Ferrous Iron- and Ammonium-Rich Diffuse Vents Support Habitat-Specific
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

AEOLIAN ISLANDS STROMBOLI Day One: Stromboli
AEOLIAN ISLANDS STROMBOLI Day One: Stromboli Stromboli is the northernmost island of the archipelago and the most spectacular for its continuously active volcano. On the way, you will stop for a nice swim in the clear blue water. At sunset the lava from the volcano glowes as it makes its way down to meet the sea with a loud hiss. To watch the eruptions is especially impressive at night Enjoy the volcanic eruptions lighting up the night sky, a fiery display of nature’s incredible power, while you taste the tasteful delicacies of the chef under the stars. PANAREA Day Two/Three: Panarea Morning departure for Panarea. Panerea is tiny, compared to the other islands, but is the most picturesque with its stark white houses and brilliant yellow ginestra bushes. This island is chic and exclusive. By boat is the best way to explore its coastline and enjoy its clear waters that, together with a thriving underwater landscape make this island the best place for snorkeling and diving in the Aeolians. The island’s charm lies not only in its natural beauty but in its unspoiled character and car-free tracks. Most of the beaches are only accessible by sea, like Lisca Bianca, Basiluzzo and Spinazzola. Panerea is also called the "jet-set" island as it is crowded with young celebrities and its nightlife is particularly lively. SALINA Day Four: Salina Salina is a lush island with thick yellow gorse bushes and terraced vineyards. The island is also known for its delicious Malvasia wine, produced in both red and white varieties. -

Journal of Geodynamics Structural Features Of
Journal of Geodynamics 47 (2009) 288–292 Contents lists available at ScienceDirect Journal of Geodynamics journal homepage: http://www.elsevier.com/locate/jog Technical note Structural features of Panarea volcano in the frame of the Aeolian Arc (Italy): Implications for the 2002–2003 unrest Valerio Acocella a,∗, Marco Neri b, Thomas R. Walter c a Dip. Sc. Geologiche Roma Tre. L.S.L. Murialdo 1, 00146 Rome, Italy b Istituto Nazionale di Geofisica e Vulcanologia, Sezione di Catania, Piazza Roma 2, 95123 Catania, Italy c Deutsches GeoForschungsZentrum (GFZ) Potsdam, Dept. Physics of the Earth, Telegrafenberg, 14473 Potsdam, Germany article info abstract Article history: Panarea, characterized by gas unrest in 2002–2003, is the volcanic island with the least constrained Received 8 October 2008 structure in the eastern-central Aeolian Arc (Italy). Based on structural measurements, we define here its Received in revised form 13 January 2009 deformation pattern relative to the Arc. The main deformations are subvertical extension fractures (63% Accepted 13 January 2009 of data), normal faults (25%) and dikes (12%). The mean orientation of the extension fractures and faults is ∼N38◦E, with a mean opening direction of N135◦ ± 8◦, implying extension with a moderate component Keywords: of dextral shear. These data, matched with those available for Stromboli volcano (pure opening) and Arc volcanoes Vulcano, Lipari and Salina volcanoes (predominant dextral motions) along the eastern-central Arc, suggest Extension Unrest a progressive westward rotation of the extension direction and an increase in the dextral shear. The dextral shear turns into compression in the western arc. The recent unrest at Panarea, coeval to that of nearby Stromboli, may also be explained by the structural context, as both volcanoes lie along the portion of the Arc subject to extension. -

Structural Features of Panarea Volcano in the Frame of the Aeolian Arc (Italy): Implications for the 2002–2003 Unrest V
Structural features of Panarea volcano in the frame of the Aeolian Arc (Italy): implications for the 2002–2003 unrest V. Acocella, M. Neri, T. Walter To cite this version: V. Acocella, M. Neri, T. Walter. Structural features of Panarea volcano in the frame of the Aeolian Arc (Italy): implications for the 2002–2003 unrest. Journal of Geodynamics, Elsevier, 2009, 47 (5), pp.288. 10.1016/j.jog.2009.01.004. hal-00518307 HAL Id: hal-00518307 https://hal.archives-ouvertes.fr/hal-00518307 Submitted on 17 Sep 2010 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Accepted Manuscript Title: Structural features of Panarea volcano in the frame of the Aeolian Arc (Italy): implications for the 2002–2003 unrest Authors: V. Acocella, M. Neri, T. Walter PII: S0264-3707(09)00017-9 DOI: doi:10.1016/j.jog.2009.01.004 Reference: GEOD 876 To appear in: Journal of Geodynamics Received date: 8-10-2008 Revised date: 13-1-2009 Accepted date: 13-1-2009 Please cite this article as: Acocella, V., Neri, M., Walter, T., Structural features of Panarea volcano in the frame of the Aeolian Arc (Italy): implications for the 2002–2003 unrest, Journal of Geodynamics (2008), doi:10.1016/j.jog.2009.01.004 This is a PDF file of an unedited manuscript that has been accepted for publication. -

Volume 2, Issue 1
Volume 2: Issue 1 Spring/Summer, 2014 Editor’s Note CONTENTS Dear Members; While it has been barely a year since the Island and Coastal Archaeology Interest Group was first launched, in that short period we have seen a number of exciting developments as membership and Editor’s Note ............. 1 enthusiasm for the organization grows. Membership now stands at more than 1100 individuals. At the annual ICA meeting in Austin in Report on ICA April, it was a standing room only crowd as members came together to discuss the mission and future agenda of the Interest Group. Meeting ..................... 2 Particularly encouraging was the strong attendance by students and junior colleagues, marking the interest of a next generation of Meetings, scholars in island and coastal archaeology. As reported on page 2, the Announcements & lively discussion produced a list of action items for the coming year and led to the establishment of formal Executive and Meeting Calls for Papers ......... 3 Committees. Award & Funding This issue of The Current continues the regular features—Meetings, Announcements, and Calls for Papers; Research Highlights; Recent Announcements ...... 6 Publications—designed to promote the exchange of ideas and information between members. Simon Stoddart provides an update Research on the FRAGSUS project on Malta where remote sensing is being conducted to understand the dynamics of occupation during the 4th Highlights .................. 7 and 3rd millennium B.C. As Sailors and Honda report on page 10, remote sensing and complimentary techniques were key in Recent identifying a submerged alignment off the coast of Kualoa, Hawai'I, Publications............ 14 that represents a probable fish pond enclosure. -

ECO2-7 Cruise Report
CRUISE REPORT ECO2-7 (Panarea Island, Italy) May 21-31, 2013 Authors: Stan Beaubien, Cinzia De Vittor, Annalisa Franzo Citation: Beaubien S.E., De Vittor C, and Franzo A. (2013) Physical, chemical and biological survey at submarine CO 2 vents off Panarea Island (Italy): cruise report ECO2-7. Università di Roma La Sapienza, Roma, Italy / Istituto Nazionale di Oceanografia e Geofisica Sperimentale, Trieste, Italy, 22 pp. Summary 1 Introduction...................................................................................................... 3 2 Objectives ......................................................................................................... 5 3 Summary of Work ............................................................................................. 7 3.1 Participants ............................................................................................... 7 3.2 Narrative ................................................................................................... 7 4 Description of Work........................................................................................... 9 4.1 Water column sampling (Basiluzzo Island) .................................................. 9 4.2 Sediment sampling (Basiluzzo and Panarea Islands)...................................10 4.3 Primary and secondary benthic production (Basiluzzo Island) .....................11 4.4 Dissolved constituents flux (Panarea Island)...............................................12 4.5 pCO 2 probe transect (Bottaro Island)..........................................................14 -

Shallow-Water Geothermal Activity Offshore Panarea
SHALLOW-WATER GEOTHERMAL ACTIVITY OFFSHORE PANAREA, AEOLIAN ISLAND ARC, ITALY by Samantha Pascarelli A thesis submitted to the Faculty and the Board of Trustees of the Colorado School of Mines in partial fulfillment of the requirements for the degree of Master of Science (Geology). Golden, Colorado Date _________________________ Signed: _________________________ Samantha Pascarelli Signed: _________________________ Dr. Thomas Monecke Thesis Advisor Golden, Colorado Date _________________________ Signed: _________________________ Dr. Wendy Bohrson Professor and Department Head Geology and Geological Engineering ii ABSTRACT Panarea island represents a partially emerged arc volcano forming part of the Aeolian volcanic chain in the southern Tyrrhenian Sea. The island rises from the western sector of a submarine platform representing the eroded summit of a large submarine stratovolcano. Seafloor observations on the eastern portion of the submarine platform have been conducted using a remotely operated vehicle. The seafloor survey resulted in the discovery of a new field of hydrothermal venting between the Secca dei Panarelli and Basiluzzo islet that is typified by quiescent gas venting and the discharge of thermal waters at water depths ranging from 60 to 100 m. The geothermal activity is associated with a wide range of seafloor morphological features, many of which are transient in nature and will not likely be preserved in the geological record. This includes widespread anhydrite-gypsum deposits that have been drilling to a maximum penetration of 5 m below seafloor using a lander-type drilling device. The massive to brecciated hydrothermal deposits contain > 80 % anhydrite and gypsum in addition to alteration minerals formed during subseafloor replacement and infiltration of the volcaniclastic host. -

Index of Places ∗
∗ Index of places ∗ For the name of places, the English version has been preferred, when available. Acate, 59 n. 16, 60. Achaia, 13. Acholla, 43. Adriatic/Adriatico, 13-14, 24-25, 26 n. 2, 64 bibl. Aegades/Egadi, 23, 70 n. 1, 72, 75. Aegean/Egeo, 5, 9, 12, 15, 17 bibl., 23. Aegimuros, 25. Aeolian islands/Isole Eolie, 6-8, 14, 73 e nn. 9, 11-12, 74 e n. 17, 75 nn. 21- 22. Africa (Ifriqiya, Canale di), 16 bibl., 24-26, 34-36 bibl., 49-50 bibl., 54, 62, 70 n. 1, 77, 79-80. Agnone (see Casalicchio-Agnone). Agrigento (Akragas), 7, 16 bibl., 59 n. 16, 60, 69 n. 1, 80 n. 48. Aizanoi, 56 n. 11. Akragas (see Agrigento). Albania, 13-14. Alexandria, 24, 59 n. 16, 61. Alicudi (Ericode), 69 n. 1, 71-73. Amalfi, 74, 78. Amorino, 77. Andalus, 76. Aphiona (Kephal(l)onia), 13, 18 bibl. Argos, 13, 19 fig. 1.8. Asia Minor/Asia Minore, 54. Aspis Hill (Argos), 13, 19 fig. 1.8. Aspis/Clupea (see Kelybia). Athens/Atene, 18 bibl., 31, 54. Balearic archipelagos/Baleari, 24. Balkans/Balcani (Aegean B., Adriatic B.), 2, 6, 9, 12-15. 110 Index of places Basiluzzo, 69-70 e n. 1, 72. Birgi, 46. Bithynia/Bitinia, 59 n. 16. Bizacena, 79. Bon (cape), 23-26, 31. Bosnia, 14. Brandenburg, 85. Brochtorff Circle Tomb (Gozo), 11-12, 14, bibl. 17. Bronte, 7-8, 19 fig. 1.3. Buda (Maison), 93. Burnum,56 n. 11. Byzacium, 31. Calabria, 6, 14, 18 bibl., 73 n. 10, 97 bibl. -

Sicily and the Aeolian Islands
SICILY AND THE AEOLIAN ISLANDS The Aeolian Islands are a dramatic series of islands and tiny islets off the north coast of Sicily, all created, and some still being created, by volcanic activity. Sailing from the Sicilian port of Capo D'Orlando, we will visit the islands of Vulcano (where 'volcano' was derived from), Lipari, Salina, Stromboli and Panarea. There will be plenty of time for swimming and exploring the islands, and in the evenings we will explore the restaurants of the ports and villages where we spend the night. Our on board chef will provide some fine lunches using local ingredients and recipes. The cuisine is very important for us and our highly skilled chef is a specialist in the preparation of traditional Sicilian dishes, such ITINERARY as pasta with sardines, fish couscous, busiati (a type of pasta) and many other specialities, served in a comfortable dining Day 1: Capo D’orlando room surrounded by breathtaking scenery. Embark our gulet in Capo D'orlando at 16.30. Welcome cocktail What's included: on board and beginning of the cruise. We depart and sail to Panarea Island. Day 2: Panarea Breakfast on board. We will stop for a swim at the uninhabited volcanic (now extinct) islets of Lisca Bianca and Bottaro, a great place to spot a submarine volcanic formations at few metres of depth. Lunch on board and before we sail around the islets of 01432 507 280 (within UK) [email protected] | small-cruise-ships.com Lisca Nera and Dattilo before arriving in Panarea. We go ashore Canneto. -

Filicudi M Petrolchimichiera IMO II Da 21.600 Dwt Costruita Da Factorias Vulcano Di Vigo Per La Compagnia Armatrice Augusta Due
NAVI VISITATE PER VOI / SHIPS VISITED FOR YOU Filicudi M Petrolchimichiera IMO II da 21.600 dwt costruita da Factorias Vulcano di Vigo per la compagnia armatrice Augusta Due. L’unità, che ha ottenuto la notazione Green Star, è la prima nave in esercizio per la quale sono stati verificati i calcoli per una vita “a fatica”di 30 anni ed è stata noleggiata per un lungo periodo alla ExxonMobil nostro servizio M/t “Filicudi M” In February, a little more than 13 months after the sign- el mese di febbraio, a poco più di 13 mesi dalla firma ing of the contract, the Factorias Vulcano shipyard of del contratto, il cantiere Factorias Vulcano di Vigo ha Vigo delivered to the shipowners Augusta Due (Mednav consegnato alla compagnia armatrice Augusta Due N Group) the m/t “Filicudi M”, an IMO II petro-chemical (Gruppo Mednav) la m/c Filicudi M, unità petrolchimica tanker having an overall cargo capacity of 25,206.8 cubic IMO II avente una capacità di carico complessiva di 25206,8 metres. metri cubi distribuiti in 16 cisterne. The ship, built under RINA surveillance has the Green La nave, costruita sotto la sorveglianza del RINA che ha rila- Star notation, flies the Italian flag and was designed and sciato la notazione Green Star in ottemperanza alle più recen- built in such way as to offer maximum flexibility in the ti normative inerenti la salvaguardia dell’ambiente marino, transport of petroleum products and derivates, having 8 batte bandiera italiana, è costata circa 47 milioni di Euro e, degrees of segregation. -
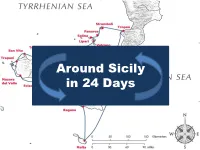
Around Sicily in 24 Days
Around Sicily in 24 Days Bonaire Dufour 385 Dufour 2 3 Onda Blu Jeanneau Sun Odyssey 37.2 Jeanneau Sun Odyssey 4 5 6 Day 1 29th June 2011 • Malta to Siracusa • 82 NM – longest leg • Approx. 13 hrs • Sailed/motor sailed most of the way • Berthed at Marina Yachting at €55 p.n. 7 8 Leaving Marsamxett Harbour … 9 … soon after 10 Day 2 30th June 2011 Siracusa • Caffè del professore • Visited the inviting open air market • Tour of the old town 11 12 13 14 Piazza del Duomo 15 Duomo Chiesa di S. Lucia The burial of Santa Lucia 16 Biography of Michelangelo Merisi da Caravaggio 17 Day 3 1st July 2011 • Siracusa to Riposto • 43 nautical miles • Berthed at Marina di Riposto for €99! • Next time try Giardini Naxos weather permitting 18 19 20 21 Riposto with Etna in the background 22 23 Ristorante Le Botti Day 4 2nd July 2011 • Riposto to Tropea - 70 nautical miles • Wind on the nose, strong current and busy traffic in the Strait • Then calm sea and no wind; sighted dolphins • Berthed at Marina di Tropea for €63 p.n. • High fixed pontoons, so passerella helpful 24 25 Taormina from the sea 26 27 28 Villa San Giovanni to Messina ferry 29 Bonaire in calm sea north of Messina Strait Marina - Tropea30 The hard way up – 200 steps 31 32 33 Cattedrale Cathedral - detail 34 35 Piazza Ercole at night Day 5 3rd July 2011 Tropea • Visited the old town • Beach in the afternoon 36 Typical narrow street 37 38 Piazza Ercole during the day Famous Tropea red onions 39 40 Beautiful beaches and transparent waters 41 42 43 Day 6 4th July 2011 • Tropea to Stromboli • 33 -

Evidence of Active Subsidence at Basiluzzo Island (Aeolian Islands
*Manuscript Click here to view linked References Evidence of active subsidence at Basiluzzo island (Aeolian islands, southern Italy)inferred from a Roman age wharf (a)Marco Anzidei,(a)Alessandra Esposito,(b)Alessandra Benini (a)Istituto Nazionale di Geofisica e Vulcanologia. Via di Vigna Murata 605, Rome, Italy (b)Università della Calabria, Dipartimento di Studi Umanistici, Rende (CS), Italy Abstract The Aeolian Arc (Southern Tyrrhenian Sea, Italy) is one of the most active volcanic areas of the Mediterranean basin, affected by volcanic/hydrothermal and seismic activity. Ancient populations settled this region since historical times, buildingcoastal installations currently used as valuable archaeological indicators of relative sea level changes and vertical land movements. In this study we show and discuss data on the relative sea level change estimated from a submerged wharf of Roman age dated between 50 B.C. and 50 A.D.,located at Basiluzzo Island,. This structurehas been studied through marine surveys and archaeological interpretations and is presently located at a corrected depth of -4.10±0.2 m.We explain this submergence by a cumulative effect of the relative sea level change caused by the regional glacio-hydro-isostatic signal,active since the end of the last glacial maximum,andthe local volcano-tectonic land subsidence.Finally, a total subsidence rate of 2.05±0.1 mm/yr-1, with a volcano-tectonic contribution of 1.43±0.1 mm/yr-1 for the last 2ka BP, is inferred from the comparison against the latest predicted sea level curve for the Southern Tyrrhenian Sea, suggestingnew evaluations of the volcano-tectonic hazard for this area of the Aeolian islands. -

Aeolian Islands) (Italy) Addendum to 1999 Iucn Evaluation
WORLD HERITAGE NOMINATION – IUCN TECHNICAL EVALUATION ISOLE EOLIE (AEOLIAN ISLANDS) (ITALY) ADDENDUM TO 1999 IUCN EVALUATION 1. DOCUMENTATION The above nomination was reviewed by the Bureau at its twenty-third extraordinary session (29 November to 4 December, 1999) which reached the following conclusion: “The Bureau noted that the site has the potential to meet natural criterion (i). The Bureau decided to refer the nomination back to allow the State Party to provide additional information and to address the exclusion of human use areas and to propose more sharply defined boundaries for the Nature Reserves and buffer zones. The volcanic landforms of the site represent classic features in the continuing study of volcanology worldwide. With scientific studies on the site from at least the 18th century, the islands have provided two of the types of eruptions (Vulcanian and Strombolian) to vulcanology and geology textbooks and so have featured prominently in the education of all geoscientists for over 200 years. They continue to provide a rich field for volcanological studies of on-going geological processes in the development of landforms. The area also has a long history of land use, and subsequent abandonment, which has lead to an on-going processes of maquis recovery.” Following the Bureau’s extraordinary meeting the World Heritage Committee, at its twenty-third session noted the examination and, after debate, noted that Italy and 1999 IUCN would clarify the management issues and that the site would be presented to the twenty-fourth Bureau session. Additional information has been received from the Italian authorities in response to this request and is the subject of IUCN’s review below.