Genetic Dissection of a Supergene Implicates Tfap2a in Craniofacial Evolution of Threespine Sticklebacks
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CLASP2 Antibody Product Type
PRODUCT INFORMATION Product name: CLASP2 antibody Product type: Primary antibodies Description: Rabbit polyclonal to CLASP2 Immunogen:3 synthetic peptides (human) conjugated to KLH Reacts with:Hu, Ms Tested applications:ELISA, WB and IF GENE INFORMATION Gene Symbol: CLASP2 Gene Name:cytoplasmic linker associated protein 2 Ensembl ID:ENSG00000163539 Entrez GeneID:23122 GenBank Accession number:AB014527 Swiss-Prot:O75122 Molecular weight of CLASP2: 165.9 & 108.6kDa Function:Microtubule plus-end tracking protein that promotes the stabilization of dynamic microtubules. Involved in the nucleation of noncentrosomal microtubules originating from the trans-Golgi network (TGN). Required for the polarization of the cytoplasmic microtubule arrays in migrating cells towards the leading edge of the cell. May act at the cell cortex to enhance the frequency of rescue of depolymerizing microtubules by attaching their plus- ends to cortical platforms composed of ERC1 and PHLDB2. This cortical microtubule stabilizing activity is regulated at least in part by phosphatidylinositol 3-kinase signaling. Also performs a similar stabilizing function at the kinetochore which is essential for the bipolar alignment of chromosomes on the mitotic spindle. Acts as a mediator of ERBB2- dependent stabilization of microtubules at the cell cortex. Expected subcellular localization:Cytoplasm › cytoskeleton. Cytoplasm › cytoskeleton › microtubule organizing center › centrosome. Chromosome › centromere › kinetochore. Cytoplasm › cytoskeleton › spindle. Golgi apparatus. Golgi apparatus › trans-Golgi network. Cell membrane. Cell projection › ruffle membrane. Note: Localizes to microtubule plus ends. Localizes to centrosomes, kinetochores and the mitotic spindle from prometaphase. Subsequently localizes to the spindle midzone from anaphase and to the midbody from telophase. In migrating cells localizes to the plus ends of microtubules within the cell body and to the entire microtubule lattice within the lamella. -

Mouse Phldb2 Knockout Project (CRISPR/Cas9)
https://www.alphaknockout.com Mouse Phldb2 Knockout Project (CRISPR/Cas9) Objective: To create a Phldb2 knockout Mouse model (C57BL/6J) by CRISPR/Cas-mediated genome engineering. Strategy summary: The Phldb2 gene (NCBI Reference Sequence: NM_001252442 ; Ensembl: ENSMUSG00000033149 ) is located on Mouse chromosome 16. 19 exons are identified, with the ATG start codon in exon 2 and the TAG stop codon in exon 19 (Transcript: ENSMUST00000076333). Exon 2 will be selected as target site. Cas9 and gRNA will be co-injected into fertilized eggs for KO Mouse production. The pups will be genotyped by PCR followed by sequencing analysis. Note: Mice homozygous for a conditional allele activated in neurons exhibit impaired LTP. Exon 2 starts from the coding region. Exon 2 covers 33.87% of the coding region. The size of effective KO region: ~1337 bp. The KO region does not have any other known gene. Page 1 of 9 https://www.alphaknockout.com Overview of the Targeting Strategy Wildtype allele 5' gRNA region gRNA region 3' 1 2 19 Legends Exon of mouse Phldb2 Knockout region Page 2 of 9 https://www.alphaknockout.com Overview of the Dot Plot (up) Window size: 15 bp Forward Reverse Complement Sequence 12 Note: The 2000 bp section upstream of Exon 2 is aligned with itself to determine if there are tandem repeats. Tandem repeats are found in the dot plot matrix. The gRNA site is selected outside of these tandem repeats. Overview of the Dot Plot (down) Window size: 15 bp Forward Reverse Complement Sequence 12 Note: The 2000 bp section downstream of Exon 2 is aligned with itself to determine if there are tandem repeats. -

SUPPLEMENTARY MATERIAL Bone Morphogenetic Protein 4 Promotes
www.intjdevbiol.com doi: 10.1387/ijdb.160040mk SUPPLEMENTARY MATERIAL corresponding to: Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells SUMIYO MIMURA, MIKA SUGA, KAORI OKADA, MASAKI KINEHARA, HIROKI NIKAWA and MIHO K. FURUE* *Address correspondence to: Miho Kusuda Furue. Laboratory of Stem Cell Cultures, National Institutes of Biomedical Innovation, Health and Nutrition, 7-6-8, Saito-Asagi, Ibaraki, Osaka 567-0085, Japan. Tel: 81-72-641-9819. Fax: 81-72-641-9812. E-mail: [email protected] Full text for this paper is available at: http://dx.doi.org/10.1387/ijdb.160040mk TABLE S1 PRIMER LIST FOR QRT-PCR Gene forward reverse AP2α AATTTCTCAACCGACAACATT ATCTGTTTTGTAGCCAGGAGC CDX2 CTGGAGCTGGAGAAGGAGTTTC ATTTTAACCTGCCTCTCAGAGAGC DLX1 AGTTTGCAGTTGCAGGCTTT CCCTGCTTCATCAGCTTCTT FOXD3 CAGCGGTTCGGCGGGAGG TGAGTGAGAGGTTGTGGCGGATG GAPDH CAAAGTTGTCATGGATGACC CCATGGAGAAGGCTGGGG MSX1 GGATCAGACTTCGGAGAGTGAACT GCCTTCCCTTTAACCCTCACA NANOG TGAACCTCAGCTACAAACAG TGGTGGTAGGAAGAGTAAAG OCT4 GACAGGGGGAGGGGAGGAGCTAGG CTTCCCTCCAACCAGTTGCCCCAAA PAX3 TTGCAATGGCCTCTCAC AGGGGAGAGCGCGTAATC PAX6 GTCCATCTTTGCTTGGGAAA TAGCCAGGTTGCGAAGAACT p75 TCATCCCTGTCTATTGCTCCA TGTTCTGCTTGCAGCTGTTC SOX9 AATGGAGCAGCGAAATCAAC CAGAGAGATTTAGCACACTGATC SOX10 GACCAGTACCCGCACCTG CGCTTGTCACTTTCGTTCAG Suppl. Fig. S1. Comparison of the gene expression profiles of the ES cells and the cells induced by NC and NC-B condition. Scatter plots compares the normalized expression of every gene on the array (refer to Table S3). The central line -

Long-Range Chromosome Interactions Mediated by Cohesin Shape Circadian Gene Expression
RESEARCH ARTICLE Long-Range Chromosome Interactions Mediated by Cohesin Shape Circadian Gene Expression Yichi Xu1,2, Weimin Guo1, Ping Li3, Yan Zhang3, Meng Zhao1,4, Zenghua Fan1,2, Zhihu Zhao3, Jun Yan1,4* 1 CAS-MPG Partner Institute for Computational Biology, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China, 2 University of Chinese Academy of Sciences, Shanghai, China, 3 Beijing Institute of Biotechnology, Beijing, China, 4 Institute of Neuroscience, State Key Laboratory a11111 of Neuroscience, CAS Center for Excellence in Brain Science, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences, Shanghai, China * [email protected] Abstract OPEN ACCESS Mammalian circadian rhythm is established by the negative feedback loops consisting of a Citation: Xu Y, Guo W, Li P, Zhang Y, Zhao M, Fan Z, et al. (2016) Long-Range Chromosome Interactions set of clock genes, which lead to the circadian expression of thousands of downstream Mediated by Cohesin Shape Circadian Gene genes in vivo. As genome-wide transcription is organized under the high-order chromosome Expression. PLoS Genet 12(5): e1005992. structure, it is largely uncharted how circadian gene expression is influenced by chromo- doi:10.1371/journal.pgen.1005992 some architecture. We focus on the function of chromatin structure proteins cohesin as well — Editor: Achim Kramer, Charité Universitätsmedizin as CTCF (CCCTC-binding factor) in circadian rhythm. Using circular chromosome confor- Berlin, GERMANY mation capture sequencing, we systematically examined the interacting loci of a Bmal1- Received: October 10, 2015 bound super-enhancer upstream of a clock gene Nr1d1 in mouse liver. These interactions Accepted: March 27, 2016 are largely stable in the circadian cycle and cohesin binding sites are enriched in the interac- Published: May 2, 2016 tome. -

Genomic and Transcriptome Analysis Revealing an Oncogenic Functional Module in Meningiomas
Neurosurg Focus 35 (6):E3, 2013 ©AANS, 2013 Genomic and transcriptome analysis revealing an oncogenic functional module in meningiomas XIAO CHANG, PH.D.,1 LINGLING SHI, PH.D.,2 FAN GAO, PH.D.,1 JONATHAN RUssIN, M.D.,3 LIYUN ZENG, PH.D.,1 SHUHAN HE, B.S.,3 THOMAS C. CHEN, M.D.,3 STEVEN L. GIANNOTTA, M.D.,3 DANIEL J. WEISENBERGER, PH.D.,4 GAbrIEL ZADA, M.D.,3 KAI WANG, PH.D.,1,5,6 AND WIllIAM J. MAck, M.D.1,3 1Zilkha Neurogenetic Institute, Keck School of Medicine, University of Southern California, Los Angeles, California; 2GHM Institute of CNS Regeneration, Jinan University, Guangzhou, China; 3Department of Neurosurgery, Keck School of Medicine, University of Southern California, Los Angeles, California; 4USC Epigenome Center, Keck School of Medicine, University of Southern California, Los Angeles, California; 5Department of Psychiatry, Keck School of Medicine, University of Southern California, Los Angeles, California; and 6Division of Bioinformatics, Department of Preventive Medicine, Keck School of Medicine, University of Southern California, Los Angeles, California Object. Meningiomas are among the most common primary adult brain tumors. Although typically benign, roughly 2%–5% display malignant pathological features. The key molecular pathways involved in malignant trans- formation remain to be determined. Methods. Illumina expression microarrays were used to assess gene expression levels, and Illumina single- nucleotide polymorphism arrays were used to identify copy number variants in benign, atypical, and malignant me- ningiomas (19 tumors, including 4 malignant ones). The authors also reanalyzed 2 expression data sets generated on Affymetrix microarrays (n = 68, including 6 malignant ones; n = 56, including 3 malignant ones). -

Genetic Factors in Nonsyndromic Orofacial Clefts
Published online: 2021-02-12 THIEME Review Article 101 Genetic Factors in Nonsyndromic Orofacial Clefts Mahamad Irfanulla Khan1 Prashanth C.S.2 Narasimha Murthy Srinath3 1 Department of Orthodontics & Dentofacial Orthopedics, The Oxford Address for correspondence Mahamad Irfanulla Khan, BDS, MDS, Dental College, Bangalore, Karnataka, India Department of Orthodontics & Dentofacial Orthopedics, The Oxford 2 Department of Orthodontics & Dentofacial Orthopedics, DAPM R.V. Dental College, Bangalore, Karnataka 560068, India Dental College, Bangalore, Karnataka, India (e-mail: [email protected]). 3 Department of Oral & Maxillofacial Surgery, Krishnadevaraya College of Dental Sciences, Bangalore, Karnataka, India Global Med Genet 2020;7:101–108. Abstract Orofacial clefts (OFCs) are the most common congenital birth defects in humans and immediately recognized at birth. The etiology remains complex and poorly understood and seems to result from multiple genetic and environmental factors along with gene– environment interactions. It can be classified into syndromic (30%) and nonsyndromic (70%) clefts. Nonsyndromic OFCs include clefts without any additional physical or Keywords cognitive deficits. Recently, various genetic approaches, such as genome-wide associ- ► orofacial clefts ation studies (GWAS), candidate gene association studies, and linkage analysis, have ► nonsyndromic identified multiple genes involved in the etiology of OFCs. ► genetics This article provides an insight into the multiple genes involved in the etiology of OFCs. ► gene mutation Identification of specific genetic causes of clefts helps in a better understanding of the ► genome-wide molecular pathogenesis of OFC. In the near future, it helps to provide a more accurate association study diagnosis, genetic counseling, personalized medicine for better clinical care, and ► linkage analysis prevention of OFCs. -
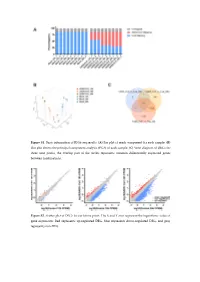
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Caracterización De Complejos CDK-Ciclina Atípicos Humanos Eva Quandt Herrera
Caracterización de complejos CDK-Ciclina atípicos humanos Eva Quandt Herrera ADVERTIMENT. La consulta d’aquesta tesi queda condicionada a l’acceptació de les següents condicions d'ús: La difusió d’aquesta tesi per mitjà del servei TDX (www.tesisenxarxa.net) ha estat autoritzada pels titulars dels drets de propietat intel·lectual únicament per a usos privats emmarcats en activitats d’investigació i docència. No s’autoritza la seva reproducció amb finalitats de lucre ni la seva difusió i posada a disposició des d’un lloc aliè al servei TDX. No s’autoritza la presentació del seu contingut en una finestra o marc aliè a TDX (framing). Aquesta reserva de drets afecta tant al resum de presentació de la tesi com als seus continguts. En la utilització o cita de parts de la tesi és obligat indicar el nom de la persona autora. ADVERTENCIA. La consulta de esta tesis queda condicionada a la aceptación de las siguientes condiciones de uso: La difusión de esta tesis por medio del servicio TDR (www.tesisenred.net) ha sido autorizada por los titulares de los derechos de propiedad intelectual únicamente para usos privados enmarcados en actividades de investigación y docencia. No se autoriza su reproducción con finalidades de lucro ni su difusión y puesta a disposición desde un sitio ajeno al servicio TDR. No se autoriza la presentación de su contenido en una ventana o marco ajeno a TDR (framing). Esta reserva de derechos afecta tanto al resumen de presentación de la tesis como a sus contenidos. En la utilización o cita de partes de la tesis es obligado indicar el nombre de la persona autora. -

Integrated Computational Approach to the Analysis of RNA-Seq Data Reveals New Transcriptional Regulators of Psoriasis
OPEN Experimental & Molecular Medicine (2016) 48, e268; doi:10.1038/emm.2016.97 & 2016 KSBMB. All rights reserved 2092-6413/16 www.nature.com/emm ORIGINAL ARTICLE Integrated computational approach to the analysis of RNA-seq data reveals new transcriptional regulators of psoriasis Alena Zolotarenko1, Evgeny Chekalin1, Alexandre Mesentsev1, Ludmila Kiseleva2, Elena Gribanova2, Rohini Mehta3, Ancha Baranova3,4,5,6, Tatiana V Tatarinova6,7,8, Eleonora S Piruzian1 and Sergey Bruskin1,5 Psoriasis is a common inflammatory skin disease with complex etiology and chronic progression. To provide novel insights into the regulatory molecular mechanisms of the disease, we performed RNA sequencing analysis of 14 pairs of skin samples collected from patients with psoriasis. Subsequent pathway analysis and extraction of the transcriptional regulators governing psoriasis-associated pathways was executed using a combination of the MetaCore Interactome enrichment tool and the cisExpress algorithm, followed by comparison to a set of previously described psoriasis response elements. A comparative approach allowed us to identify 42 core transcriptional regulators of the disease associated with inflammation (NFκB, IRF9, JUN, FOS, SRF), the activity of T cells in psoriatic lesions (STAT6, FOXP3, NFATC2, GATA3, TCF7, RUNX1), the hyper- proliferation and migration of keratinocytes (JUN, FOS, NFIB, TFAP2A, TFAP2C) and lipid metabolism (TFAP2, RARA, VDR). In addition to the core regulators, we identified 38 transcription factors previously not associated with the disease that can clarify the pathogenesis of psoriasis. To illustrate these findings, we analyzed the regulatory role of one of the identified transcription factors (TFs), FOXA1. Using ChIP-seq and RNA-seq data, we concluded that the atypical expression of the FOXA1 TF is an important player in the disease as it inhibits the maturation of naive T cells into the (CD4+FOXA1+CD47+CD69+PD-L1(hi) FOXP3 − ) regulatory T cell subpopulation, therefore contributing to the development of psoriatic skin lesions. -

ID AKI Vs Control Fold Change P Value Symbol Entrez Gene Name *In
ID AKI vs control P value Symbol Entrez Gene Name *In case of multiple probesets per gene, one with the highest fold change was selected. Fold Change 208083_s_at 7.88 0.000932 ITGB6 integrin, beta 6 202376_at 6.12 0.000518 SERPINA3 serpin peptidase inhibitor, clade A (alpha-1 antiproteinase, antitrypsin), member 3 1553575_at 5.62 0.0033 MT-ND6 NADH dehydrogenase, subunit 6 (complex I) 212768_s_at 5.50 0.000896 OLFM4 olfactomedin 4 206157_at 5.26 0.00177 PTX3 pentraxin 3, long 212531_at 4.26 0.00405 LCN2 lipocalin 2 215646_s_at 4.13 0.00408 VCAN versican 202018_s_at 4.12 0.0318 LTF lactotransferrin 203021_at 4.05 0.0129 SLPI secretory leukocyte peptidase inhibitor 222486_s_at 4.03 0.000329 ADAMTS1 ADAM metallopeptidase with thrombospondin type 1 motif, 1 1552439_s_at 3.82 0.000714 MEGF11 multiple EGF-like-domains 11 210602_s_at 3.74 0.000408 CDH6 cadherin 6, type 2, K-cadherin (fetal kidney) 229947_at 3.62 0.00843 PI15 peptidase inhibitor 15 204006_s_at 3.39 0.00241 FCGR3A Fc fragment of IgG, low affinity IIIa, receptor (CD16a) 202238_s_at 3.29 0.00492 NNMT nicotinamide N-methyltransferase 202917_s_at 3.20 0.00369 S100A8 S100 calcium binding protein A8 215223_s_at 3.17 0.000516 SOD2 superoxide dismutase 2, mitochondrial 204627_s_at 3.04 0.00619 ITGB3 integrin, beta 3 (platelet glycoprotein IIIa, antigen CD61) 223217_s_at 2.99 0.00397 NFKBIZ nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, zeta 231067_s_at 2.97 0.00681 AKAP12 A kinase (PRKA) anchor protein 12 224917_at 2.94 0.00256 VMP1/ mir-21likely ortholog -

Human-Specific Tandem Repeat Expansion and Differential Gene Expression During Primate Evolution
Human-specific tandem repeat expansion and differential gene expression during primate evolution Arvis Sulovaria, Ruiyang Lia, Peter A. Audanoa, David Porubskya, Mitchell R. Vollgera, Glennis A. Logsdona, Human Genome Structural Variation Consortium1, Wesley C. Warrenb, Alex A. Pollenc, Mark J. P. Chaissona,d, and Evan E. Eichlera,e,2 aDepartment of Genome Sciences, University of Washington School of Medicine, Seattle, WA 98195; bBond Life Sciences Center, University of Missouri, Columbia, MO 65201; cDepartment of Neurology, University of California, San Francisco, CA 94143; dQuantitative and Computational Biology, University of Southern California, Los Angeles, CA 90089; and eHoward Hughes Medical Institute, University of Washington, Seattle, WA 98195 Edited by Stephen T. Warren, Emory University School of Medicine, Atlanta, GA, and approved October 1, 2019 (received for review July 17, 2019) Short tandem repeats (STRs) and variable number tandem repeats Despite their established importance in population genetics (VNTRs) are important sources of natural and disease-causing and disease association, tandem repeats, particularly VNTRs, variation, yet they have been problematic to resolve in reference are among the least characterized forms of genetic variation in genomes and genotype with short-read technology. We created a the human genome (13, 14). Their repetitive nature and some- framework to model the evolution and instability of STRs and VNTRs times extreme GC content make them particularly challenging to in apes. We phased and assembled 3 ape genomes (chimpanzee, sequence and assemble with standard whole-genome shotgun gorilla, and orangutan) using long-read and 10x Genomics linked- sequencing assembly strategies, including next generation se- read sequence data for 21,442 human tandem repeats discovered in quencing approaches that depend on bridge amplification (15). -

Identification of Shared and Unique Gene Families Associated with Oral
International Journal of Oral Science (2017) 9, 104–109 OPEN www.nature.com/ijos ORIGINAL ARTICLE Identification of shared and unique gene families associated with oral clefts Noriko Funato and Masataka Nakamura Oral clefts, the most frequent congenital birth defects in humans, are multifactorial disorders caused by genetic and environmental factors. Epidemiological studies point to different etiologies underlying the oral cleft phenotypes, cleft lip (CL), CL and/or palate (CL/P) and cleft palate (CP). More than 350 genes have syndromic and/or nonsyndromic oral cleft associations in humans. Although genes related to genetic disorders associated with oral cleft phenotypes are known, a gap between detecting these associations and interpretation of their biological importance has remained. Here, using a gene ontology analysis approach, we grouped these candidate genes on the basis of different functional categories to gain insight into the genetic etiology of oral clefts. We identified different genetic profiles and found correlations between the functions of gene products and oral cleft phenotypes. Our results indicate inherent differences in the genetic etiologies that underlie oral cleft phenotypes and support epidemiological evidence that genes associated with CL/P are both developmentally and genetically different from CP only, incomplete CP, and submucous CP. The epidemiological differences among cleft phenotypes may reflect differences in the underlying genetic causes. Understanding the different causative etiologies of oral clefts is