1 | Page October 12, 2016 Andrew Slavitt Acting Administrator
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Medical Review(S) Clinical Review
CENTER FOR DRUG EVALUATION AND RESEARCH APPLICATION NUMBER: 200327 MEDICAL REVIEW(S) CLINICAL REVIEW Application Type NDA Application Number(s) 200327 Priority or Standard Standard Submit Date(s) December 29, 2009 Received Date(s) December 30, 2009 PDUFA Goal Date October 30, 2010 Division / Office Division of Anti-Infective and Ophthalmology Products Office of Antimicrobial Products Reviewer Name(s) Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD Review Completion October 29, 2010 Date Established Name Ceftaroline fosamil for injection (Proposed) Trade Name Teflaro Therapeutic Class Cephalosporin; ß-lactams Applicant Cerexa, Inc. Forest Laboratories, Inc. Formulation(s) 400 mg/vial and 600 mg/vial Intravenous Dosing Regimen 600 mg every 12 hours by IV infusion Indication(s) Acute Bacterial Skin and Skin Structure Infection (ABSSSI); Community-acquired Bacterial Pneumonia (CABP) Intended Population(s) Adults ≥ 18 years of age Template Version: March 6, 2009 Reference ID: 2857265 Clinical Review Ariel Ramirez Porcalla, MD, MPH Neil Rellosa, MD NDA 200327: Teflaro (ceftaroline fosamil) Table of Contents 1 RECOMMENDATIONS/RISK BENEFIT ASSESSMENT ......................................... 9 1.1 Recommendation on Regulatory Action ........................................................... 10 1.2 Risk Benefit Assessment.................................................................................. 10 1.3 Recommendations for Postmarketing Risk Evaluation and Mitigation Strategies ........................................................................................................................ -

Surgical Decolonization and Prophylaxis
Surgical Decolonization and Prophylaxis SurgicalSurgical Decolonization Decolonization and Prophylaxis and Prophylaxis DECOLONIZATION DECOLONIZATION Nasal Screening Result Recommended Intervention Nasal Screening Result Recommended Intervention MRSA Negative • No decolonization required MSSAMRSA NegativeNegative • No decolonization required MSSA PositiveNegative • Intranasal mupirocin twice daily x 5 days MSSA Positive • Intranasal mupirocin twice daily x 5 days MRSA Positive • Intranasal mupirocin twice daily x 5 days, MRSA Positive AND• Intranasal mupirocin twice daily x 5 days, •ANDChlorhexidine bathing one day prior to surgery ANTIMICROBIAL• ChlorhexidinePROPHYLAXISbathing one day prior to surgery ANTIMICROBIAL PROPHYLAXIS CLINICAL CONSIDERATIONS • Preoperative dose-timing CLINICAL CONSIDERATIONS • PreoperativeWithin 60 minutesdose-timing of surgical incision WithinExceptions: 60 minutes vancomycin of surgicaland fluoroquinolones incision within 120 minutes of surgical incisionExceptions: vancomycin and fluoroquinolones within 120 minutes of surgical • Weightincision-based dosing • WeightCefazolin-based: 2 gm dosingfor patients <120 kg, and 3 gm for patients ≥120 kg Vancomycin:Cefazolin: 2 gm usefor ABW patients <120 kg, and 3 gm for patients ≥120 kg Gentamicin:Vancomycin: use use ABW ABW unless ABW is >120% of their IBW, in which case use AdjBWGentamicin:(see below use ABW for equation)unless ABW is >120% of their IBW, in which case use • DurationAdjBW of(see prophylaxis below for equation) • DurationA single of dose, prophylaxis -

Severe Sepsis Tipsheet
Severe Sepsis Tipsheet SEVERE SEPSIS A, B, and C of the following must be met within 6 hours of each other. A. Documentation of a suspected infection. There may be reference to “possible infection,” “suspect infection,” “rule out infection,” or similar documentation. B. TWO or more manifestations of systemic infection according to the Systemic Inflammatory Response Syndrome (SIRS) criteria, which are: • Temperature > 38.3 C or < 36.0 C (> 100.9 F or < 96.8 F) • Heart rate (pulse) > 90 • Respiration > 20 per minute • White blood cell count > 12,000 or < 4,000 or > 10% bands C. Organ dysfunction, evidenced by any one of the following: • Systolic blood pressure (SBP) < 90 mmHg, or mean arterial pressure < 65 mmHg, or a systolic blood pressure decrease of more than 40 mmHg. Physician/APN/PA documentation must be present in the medical record indicating a > 40 mmHg decrease in SBP has occurred and is related to infection, severe sepsis or septic shock and not other causes. • Acute respiratory failure as evidenced by a new need for invasive or non-invasive mechanical ventilation. To use acute respiratory failure as a sign of organ dysfunction there must be: • Documentation of acute respiratory failure AND • Documentation the patient is on mechanical ventilation • Invasive mechanical ventilation requires an endotracheal or tracheostomy tube. Non-invasive mechanical ventilation may be referred to as BiPAP or CPAP. • New need for mechanical ventilation indicates the patient was not using the same type of mechanical ventilation prior to the current acute respiratory failure. • Use the time at which there is documentation the patient has both acute respiratory failure and is on mechanical ventilation. -

Antimicrobial Stewardship Guidance
Antimicrobial Stewardship Guidance Federal Bureau of Prisons Clinical Practice Guidelines March 2013 Clinical guidelines are made available to the public for informational purposes only. The Federal Bureau of Prisons (BOP) does not warrant these guidelines for any other purpose, and assumes no responsibility for any injury or damage resulting from the reliance thereof. Proper medical practice necessitates that all cases are evaluated on an individual basis and that treatment decisions are patient-specific. Consult the BOP Clinical Practice Guidelines Web page to determine the date of the most recent update to this document: http://www.bop.gov/news/medresources.jsp Federal Bureau of Prisons Antimicrobial Stewardship Guidance Clinical Practice Guidelines March 2013 Table of Contents 1. Purpose ............................................................................................................................................. 3 2. Introduction ...................................................................................................................................... 3 3. Antimicrobial Stewardship in the BOP............................................................................................ 4 4. General Guidance for Diagnosis and Identifying Infection ............................................................. 5 Diagnosis of Specific Infections ........................................................................................................ 6 Upper Respiratory Infections (not otherwise specified) .............................................................................. -
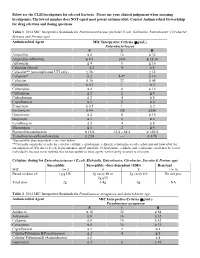
Below Are the CLSI Breakpoints for Selected Bacteria. Please Use Your Clinical Judgement When Assessing Breakpoints
Below are the CLSI breakpoints for selected bacteria. Please use your clinical judgement when assessing breakpoints. The lowest number does NOT equal most potent antimicrobial. Contact Antimicrobial Stewardship for drug selection and dosing questions. Table 1: 2014 MIC Interpretive Standards for Enterobacteriaceae (includes E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia and Proteus spp) Antimicrobial Agent MIC Interpretive Criteria (g/mL) Enterobacteriaceae S I R Ampicillin ≤ 8 16 ≥ 32 Ampicillin-sulbactam ≤ 8/4 16/8 ≥ 32/16 Aztreonam ≤ 4 8 ≥ 16 Cefazolin (blood) ≤ 2 4 ≥ 8 Cefazolin** (uncomplicated UTI only) ≤ 16 ≥ 32 Cefepime* ≤ 2 4-8* ≥ 16 Cefotetan ≤ 16 32 ≥ 64 Ceftaroline ≤ 0.5 1 ≥ 2 Ceftazidime ≤ 4 8 ≥ 16 Ceftriaxone ≤ 1 2 ≥ 4 Cefpodoxime ≤ 2 4 ≥ 8 Ciprofloxacin ≤ 1 2 ≥ 4 Ertapenem ≤ 0.5 1 ≥ 2 Fosfomycin ≤ 64 128 ≥256 Gentamicin ≤ 4 8 ≥ 16 Imipenem ≤ 1 2 ≥ 4 Levofloxacin ≤ 2 4 ≥ 8 Meropenem ≤ 1 2 ≥ 4 Piperacillin-tazobactam ≤ 16/4 32/4 – 64/4 ≥ 128/4 Trimethoprim-sulfamethoxazole ≤ 2/38 --- ≥ 4/76 *Susceptibile dose-dependent – see chart below **Cefazolin can predict results for cefaclor, cefdinir, cefpodoxime, cefprozil, cefuroxime axetil, cephalexin and loracarbef for uncomplicated UTIs due to E.coli, K.pneumoniae, and P.mirabilis. Cefpodoxime, cefinidir, and cefuroxime axetil may be tested individually because some isolated may be susceptible to these agents while testing resistant to cefazolin. Cefepime dosing for Enterobacteriaceae ( E.coli, Klebsiella, Enterobacter, Citrobacter, Serratia & Proteus spp) Susceptible Susceptible –dose-dependent (SDD) Resistant MIC </= 2 4 8 >/= 16 Based on dose of: 1g q12h 1g every 8h or 2g every 8 h Do not give 2g q12 Total dose 2g 3-4g 6g NA Table 2: 2014 MIC Interpretive Standards for Pseudomonas aeruginosa and Acinetobacter spp. -

AMEG Categorisation of Antibiotics
12 December 2019 EMA/CVMP/CHMP/682198/2017 Committee for Medicinal Products for Veterinary use (CVMP) Committee for Medicinal Products for Human Use (CHMP) Categorisation of antibiotics in the European Union Answer to the request from the European Commission for updating the scientific advice on the impact on public health and animal health of the use of antibiotics in animals Agreed by the Antimicrobial Advice ad hoc Expert Group (AMEG) 29 October 2018 Adopted by the CVMP for release for consultation 24 January 2019 Adopted by the CHMP for release for consultation 31 January 2019 Start of public consultation 5 February 2019 End of consultation (deadline for comments) 30 April 2019 Agreed by the Antimicrobial Advice ad hoc Expert Group (AMEG) 19 November 2019 Adopted by the CVMP 5 December 2019 Adopted by the CHMP 12 December 2019 Official address Domenico Scarlattilaan 6 ● 1083 HS Amsterdam ● The Netherlands Address for visits and deliveries Refer to www.ema.europa.eu/how-to-find-us Send us a question Go to www.ema.europa.eu/contact Telephone +31 (0)88 781 6000 An agency of the European Union © European Medicines Agency, 2020. Reproduction is authorised provided the source is acknowledged. Categorisation of antibiotics in the European Union Table of Contents 1. Summary assessment and recommendations .......................................... 3 2. Introduction ............................................................................................ 7 2.1. Background ........................................................................................................ -

Antibiotic Review
8/28/19 Disclosure 2 E. Monee’ Carter-Griffin DNP, MA, RN, ACNP-BC Antibiotic has no financial relationships with commercial Review interests to disclose. Any unlaBeled and/or E. Monee’ Carter-Griffin, DNP, MA, RN, ACNP-BC unapproved uses of drugs or products will Be Associate Chair for Advanced Practice Nursing disclosed. University of Texas at Arlington, College of Nursing & Health Innovation Dallas Pulmonary & Critical Care, PA • Identify the general characteristics of • SusceptiBility à determine which antibiotics. antibiotics a bacteria is sensitive to • Discuss the mechanism of action, • à pharmacokinetics, and spectrum of activity MIC lowest concentration of drug that inhiBits growth of the Bacteria for the most common antiBiotic drug classes. Antibiotic Objectives • Identify commonly prescriBed antiBiotics • Trough à lowest concentration of within the drug classes including dosage Lingo drug in bloodstream range and indication for prescribing. – Collect prior to administration of • Identify special considerations for specific drug antibiotics and/or drug classes. • Peak à highest concentration of • Practice appropriate prescriBing for common drug in bloodstream bacterial infections. • Review Basic antiBiotic stewardship 3 4 principles. • Penicillins • Bactericidal or bacteriostatic • Cephalosporins • Narrow or Broad spectrum • Carbapenems General • Can elicit allergic responses Antibiotic • MonoBactam Characteristics • Affect normal Body flora Drug Classes • Macrolides • Fluoroquinolones • Sulfonamides • Tetracyclines 5 6 1 8/28/19 • Penicillins – Natural penicillins 8 – Aminopenicillins Penicillins Penicillins – Anti-staphylococcal penicillins • Mechanism of action àbactericidal à – Anti-pseudomonal penicillins interrupts cell wall synthesis 7 • Pharmacokinetics • Spectrum of Activity – Gram positive organisms Natural – Penicillin G à mostly given IM, But Natural • Streptococcus species (e.g. S. Penicillins one variation can Be given IV Penicillins pyogenes) • Poorly aBsorBed via PO • Some enterococcus species (e.g. -

Computational Antibiotics Book
Andrew V DeLong, Jared C Harris, Brittany S Larcart, Chandler B Massey, Chelsie D Northcutt, Somuayiro N Nwokike, Oscar A Otieno, Harsh M Patel, Mehulkumar P Patel, Pratik Pravin Patel, Eugene I Rowell, Brandon M Rush, Marc-Edwin G Saint-Louis, Amy M Vardeman, Felicia N Woods, Giso Abadi, Thomas J. Manning Computational Antibiotics Valdosta State University is located in South Georgia. Computational Antibiotics Index • Computational Details and Website Access (p. 8) • Acknowledgements (p. 9) • Dedications (p. 11) • Antibiotic Historical Introduction (p. 13) Introduction to Antibiotic groups • Penicillin’s (p. 21) • Carbapenems (p. 22) • Oxazolidines (p. 23) • Rifamycin (p. 24) • Lincosamides (p. 25) • Quinolones (p. 26) • Polypeptides antibiotics (p. 27) • Glycopeptide Antibiotics (p. 28) • Sulfonamides (p. 29) • Lipoglycopeptides (p. 30) • First Generation Cephalosporins (p. 31) • Cephalosporin Third Generation (p. 32) • Fourth-Generation Cephalosporins (p. 33) • Fifth Generation Cephalosporin’s (p. 34) • Tetracycline antibiotics (p. 35) Computational Antibiotics Antibiotics Covered (in alphabetical order) Amikacin (p. 36) Cefempidone (p. 98) Ceftizoxime (p. 159) Amoxicillin (p. 38) Cefepime (p. 100) Ceftobiprole (p. 161) Ampicillin (p. 40) Cefetamet (p. 102) Ceftoxide (p. 163) Arsphenamine (p. 42) Cefetrizole (p. 104) Ceftriaxone (p. 165) Azithromycin (p.44) Cefivitril (p. 106) Cefuracetime (p. 167) Aziocillin (p. 46) Cefixime (p. 108) Cefuroxime (p. 169) Aztreonam (p.48) Cefmatilen ( p. 110) Cefuzonam (p. 171) Bacampicillin (p. 50) Cefmetazole (p. 112) Cefalexin (p. 173) Bacitracin (p. 52) Cefodizime (p. 114) Chloramphenicol (p.175) Balofloxacin (p. 54) Cefonicid (p. 116) Cilastatin (p. 177) Carbenicillin (p. 56) Cefoperazone (p. 118) Ciprofloxacin (p. 179) Cefacetrile (p. 58) Cefoselis (p. 120) Clarithromycin (p. 181) Cefaclor (p. -

Management of Penicillin and Beta-Lactam Allergy
Management of Penicillin and Beta-Lactam Allergy (NB Provincial Health Authorities Anti-Infective Stewardship Committee, September 2017) Key Points • Beta-lactams are generally safe; allergic and adverse drug reactions are over diagnosed and over reported • Nonpruritic, nonurticarial rashes occur in up to 10% of patients receiving penicillins. These rashes are usually not allergic and are not a contraindication to the use of a different beta-lactam • The frequently cited risk of 8 to 10% cross-reactivity between penicillins and cephalosporins is an overestimate based on studies from the 1970’s that are now considered flawed • Expect new intolerances (i.e. any allergy or adverse reaction reported in a drug allergy field) to be reported after 0.5 to 4% of all antimicrobial courses depending on the gender and specific antimicrobial. Expect a higher incidence of new intolerances in patients with three or more prior medication intolerances1 • For type-1 immediate hypersensitivity reactions (IgE-mediated), cross-reactivity among penicillins (table 1) is expected due to similar core structure and/or major/minor antigenic determinants, use not recommended without desensitization • For type-1 immediate hypersensitivity reactions, cross-reactivity between penicillins (table 1) and cephalosporins is due to similarities in the side chains; risk of cross-reactivity will only be significant between penicillins and cephalosporins with similar side chains • Only type-1 immediate hypersensitivity to a penicillin manifesting as anaphylaxis, bronchospasm, -

Antibiotic Use for Sepsis in Neonates and Children: 2016 Evidence Update
Antibiotic Use for Sepsis in Neonates and Children: 2016 Evidence Update Aline Fuchsa, Julia Bielickia,b, Shrey Mathurb, Mike Sharlandb, Johannes N. Van Den Ankera,c a Paediatric Pharmacology and Pharmacometrics, University Children's Hospital Basel, Basel, Switzerland b Paediatric Infectious Disease Research Group, Institute for Infection and Immunity, St George's University of London, London, United Kingdom c Division of Clinical Pharmacology, Children’s National Health System, Washington, DC, USA WHO-Reviews 1 TABLE OF CONTENTS 1. INTRODUCTION ............................................................................................................................... 3 1.1. Aims ......................................................................................................................................... 3 1.2. Background ............................................................................................................................. 3 1.2.1. Definition and diagnosis ................................................................................................. 3 Neonatal Sepsis ............................................................................................................................... 3 Paediatric Sepsis ............................................................................................................................. 4 Community versus hospital acquired sepsis .................................................................................. 5 1.2.2. Microbiology .................................................................................................................. -

Comparative Activity of Newer Antibiotics Against Gram-Negative Bacilli
CONTRIBUTION • Comparative activity of newer antibiotics against gram-negative bacilli CYNTHIA C. KNAPP, MS AND JOHN A. WASHINGTON, MD • The in vitro activities of cefoperazone, cefotaxime, ceftriaxone, ceftazidime, azlocillin, mezlocillin, piperacillin, ticarcillin/clavulanate, aztreonam, imipenem, and ciprofloxacin were concurrently deter- mined against over 1,000 isolates of gram-negative bacilli from clinical specimens of patients at the Cleve- land Clinic. Cephalosporins, penicillins, and aztreonam were active against species of Enterobacteriaceae other than Citrobacter freundii, Enterobacter aerogenes, and Enterobacter cloacae. Ceftazidime was the most active cephalosporin against Pseudomonas aeruginosa. Against the Enterobacteriaceae, the rank order of activity of penicillins was ticarcillin/clavulanate > piperacillin > mezlocillin > azlocillin. Against P. aer- uginosa, the rank order of activity of penicillins was piperacillin > ticarcillin/clavulanate > azlocillin > mezlocillin. Aztreonam was less active v P. aeruginosa than ceftazidime, cefoperazone, or piperacillin. The most active antimicrobials against all isolates tested were imipenem and ciprofloxacin. • INDEX TERMS: ANTIBIOTICS, LACTAM; GRAM-NEGATIVE BACTERIA • CLEVE CLIN J MED 1989; 56:161-166 HE RECENT introduction of ciprofloxacin for and penicillins, as well as the monobactam, aztreonam, clinical use follows closely a period of active re- and the carbapenem, imipenem.1-3 search in and development of expanded spec- We compared the susceptibility of more than 1,000 trum p-lactam antibiotics, including cephalo- clinical bacterial isolates to four expanded spectrum Tsporins, penicillins, monobactams, and carbapenems. cephalosporins (cefoperazone, cefotaxime, ceftriaxone, Although numerous published studies compare the ac- and ceftazidime), four expanded spectrum penicillins tivity of ciprofloxacin with other quinolones, only a (azlocillin, mezlocillin, piperacillin, and ticarcil- limited number of studies compare the activity of ci- lin/clavulanate), aztreonam, imipenem, and ciproflox- acin. -

Surgical Prophylaxis Antibiotic Recommendations Updated 2017
Surgical Prophylaxis Antibiotic Recommendations Updated 2017 1. Prophylactic antibiotic is to be initiated within 1 hour prior to incision Exception: Due to the longer infusion time required, initiate vancomycin and ciprofloxacin within 2 hours prior to incision 2. Anesthesia to give preoperative antibiotic dose and document time on anesthesia record. Exception: Antibiotics in Endoscopy, Labor and Delivery and Birth Care Center 3. Chose alternative antibiotic if patient is allergic to recommended first line antibiotic. Avoid giving cefazolin or cefotetan if the patient has allergies to cephalosporins or a life- threatening allergy to penicillin (angioedema, bronchoconstriction, cardiac arrhythmias, hypotension, or respiratory distress) 4. Pediatric Patients: Maximum dose is dose listed for wt <80 kg. For pediatric patients >80 kg, follow adult dosing. 5. For patients colonized with MRSA: Give vancomycin as the pre-op antibiotics for cardiac, vascular, orthopedic, neurological, and head and neck surgeries. For all other surgeries, give vancomycin in addition to the antibiotics listed below. To determine if a patient is colonized with MRSA, please see flow sheet below. 6. Per Clinical Practice Restricted/High Risk Medications with Special Requirements Policy: Pre-operative antibiotics for prophylaxis in adult patients will be automatically substituted to the antibiotics listed on Adult Surgical Pre-Operative Antibiotic Prophylaxis order set #347. For surgeries/procedures not addressed on the order set, anesthesia is to discuss antibiotic selection with the surgeon. 7. Antibiotics listed below are listed in order of preference. The first line antibiotic has been designated with an asterisks(*). Dose Surgical Procedure Drug Pediatrics (<80 kg), Wt <80 kg Wt 80-119 kg Wt ≥ 120 kg If applicable Coronary Artery Bypass Graft Cardiac Surgery cefazolin IV* 1 gm 2 gm 3 gm Vascular Surgery All CABG, Valves and other CV procedures Pacemakers, AICD’s, pocket vancomycin IV (MRSA or 1 gm 1.5 gm 2 gm relocation and lead revisions, allergy) generator replacements.