Clinical Policy: Aripiprazole Long-Acting Injections (Abilify
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Original Research Published, Reproduced, Transmitted, Modified, Posted, Sold, Licensed, Or Used for Commercial Purposes
This work may not be copied, distributed, displayed, Original Research published, reproduced, transmitted, modified, posted, sold, licensed, or used for commercial purposes. By downloading this file, you are agreeing to the Efficacy and Effectiveness of Depot publisher’s Terms & Conditions. Versus Oral Antipsychotics in Schizophrenia: Synthesizing Results Across Different Research Designs Noam Y. Kirson, PhD; Peter J. Weiden, MD; Sander Yermakov, MS; Wayne Huang, MPP; Thomas Samuelson, BA; Steve J. Offord, PhD; Paul E. Greenberg, MS, MA; and Bruce J. O. Wong, MD ABSTRACT he cornerstone of long-term maintenance therapy Objective: Nonadherence is a major challenge in schizophrenia Tof schizophrenia patients is relapse prevention. treatment. While long-acting (depot) antipsychotic medications are Relapse prevention is necessary—albeit not sufficient— often recommended to address adherence problems, evidence on for eventual successful rehabilitation.1 In practice, the the comparative effectiveness of depot versus oral antipsychotics is effectiveness of maintenance antipsychotic treatment is inconsistent. We hypothesize that this inconsistency could be due to often undermined by poor adherence to therapy. Not systematic differences in study design. This review evaluates the effect only is nonadherence the single greatest modifiable of study design on the comparative effectiveness of antipsychotic risk factor for relapse,2,3 it is also often undetected, formulations. The optimal use of different antipsychotic formulations resulting in lost opportunities to employ psychosocial in a general clinical setting depends on better understanding of the underlying reasons for differences in effectiveness across research interventions for adherence, as well as uncertainty as designs. to the relative contribution of lack of efficacy versus Data Sources: A PubMed literature review targeted English-language adherence problems to poor outcomes. -

When Clozapine Is Not Tolerated Mitchell J
University of North Dakota UND Scholarly Commons Nursing Capstones Department of Nursing 10-28-2018 When Clozapine is Not Tolerated Mitchell J. Relf Follow this and additional works at: https://commons.und.edu/nurs-capstones Recommended Citation Relf, Mitchell J., "When Clozapine is Not Tolerated" (2018). Nursing Capstones. 268. https://commons.und.edu/nurs-capstones/268 This Independent Study is brought to you for free and open access by the Department of Nursing at UND Scholarly Commons. It has been accepted for inclusion in Nursing Capstones by an authorized administrator of UND Scholarly Commons. For more information, please contact [email protected]. Running head: WHEN CLOZAPINE IS NOT TOLERATED 1 WHEN CLOZAPINE IS NOT TOLERATED by Mitchell J. Relf Masters of Science in Nursing, University of North Dakota, 2018 An Independent Study Submitted to the Graduate Faculty of the University of North Dakota in partial fulfillment of the requirements for the degree of Master of Science Grand Forks, North Dakota December 2018 WHEN CLOZAPINE IS NOT TOLERATED 2 PERMISSION Title When Clozapine is Not Tolerated Department Nursing Degree Master of Science In presenting this independent study in partial fulfillment of the requirements for a graduate degree from the University of North Dakota, I agree that the College of Nursing and Professional Disciplines of this University shall make it freely available for inspection. I further agree that permission for extensive copying or electronic access for scholarly purposes may be granted by the professor who supervised my independent study work or, in her absence, by the chairperson of the department or the dean of the School of Graduate Studies. -

Treatment of Psychosis: 30 Years of Progress
Journal of Clinical Pharmacy and Therapeutics (2006) 31, 523–534 REVIEW ARTICLE Treatment of psychosis: 30 years of progress I. R. De Oliveira* MD PhD andM.F.Juruena MD *Department of Neuropsychiatry, School of Medicine, Federal University of Bahia, Salvador, BA, Brazil and Department of Psychological Medicine, Institute of Psychiatry, King’s College, University of London, London, UK phrenia. Thirty years ago, psychiatrists had few SUMMARY neuroleptics available to them. These were all Background: Thirty years ago, psychiatrists had compounds, today known as conventional anti- only a few choices of old neuroleptics available to psychotics, and all were liable to cause severe extra them, currently defined as conventional or typical pyramidal side-effects (EPS). Nowadays, new antipsychotics, as a result schizophrenics had to treatments are more ambitious, aiming not only to suffer the severe extra pyramidal side effects. improve psychotic symptoms, but also quality of Nowadays, new treatments are more ambitious, life and social reinsertion. We briefly but critically aiming not only to improve psychotic symptoms, outline the advances in diagnosis and treatment but also quality of life and social reinsertion. Our of schizophrenia, from the mid 1970s up to the objective is to briefly but critically review the present. advances in the treatment of schizophrenia with antipsychotics in the past 30 years. We conclude DIAGNOSIS OF SCHIZOPHRENIA that conventional antipsychotics still have a place when just the cost of treatment, a key factor in Up until the early 1970s, schizophrenia diagnoses poor regions, is considered. The atypical anti- remained debatable. The lack of uniform diagnostic psychotic drugs are a class of agents that have criteria led to relative rates of schizophrenia being become the most widely used to treat a variety of very different, for example, in New York and psychoses because of their superiority with London, as demonstrated in an important study regard to extra pyramidal symptoms. -

Lumateperone Monograph
Lumateperone (Caplyta®) Classification Atypical antipsychotic Pharmacology The mechanism of action of lumateperone tosylate (Caplyta, ITI-007) in the treatment of schizophrenia is unknown but it’s thought to simultaneously modulate serotonin, dopamine, and glutamate neurotransmission. Specifically, lumateperone acts as a potent 5-HT2A receptor antagonist, a D2 receptor presynaptic partial agonist and postsynaptic antagonist, a D1 receptor-dependent modulator of glutamate, and a serotonin reuptake inhibitor. Indication Treatment of schizophrenia in adults Boxed Warning Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death. Caplyta is not approved for the treatment of patients with dementia-related psychosis. Pharmacokinetics Pharmacokinetic Parameter Details Absorption Rapidly absorbed. Absolute bioavailability is about 4.4%. Cmax reached approximately 1 (fasting) to 2 h (food) post dosing. Ingestion of high fat meal lowers mean Cmax by 33% and increases mean AUC by 9% Distribution Protein binding = 97.4%. Volume of distribution (IV) = 4.1 L/kg Metabolism Extensively metabolized. T1/2 = 13-21 hours for lumateperone and metabolites Excretion Less than 1% excreted unchanged in urine 1 Texas Health and Human Services ● hhs.texas.gov Dosage/Administration 42 mg by mouth once daily with food. Dose titration is not required. Use in Special Population Pregnancy Neonates exposed to antipsychotic drugs during the third trimester are at risk for extrapyramidal and/or withdrawal symptoms following delivery. Available data from Caplyta use in pregnant women are insufficient to establish any drug associated risks for birth defects, miscarriage, or adverse maternal or fetal outcomes. There are risks to the mother associated with untreated schizophrenia and with exposure to antipsychotics, including Caplyta, during pregnancy. -

Papers and Originals
BRITISH MEDICAL JOURNAL 19 FEBRUARY 1972 463 PAPERS AND ORIGINALS Drug Interaction: Inhibitory Effect of Neuroleptics on Metabolism of Tricyclic Antidepressants in Man LARS F. GRAM, KERSTIN FREDRICSON OVERO British Medical Journal, 1972, 1, 463-465 excretion (Anders, 1971). There are relatively few reports on interaction concerning neuroleptics or tricyclic antidepressants. Summary Chlorpromazine has both stimulatory and inhibitory effects on the metabolism of hexobarbitone in Total urinary excretion of radioactivity after oral or experimental animals of a test of (Riimke and Bout, 1960-1). Furthermore, chlorpromazine intravenous administration dose '4C-imi- accelerates the metabolism pramine was measured in were tested ofmeprobamate (Kato and Vassanelli, eight patients. They 1962). Tricyclic antidepressants have been shown to inhibit the before, during, and after treatment with neuroleptics. metabolism Excretion diminished while the were of tremorine and oxotremorine in rats in vitro and patients being in vivo (Hammer and Sjoqvist, 1967). It has also been found treated with perphenazine, haloperidol, or chlorproma- that zine, not treatment. desipramine hydrochloride inhibits the metabolism of though during flupenthixol amphetamine in isolated, perfused rat liver (Dingell and Bass, Total urinary excretion of radioactivity and plasma 1969). Pretreatment levels of metabolites and were measured with phenobarbitone decreased steady-state unchanged drug plasma levels of desipramine and nortriptyline in man (Sjoqvist in five patients after a test dose of 14C-nortriptyline. Each et was before and al. 1968). These findings were later confirmed in a twin study patient tested again during perphenazine (Alexanderson et al., 1969). Forrest et treatment. In all patients perphenazine treatment caused: al. (1970) found increased (1) decrease of total urinary excretion, (2) decreased urinary excretion of chlorpromazine metabolites when patients plasma level of metabolites, and (3) increased plasma were given additional treatment with phenobarbitone. -

Lurasidone (Latuda) Reference Number: CP.PMN.50 Effective Date: 09.01.15 Last Review Date: 02.21 Line of Business: Commercial, HIM, Medicaid Revision Log
Clinical Policy: Lurasidone (Latuda) Reference Number: CP.PMN.50 Effective Date: 09.01.15 Last Review Date: 02.21 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description Lurasidone (Latuda®) is an atypical antipsychotic. FDA Approved Indication(s) Latuda is indicated for the treatment of: • Schizophrenia in adults and adolescents (13 to 17 years) • Depressive episode associated with bipolar I disorder (bipolar depression) in adults and pediatric patients (10 to 17 years) as monotherapy • Depressive episode associated with bipolar I disorder (bipolar depression) in adults as adjunctive therapy with lithium or valproate Policy/Criteria Provider must submit documentation (such as office chart notes, lab results or other clinical information) supporting that member has met all approval criteria. It is the policy of health plans affiliated with Centene Corporation® that Latuda is medically necessary when the following criteria are met: I. Initial Approval Criteria A. Bipolar Disorder (must meet all): 1. Diagnosis of bipolar disorder; 2. Age ≥ 10 years; 3. Failure of two preferred atypical antipsychotics (e.g., aripiprazole, ziprasidone, quetiapine, risperidone, or olanzapine) at up to maximally indicated doses, each used for ≥ 4 weeks, unless all are contraindicated or clinically significant adverse effects are experienced; 4. Dose does not exceed 120 mg per day for adults and 80 mg per day for pediatric patients. Approval duration: Medicaid/HIM – 12 months Commercial – Length of Benefit B. Schizophrenia (must meet all): 1. Diagnosis of schizophrenia; 2. Age ≥ 13 years; 3. Failure of two preferred atypical antipsychotics (e.g., aripiprazole, ziprasidone, quetiapine, risperidone, or olanzapine) at up to maximally indicated doses, each used Page 1 of 6 CLINICAL POLICY Lurasidone for ≥ 4 weeks, unless all are contraindicated or clinically significant adverse effects are experienced; 4. -

Atypical Antipsychotics TCO 02.2018
Therapeutic Class Overview Atypical Antipsychotics INTRODUCTION • Antipsychotic medications have been used for over 50 years to treat schizophrenia and a variety of other psychiatric disorders (Miyamato et al 2005). • Antipsychotic medications generally exert their effect in part by blocking dopamine (D)-2 receptors (Jibson et al 2017). • Antipsychotics are divided into 2 distinct classes based on their affinity for D2 and other neuroreceptors: typical antipsychotics, also called first-generation antipsychotics (FGAs), and atypical antipsychotics, also called second- generation antipsychotics (SGAs) (Miyamato et al 2005). • Atypical antipsychotics do not have a uniform pharmacology or mechanism of action; these differences likely account for the different safety and tolerability profiles of these agents (Clinical Pharmacology 2020, Jibson et al 2017). The atypical antipsychotics differ from the early antipsychotics in that they have affinity for the serotonin 5-HT2 receptor in addition to D2. Clozapine is an antagonist at all dopamine receptors (D1-5), with lower affinity for D1 and D2 receptors and high affinity for D4 receptors. Aripiprazole and brexpiprazole act as partial agonists at the D2 receptor, functioning as an ○ agonist when synaptic dopamine levels are low and as an antagonist when they are high. Cariprazine is a partial agonist at D2 and D3. Pimavanserin does not have dopamine blocking activity and is primarily an inverse agonist at 5-HT2A receptors. The remaining atypical antipsychotics share the similarity of D2 and 5-HT2A -
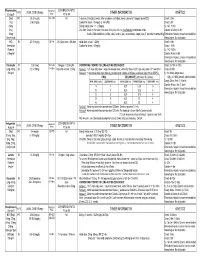
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients Research Questions: 1. How many schizophrenia patients are prescribed recommended first-line second-generation treatments for schizophrenia? 2. How many schizophrenia patients switch to an injectable antipsychotic after stabilization on an oral antipsychotic? 3. How many schizophrenia patients are prescribed 2 or more concomitant antipsychotics? 4. Are claims for long-acting injectable antipsychotics primarily billed as pharmacy or physician administered claims? 5. Does adherence to antipsychotic therapy differ between patients with claims for different routes of administration (oral vs. long-acting injectable)? Conclusions: In total, 4663 schizophrenia patients met inclusion criteria, and approximately 14% of patients (n=685) were identified as treatment naïve without claims for antipsychotics in the year before their first antipsychotic prescription. Approximately 45% of patients identified as treatment naïve had a history of remote antipsychotic use, but it is unclear if antipsychotics were historically prescribed for schizophrenia. Oral second-generation antipsychotics which are recommended as first-line treatment in the MHCAG schizophrenia algorithm were prescribed as initial treatment in 37% of treatment naive patients and 28% of all schizophrenia patients. Recommended agents include risperidone, paliperidone, and aripiprazole. Utilization of parenteral antipsychotics was limited in patients with schizophrenia. Overall only 8% of patients switched from an oral to an injectable therapy within 6 months of their first claim. Approximately, 60% of all schizophrenia patients (n=2512) had claims for a single antipsychotic for at least 12 continuous weeks and may be eligible to transition to a long-acting injectable antipsychotic. -

Schizophrenia Care Guide
August 2015 CCHCS/DHCS Care Guide: Schizophrenia SUMMARY DECISION SUPPORT PATIENT EDUCATION/SELF MANAGEMENT GOALS ALERTS Minimize frequency and severity of psychotic episodes Suicidal ideation or gestures Encourage medication adherence Abnormal movements Manage medication side effects Delusions Monitor as clinically appropriate Neuroleptic Malignant Syndrome Danger to self or others DIAGNOSTIC CRITERIA/EVALUATION (PER DSM V) 1. Rule out delirium or other medical illnesses mimicking schizophrenia (see page 5), medications or drugs of abuse causing psychosis (see page 6), other mental illness causes of psychosis, e.g., Bipolar Mania or Depression, Major Depression, PTSD, borderline personality disorder (see page 4). Ideas in patients (even odd ideas) that we disagree with can be learned and are therefore not necessarily signs of schizophrenia. Schizophrenia is a world-wide phenomenon that can occur in cultures with widely differing ideas. 2. Diagnosis is made based on the following: (Criteria A and B must be met) A. Two of the following symptoms/signs must be present over much of at least one month (unless treated), with a significant impact on social or occupational functioning, over at least a 6-month period of time: Delusions, Hallucinations, Disorganized Speech, Negative symptoms (social withdrawal, poverty of thought, etc.), severely disorganized or catatonic behavior. B. At least one of the symptoms/signs should be Delusions, Hallucinations, or Disorganized Speech. TREATMENT OPTIONS MEDICATIONS Informed consent for psychotropic -

Revision of Precautions Asenapine Maleate, Aripiprazole, Olanzapine
Published by Translated by Ministry of Health, Labour and Welfare Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material to provide convenience for users. In the event of inconsistency between the Japanese original and this English translation, the former shall prevail. Revision of Precautions Asenapine maleate, aripiprazole, olanzapine, quetiapine fumarate, clocapramine hydrochloride hydrate, chlorpromazine hydrochloride, chlorpromazine hydrochloride/promethazine hydrochloride/phenobarbital, chlorpromazine phenolphthalinate, spiperone, zotepine, timiperone, haloperidol, paliperidone, pipamperone hydrochloride, fluphenazine decanoate, fluphenazine maleate, brexpiprazole, prochlorperazine maleate, prochlorperazine mesilate, Pharmaceuticals and Medical Devices Agency Office of Safety I 3-3-2 Kasumigaseki, Chiyoda-ku, Tokyo 100-0013 Japan E-mail: [email protected] Published by Translated by Ministry of Health, Labour and Welfare Pharmaceuticals and Medical Devices Agency This English version is intended to be a reference material to provide convenience for users. In the event of inconsistency between the Japanese original and this English translation, the former shall prevail. propericiazine, bromperidol, perphenazine, perphenazine hydrochloride, perphenazine fendizoate, perphenazine maleate, perospirone hydrochloride hydrate, mosapramine hydrochloride, risperidone (oral drug), levomepromazine hydrochloride, levomepromazine maleate March 27, 2018 Non-proprietary name Asenapine maleate, -

Neuroprotection by Chlorpromazine and Promethazine in Severe Transient and Permanent Ischemic Stroke
Mol Neurobiol (2017) 54:8140–8150 DOI 10.1007/s12035-016-0280-x Neuroprotection by Chlorpromazine and Promethazine in Severe Transient and Permanent Ischemic Stroke Xiaokun Geng1,2 & Fengwu Li1 & James Yip2 & Changya Peng2 & Omar Elmadhoun2 & Jiamei Shen1 & Xunming Ji1,3 & Yuchuan Ding1,2 Received: 29 June 2016 /Accepted: 31 October 2016 /Published online: 28 November 2016 # Springer Science+Business Media New York 2016 Abstract Previous studies have demonstrated depressive or enhance C + P-induced neuroprotection. C + P therapy im- hibernation-like roles of phenothiazine neuroleptics [com- proved brain metabolism as determined by increased ATP bined chlorpromazine and promethazine (C + P)] in brain levels and NADH activity, as well as decreased ROS produc- activity. This ischemic stroke study aimed to establish neuro- tion. These therapeutic effects were associated with alterations protection by reducing oxidative stress and improving brain in PKC-δ and Akt protein expression. C + P treatments con- metabolism with post-ischemic C + P administration. ferred neuroprotection in severe stroke models by suppressing Sprague-Dawley rats were subjected to transient (2 or 4 h) the damaging cascade of metabolic events, most likely inde- middle cerebral artery occlusion (MCAO) followed by 6 or pendent of drug-induced hypothermia. These findings further 24 h reperfusion, or permanent (28 h) MCAO without reper- prove the clinical potential for C + P treatment and may direct fusion. At 2 h after ischemia onset, rats received either an us closer towards the development of an efficacious neuropro- intraperitoneal (IP) injection of saline or two doses of C + P. tective therapy. Body temperatures, brain infarct volumes, and neurological deficits were examined.