University, 2 Miami Children’s Hospital and 3 Florida Atlantic University Abstract Resumen Background: Understanding the functions of different brain areas has Estudio de la conectividad del AB46 en el control ejecutivo del lenguaje. represented a major endeavor of contemporary neurosciences. Modern Antecedentes: la comprensión de las funciones de diferentes áreas neuroimaging developments suggest cognitive functions are associated cerebrales representa una de las mayores empresas de las neurociencias with networks rather than with specifi c areas. Objectives. The purpose contemporáneas. Los estudios contemporáneos con neuroimágenes of this paper was to analyze the connectivity of Brodmann area (BA) 46 sugieren que las funciones cognitivas se asocian con redes más que con (anterior middle frontal gyrus) in relation to language. Methods: A meta- áreas específi cas. El propósito de este estudio fue analizar la conectividad analysis was conducted to assess the language network in which BA46 is del área de Brodmann 46 (BA46) (circunvolución frontal media anterior) involved. The DataBase of Brainmap was used; 19 papers corresponding con relación al lenguaje. Método: se llevó a cabo un meta-análisis para to 60 experimental conditions with a total of 245 subjects were included. determinar el circuito o red lingüística en la cual participa BA46. Se utilizó Results: Our results suggest the core network of BA46. -

Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans Ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai
Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai To cite this version: Hans ten Donkelaar, Nathalie Tzourio-Mazoyer, Jürgen Mai. Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex. Frontiers in Neuroanatomy, Frontiers, 2018, 12, pp.93. 10.3389/fnana.2018.00093. hal-01929541 HAL Id: hal-01929541 https://hal.archives-ouvertes.fr/hal-01929541 Submitted on 21 Nov 2018 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. REVIEW published: 19 November 2018 doi: 10.3389/fnana.2018.00093 Toward a Common Terminology for the Gyri and Sulci of the Human Cerebral Cortex Hans J. ten Donkelaar 1*†, Nathalie Tzourio-Mazoyer 2† and Jürgen K. Mai 3† 1 Department of Neurology, Donders Center for Medical Neuroscience, Radboud University Medical Center, Nijmegen, Netherlands, 2 IMN Institut des Maladies Neurodégénératives UMR 5293, Université de Bordeaux, Bordeaux, France, 3 Institute for Anatomy, Heinrich Heine University, Düsseldorf, Germany The gyri and sulci of the human brain were defined by pioneers such as Louis-Pierre Gratiolet and Alexander Ecker, and extensified by, among others, Dejerine (1895) and von Economo and Koskinas (1925). -

Metabolic Reduction in the Posterior Cingulate Cortex in Very Early Alzheimer’S Disease
Metabolic Reduction in the Posterior Cingulate Cortex in Very Early Alzheimer’s Disease Satoshi Minoshima, MD, PhD,* Bruno Giordani, PhD,t Stanley Berent, PhD,t$ Kirk A. Frey, MD, PhD,*$ Norman L. Foster, MD,$ and David E. Kuhl, MD* This study investigated cerebral glucose metabolism in very early Alzheimer’s disease, before a clinical diagnosis of probable Alzheimer’s disease is possible, using [ ‘8F]flu~r~de~xygluc~~epositron emission tomography. First, 66 patients with probable Alzheimer’s disease with a spectrum of dementia severity (Mini-Mental State Examination score, 0-23) were recruited and studied. Cortical metabolic activity was analyzed topographically using three-dimensional stereotactic surface projections. Regression analysis was performed for each brain pixel to predict metabolic patterns of very early disease. Predictions were tested prospectively in a group of 8 patients who complained only of memory impairment without general cognitive decline (Mini-Mental State Examination score, 25 Ifr 1) at the time of scanning but whose condition later progressed to probable Alzheimer’s disease. Both results were compared to cerebral metabolic activity in 22 age-similar normal control subjects. Prediction and analysis of actual patients consistently indicated marked metabolic reduction (21-22%) in the posterior cingulate cortex and cinguloparietal transitional area in patients with very early Alzheimer’s disease. Mean metabolic reduction in the posterior cingulate cortex was significantly greater than that in the lateral neocortices or parahippocampal cortex. The result suggests a functional importance for the posterior cingulate cortex in impairment of learning and memory, which is a feature of very early Alzheimer’s disease. Minoshima S, Giordani B, Berent S, Frey KA, Foster NL, Kuhl DE. -

A Hypothesis for the Evolution of the Upper Layers of the Neocortex Through Co-Option of the Olfactory Cortex Developmental Program
HYPOTHESIS AND THEORY published: 12 May 2015 doi: 10.3389/fnins.2015.00162 A hypothesis for the evolution of the upper layers of the neocortex through co-option of the olfactory cortex developmental program Federico Luzzati 1, 2* 1 Department of Life Sciences and Systems Biology (DBIOS), University of Turin, Turin, Italy, 2 Neuroscience Institute Cavalieri Ottolenghi, Orbassano, Truin, Italy The neocortex is unique to mammals and its evolutionary origin is still highly debated. The neocortex is generated by the dorsal pallium ventricular zone, a germinative domain that in reptiles give rise to the dorsal cortex. Whether this latter allocortical structure Edited by: contains homologs of all neocortical cell types it is unclear. Recently we described a Francisco Aboitiz, + + Pontificia Universidad Catolica de population of DCX /Tbr1 cells that is specifically associated with the layer II of higher Chile, Chile order areas of both the neocortex and of the more evolutionary conserved piriform Reviewed by: cortex. In a reptile similar cells are present in the layer II of the olfactory cortex and Loreta Medina, the DVR but not in the dorsal cortex. These data are consistent with the proposal that Universidad de Lleida, Spain Gordon M. Shepherd, the reptilian dorsal cortex is homologous only to the deep layers of the neocortex while Yale University School of Medicine, the upper layers are a mammalian innovation. Based on our observations we extended USA Fernando Garcia-Moreno, these ideas by hypothesizing that this innovation was obtained by co-opting a lateral University of Oxford, UK and/or ventral pallium developmental program. Interestingly, an analysis in the Allen brain *Correspondence: atlas revealed a striking similarity in gene expression between neocortical layers II/III Federico Luzzati, and piriform cortex. -

Anterograde and Retrograde Amnesia in Rats with Large Hippocampal Lesions
HIPPOCAMPUS 11:18–26 (2001) Anterograde and Retrograde Amnesia in Rats With Large Hippocampal Lesions Gordon Winocur,1–4* Robert M. McDonald,3 and Morris Moscovitch1,5 1Department of Psychology, Rotman Research Institute, Baycrest Centre for Geriatric Care, Toronto, Ontario, Canada 2Department of Psychology, Trent University, Peterborough, Ontario, Canada 3Department of Psychology, University of Toronto, Toronto, Ontario, Canada 4Department of Psychiatry, University of Toronto, Toronto, Ontario, Canada 5Department of Psychology, Erindale College, University of Toronto, Toronto, Ontario, Canada ABSTRACT: A test of socially acquired food preferences was used to study durable representation, available for future recall (Mil- the effects of large lesions to the hippocampal formation (HPC) on antero- ner, 1966; Squire, 1992). Two related predictions follow grade and retrograde memory in rats. In the anterograde test, rats with HPC from this position. The first pertains to anterograde lesions normally acquired the food preference but showed a faster rate of forgetting than control groups. When the food preference was acquired memory and asserts that hippocampal damage will result preoperatively, HPC groups exhibited a temporally graded retrograde amne- in impaired memory for events after long delays (long- sia in which memory was impaired when the preference was acquired within term memory, LTM), but not after relatively short delays 2 days of surgery but not at longer delays. The results support the traditional (short-term memory, STM). The rationale is that, in theory that the HPC contributes to the consolidation of newly acquired information into a durable memory trace that is represented in other brain STM, information is held and recalled for a brief period areas. -

Reversible Verbal and Visual Memory Deficits After Left Retrosplenial Infarction
Journal of Clinical Neurology / Volume 3 / March, 2007 Case Report Reversible Verbal and Visual Memory Deficits after Left Retrosplenial Infarction Jong Hun Kim, M.D.*, Kwang-Yeol Park, M.D.†, Sang Won Seo, M.D.*, Duk L. Na, M.D.*, Chin-Sang Chung, M.D.*, Kwang Ho Lee, M.D.*, Gyeong-Moon Kim, M.D.* *Department of Neurology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea †Department of Neurology, Chung-Ang University Medical Center, Chung-Ang University School of Medicine, Seoul, Korea The retrosplenial cortex is a cytoarchitecturally distinct brain structure located in the posterior cingulate gyrus and bordering the splenium, precuneus, and calcarine fissure. Functional imaging suggests that the retrosplenium is involved in memory, visuospatial processing, proprioception, and emotion. We report on a patient who developed reversible verbal and visual memory deficits following a stroke. Neuro- psychological testing revealed both anterograde and retrograde memory deficits in verbal and visual modalities. Brain diffusion-weighted and T2-weighted magnetic resonance imaging (MRI) demonstrated an acute infarction of the left retrosplenium. J Clin Neurol 3(1):62-66, 2007 Key Words : Retrosplenium, Memory, Amnesia We report on a patient who developed both verbal INTRODUCTION and visual memory deficits after an acute infarction of the retrosplenial cortex. The main structures related to human memory are the Papez circuit, the basolateral limbic circuit, and the basal forebrain, which communicate with each other through white-matter tracts. Damage to these structures (in- cluding the communication tracts) from hemorrhages, infarctions, and tumors can result in memory dis- turbances.1,2 In addition to these structures, Valenstein et al.