The Role of Bcl-2 Modifying Factor (Bmf) in Airway Epithelial Cell Death
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

Multidrug Transporter MRP4/ABCC4 As a Key Determinant of Pancreatic
www.nature.com/scientificreports OPEN Multidrug transporter MRP4/ ABCC4 as a key determinant of pancreatic cancer aggressiveness A. Sahores1, A. Carozzo1, M. May1, N. Gómez1, N. Di Siervi1, M. De Sousa Serro1, A. Yanef1, A. Rodríguez‑González2, M. Abba3, C. Shayo2 & C. Davio1* Recent fndings show that MRP4 is critical for pancreatic ductal adenocarcinoma (PDAC) cell proliferation. Nevertheless, the signifcance of MRP4 protein levels and function in PDAC progression is still unclear. The aim of this study was to determine the role of MRP4 in PDAC tumor aggressiveness. Bioinformatic studies revealed that PDAC samples show higher MRP4 transcript levels compared to normal adjacent pancreatic tissue and circulating tumor cells express higher levels of MRP4 than primary tumors. Also, high levels of MRP4 are typical of high-grade PDAC cell lines and associate with an epithelial-mesenchymal phenotype. Moreover, PDAC patients with high levels of MRP4 depict dysregulation of pathways associated with migration, chemotaxis and cell adhesion. Silencing MRP4 in PANC1 cells reduced tumorigenicity and tumor growth and impaired cell migration. Transcriptomic analysis revealed that MRP4 silencing alters PANC1 gene expression, mainly dysregulating pathways related to cell-to-cell interactions and focal adhesion. Contrarily, MRP4 overexpression signifcantly increased BxPC-3 growth rate, produced a switch in the expression of EMT markers, and enhanced experimental metastatic incidence. Altogether, our results indicate that MRP4 is associated with a more aggressive phenotype in PDAC, boosting pancreatic tumorigenesis and metastatic capacity, which could fnally determine a fast tumor progression in PDAC patients. Pancreatic ductal adenocarcinoma (PDAC) is one of the most lethal human malignancies, due to its late diag- nosis, inherent resistance to treatment and early dissemination 1. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

Bcl-2–Modifying Factor Induces Renal Proximal Tubular Cell Apoptosis in Diabetic Mice Garnet J
ORIGINAL ARTICLE Bcl-2–Modifying Factor Induces Renal Proximal Tubular Cell Apoptosis in Diabetic Mice Garnet J. Lau,1 Nicolas Godin,1 Hasna Maachi,1 Chao-Sheng Lo,1 Shyh-Jong Wu,1 Jian-Xin Zhu,1 Marie-Luise Brezniceanu,1 Isabelle Chénier,1 Joelle Fragasso-Marquis,1 Jean-Baptiste Lattouf,1 Jean Ethier,1 Janos G. Filep,2 Julie R. Ingelfinger,3 Viji Nair,4 Matthias Kretzler,4 Clemens D. Cohen,5 Shao-Ling Zhang,1 and John S.D. Chan1 – This study investigated the mechanisms underlying tubular apopto- than glomerular pathology (2 5). For example, examination sis in diabetes by identifying proapoptotic genes that are differen- of nephrons from proteinuric diabetic patients shows that tially upregulated by reactive oxygen species in renal proximal 71% of glomeruli display glomerulotubular junction abnor- tubular cells (RPTCs) in models of diabetes. Total RNAs isolated malities and 8–17% of glomeruli are atubular glomeruli (6,7). from renal proximal tubules (RPTs) of 20-week-old heterozygous The mechanisms underlying tubular atrophy are incom- db/m+, db/db,anddb/db catalase (CAT)-transgenic (Tg) mice were pletely delineated. Studies have shown that high glucose used for DNA chip microarray analysis. Real-time quantitative PCR (HG) concentrations are associated with increased reactive assays, immunohistochemistry, and mice rendered diabetic with oxygen species (ROS) production, which inhibits proximal streptozotocin were used to validate the proapoptotic gene expres- – sion in RPTs. Cultured rat RPTCs were used to confirm the apoptotic tubular function and induces apoptosis (8 10). Apoptosis activity and regulation of proapoptotic gene expression. Addition- has been detected in renal proximal tubular cells (RPTCs) ally, studies in kidney tissues from patients with and without diabe- of diabetic mice (11,12) and rats (13,14) as well as in RPTCs teswereusedtoconfirm enhanced proapoptotic gene expression in of diabetic patients (15–17), suggesting that tubular apo- RPTs. -
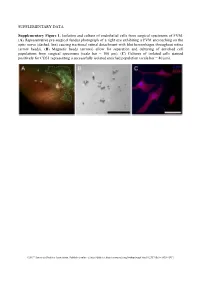
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Supplemental Information
Supplemental Information Supplemental Figures Supplemental Figure 1. Quantitation of bioluminescent imaging data in Figure 1B. 1 Supplemental Figure 2. Impact of Brg1 shRNAs on proliferation of immortalized murine embryonic fibroblasts (iMEFs), BCR-ABL/p19-/- B cell acute lymphoblastic leukemia (B-ALL), murine breast cancer cells (4T1), and BrafV600E melanoma. A) shRNA/GFP competition assay in indicated murine cell lines. Rpa3 shRNA targeting replication protein A3 serves as a positive control. shRNA targeting Renilla luciferase is included as a negative control. B) Western blotting for additional Brg1 shRNAs in the indicated cell lines transduced with the Ren or Brg1 LMN-shRNAs. A representative experiment of three biological replicates is shown. 2 Supplemental Figure 3. Effect of Brg1 shRNAs on proliferation of human leukemia cell lines. A) shRNA/GFP competition assay in the indicated human cell lines with shRen.713, shBrg1.4471, or shSpt16.3249. All shRNAs were evaluated in a timecourse of GFP% measurements at days 4, 8, 12, 16, 20, and 24. CML-BC indicates chronic myeloid leukemia blast crisis. B) Western blotting of SPT16 levels in whole cell lysates prepared from HeLa cells transduced with the indicated MLP shRNA constructs following puromycin selection. 3 Supplemental Figure 4. Expression and functional validation of SWI/SNF subunits in MLL-AF9/NrasG12D leukemia. A) RPKM values for genes encoding SWI/SNF complex subunits from polyA+ tail RNA-seq performed in RN2 cell line. B) shRNA/GFP competition assay with two independent shRNAs targeting indicated SWI/SNF subunits. Experiments were performed in RN2 cells as in Figure 1A. n=2-3. C) RT-qPCR was performed to test the knockdown efficiency of indicted SWI/SNF subunit shRNAs. -

Proteins Interact with Their Pro-Survival Partner MCL-1
Promiscuous and selective: how intrinsically disordered BH3- proteins interact with their pro-survival partner MCL-1. Liza Dahal, Tristan O.C. Kwan, Jeffery J. Hollins and Jane Clarke1 1Corresponding author, Email: [email protected] Department of Chemistry, University of Cambridge, Lensfield Road, Cambridge, CB2 1EW Highlights • Despite differences in residual helicity BH3 peptides bind equally fast to MCL-1. • Differences in stabilities results from off-rates, altering complex lifetimes. • This difference arises from variation in interactions at MCL-1 binding interface. GRAPHICAL ABSTRACT Promiscuous and selective: how intrinsically disordered BH3- proteins interact with their pro-survival partner MCL-1. Liza Dahal, Tristan O.C. Kwan, JefFery J. Hollins and Jane Clarke1 1Corresponding author, Email: [email protected] Department of Chemistry, University of Cambridge, Lensfield Road, Cambridge, CB2 1EW Abstract 1 The BCL-2 family of proteins plays a central role in regulating cell survival and apoptosis. Disordered BH3-only proteins bind promiscuously to a number of different BCL-2 proteins, with binding affinities that vary by orders of magnitude. Here we investigate the basis for these differences in affinity. We show that eight different disordered BH3 proteins all bind to their BCL-2 partner (MCL-1) very rapidly, and that the differences in sequences result in different dissociation rates. Similarly, mutation of the binding surface of MCL-1 generally affects association kinetics in the same way for all BH3 peptides but has significantly -

Database and Bioinformatic Analysis of BCL-2 Family Proteins and BH3-Only Proteins Abdel Aouacheria, Vincent Navratil, Christophe Combet
Database and Bioinformatic Analysis of BCL-2 Family Proteins and BH3-Only Proteins Abdel Aouacheria, Vincent Navratil, Christophe Combet To cite this version: Abdel Aouacheria, Vincent Navratil, Christophe Combet. Database and Bioinformatic Analysis of BCL-2 Family Proteins and BH3-Only Proteins. Gavathiotis E. BCL-2 Family Proteins. Methods in Molecular Biology, 1877, Springer Nature, pp.23-43, 2019, Methods in Molecular Biology, 978-1-4939- 8860-0. 10.1007/978-1-4939-8861-7_2. hal-02347884 HAL Id: hal-02347884 https://hal.archives-ouvertes.fr/hal-02347884 Submitted on 5 Nov 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Copyright Database and bioinformatic analysis of BCL-2 family proteins and BH3-only proteins Abdel Aouacheria 1,*, Vincent Navratil 2 and Christophe Combet 3 1 ISEM, Institut des Sciences de l’Evolution de Montpellier, Université de Montpellier, UMR 5554, CNRS, IRD, EPHE, Place Eugène Bataillon, 34095 Montpellier, France 2 PRABI, Rhône Alpes Bioinformatics Center, UCBL, Lyon1, Université de Lyon, Lyon, France. 3 Centre de Recherche en Cancérologie de Lyon, UMR Inserm U1052, CNRS 5286, Université Claude Bernard Lyon 1, Centre Léon Bérard, Lyon, France. -

UC San Diego Electronic Theses and Dissertations
UC San Diego UC San Diego Electronic Theses and Dissertations Title Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent Permalink https://escholarship.org/uc/item/7m04f0b0 Author Buchholz, Kyle Stephen Publication Date 2016 Peer reviewed|Thesis/dissertation eScholarship.org Powered by the California Digital Library University of California UNIVERSITY OF CALIFORNIA, SAN DIEGO Cardiac Stretch-Induced Transcriptomic Changes are Axis-Dependent A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Bioengineering by Kyle Stephen Buchholz Committee in Charge: Professor Jeffrey Omens, Chair Professor Andrew McCulloch, Co-Chair Professor Ju Chen Professor Karen Christman Professor Robert Ross Professor Alexander Zambon 2016 Copyright Kyle Stephen Buchholz, 2016 All rights reserved Signature Page The Dissertation of Kyle Stephen Buchholz is approved and it is acceptable in quality and form for publication on microfilm and electronically: Co-Chair Chair University of California, San Diego 2016 iii Dedication To my beautiful wife, Rhia. iv Table of Contents Signature Page ................................................................................................................... iii Dedication .......................................................................................................................... iv Table of Contents ................................................................................................................ v List of Figures ................................................................................................................... -

Non-Canonical Phosphorylation of Bmf by P38 MAPK Promotes Its
www.nature.com/cdd ARTICLE Non-canonical phosphorylation of Bmf by p38 MAPK promotes its apoptotic activity in anoikis ✉ Zhe Zhi1, Zhenlin Ouyang2, Yibo Ren1, Ying Cheng3, Peijun Liu 4, Yurong Wen2 and Yongping Shao 1,5 © The Author(s), under exclusive licence to ADMC Associazione Differenziamento e Morte Cellulare 2021 Bmf contributes to the onset of anoikis by translocating from cytoskeleton to mitochondria when cells lose attachment to the extracellular matrix. However, the structural details of Bmf cytoskeleton tethering and the control of Bmf release upon loss of anchorage remained unknown. Here we showed that cell detachment induced rapid and sustained activation of p38 MAPK in mammary epithelial cell lines. Inhibition of p38 signaling or Bmf knockdown rescued anoikis. Activated p38 MAPK could directly phosphorylate Bmf at multiple sites including a non-proline-directed site threonine 72 (T72). Crystallographic studies revealed that Bmf T72 directly participated in DLC2 binding and its phosphorylation would block Bmf/DLC2 interaction through steric hindrance. Finally, we showed that phosphomimetic mutation of T72 enhanced Bmf apoptotic activity in vitro and in a knock-in mouse model. This work unraveled a novel regulatory mechanism of Bmf activity during anoikis and provided structural basis for Bmf cytoskeleton tethering and dissociation. Cell Death & Differentiation; https://doi.org/10.1038/s41418-021-00855-3 INTRODUCTION sequences of Bim and Bmf, Lei et al. proposed that Bmf might Anoikis is a special type of programed cell death triggered when also be phosphorylated by JNK at serine 74 (S74) proximal to the cells lose attachment to extracellular matrix (ECM) [1]. -

Down-Regulation of BCL2L13 Renders Poor Prognosis in Clear Cell And
Meng et al. Cancer Cell Int (2021) 21:332 https://doi.org/10.1186/s12935-021-02039-y Cancer Cell International PRIMARY RESEARCH Open Access Down-regulation of BCL2L13 renders poor prognosis in clear cell and papillary renal cell carcinoma Fei Meng1,2,3 , Luojin Zhang4, Mingjun Zhang5, Kaiqin Ye1,3, Wei Guo1,2, Yu Liu1,2,3, Wulin Yang1,3, Zhimin Zhai4, Hongzhi Wang1,3, Jun Xiao6* and Haiming Dai1,3* Abstract Background: BCL2L13 belongs to the BCL2 super family, with its protein product exhibits capacity of apoptosis-medi- ating in diversifed cell lines. Previous studies have shown that BCL2L13 has functional consequence in several tumor types, including ALL and GBM, however, its function in kidney cancer remains as yet unclearly. Methods: Multiple web-based portals were employed to analyze the efect of BCL2L13 in kidney cancer using the data from TCGA database. Functional enrichment analysis and hubs of BCL2L13 co-expressed genes in clear cell renal cell carcinoma (ccRCC) and papillary renal cell carcinoma (pRCC) were carried out on Cytoscape. Evaluation of BCL2L13 protein level was accomplished through immunohistochemistry on parafn embedded renal cancer tissue sections. Western blotting and fow cytometry were implemented to further analyze the pro-apoptotic function of BCL2L13 in ccRCC cell line 786-0. Results: BCL2L13 expression is signifcantly decreased in ccRCC and pRCC patients, however, mutations and copy number alterations are rarely observed. The poor prognosis of ccRCC that derived from down-regulated BCL2L13 is independent of patients’ gender or tumor grade. Furthermore, BCL2L13 only weakly correlates with the genes that mutated in kidney cancer or the genes that associated with inherited kidney cancer predisposing syndrome, while actively correlates with SLC25A4. -
Bim and Bmf in Tissue Homeostasis and Malignant Disease
Oncogene (2009) 27, S41–S52 & 2009 Macmillan Publishers Limited All rights reserved 0950-9232/09 $32.00 www.nature.com/onc REVIEW Bim and Bmf in tissue homeostasis and malignant disease JD Pin˜ on1, V Labi2, A Egle1 and A Villunger2 1Laboratory for Immunological and Molecular Cancer Research, University Hospital Salzburg, Salzburg, Austria and 2Division of Developmental Immunology, Biocenter, Innsbruck Medical University, Innsbruck, Austria Among all BH3-only proteins known to date, most proteins for localization to intra-cellular membranes, information is available on the biological role and function and this was also confirmed for Bim in overexpression of Bim (Bcl-2 interacting mediator of cell death)/BOD studies (O’Connor et al., 1998). Further analysis (Bcl-2 related ovarian death agonist), whereas little is still revealed that the pro-apoptotic and Bcl-2 antagonizing known about its closest relative, Bcl-2 modifying factor potential of Bim/Bod correlated inversely with protein (Bmf). Although Bim has been implicated in the regulation length, for reasons discussed below. Although BimEL of cell death induction in multiple cell types and tissues in and BimL can be found widely expressed in most tissues response to a large number of stimuli, including growth and cell lines, BimS expression is less abundant, factor or cytokine deprivation, calcium flux, ligation of presumably because of its strong pro-apoptotic potential antigen receptors on T and B cells, glucocorticoid or loss ((O’Reilly et al., 2000) and http://symatlas.gnf.org/ of adhesion, Bmf seems to play a more restricted role by SymAtlas). Today, more than a dozen splice variants supporting Bim in some of these cell death processes. -

Mining Prognostic Markers of Asian Hepatocellular Carcinoma Patients Based on the Apoptosis-Related Genes Junbin Yan, Jielu Cao and Zhiyun Chen*
Yan et al. BMC Cancer (2021) 21:175 https://doi.org/10.1186/s12885-021-07886-6 RESEARCH ARTICLE Open Access Mining prognostic markers of Asian hepatocellular carcinoma patients based on the apoptosis-related genes Junbin Yan, Jielu Cao and Zhiyun Chen* Abstract Background: Apoptosis-related genes(Args)play an essential role in the occurrence and progression of hepatocellular carcinoma(HCC). However, few studies have focused on the prognostic significance of Args in HCC. In the study, we aim to explore an efficient prognostic model of Asian HCC patients based on the Args. Methods: We downloaded mRNA expression profiles and corresponding clinical data of Asian HCC patients from The Cancer Genome Atlas (TCGA) and International Cancer Genome Consortium (ICGC) databases. The Args were collected from Deathbase, a database related to cell death, combined with the research results of GeneCards、National Center for Biotechnology Information (NCBI) databases and a lot of literature. We used Wilcoxon-test and univariate Cox analysis to screen the differential expressed genes (DEGs) and the prognostic related genes (PRGs) of HCC. The intersection genes of DEGs and PGGs were seen as crucial Args of HCC. The prognostic model of Asian HCC patients was constructed by least absolute shrinkage and selection operator (lasso)- proportional hazards model (Cox) regression analysis. Kaplan-Meier curve, Principal Component Analysis (PCA) analysis, t-distributed Stochastic Neighbor Embedding (t-SNE) analysis, risk score curve, receiver operating characteristic (ROC) curve, and the HCC data of ICGC database and the data of Asian HCC patients of Kaplan-Meier plotter database were used to verify the model.