European Patent Office
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

5. POTENTIAL for HUMAN EXPOSURE 5.1 OVERVIEW White
WHITE PHOSPHORUS 157 5. POTENTIAL FOR HUMAN EXPOSURE 5.1 OVERVIEW White phosphorus can enter the environment from its production, use, accidental spills during loading and unloading for shipment, and accidental spills during transport. Hazardous wastes sites containing white phosphorus can also be a source of phosphorus in the environment. White phosphorus has been found in at least 77 of the 1,430 current or former EPA National Priorities List (NPL) hazardous waste sites (HazDat 1996). However, the number of sites evaluated for white phosphorus is not known. The frequency of these sites within the United States can be seen in Figure 5-l. The persistence of elemental phosphorus in the air is very short due to oxidation to phosphorus oxides and ultimately to phosphorus acids. However, the particulate phosphorus aerosol may be coated with a protective oxide layer that may prevent further oxidation and extend the lifetime of particulate phosphorus in air. Both wet and dry deposition remove unreacted elemental phosphorus and the degradation products from the air. Similarly, elemental phosphorus oxidizes and hydrolyzes in water and in soil. A small amount of elemental phosphorus is lost from soil and water by volatilization. Phosphorus is used as a fumigant in the storage of grain. Because of ease of application, pellets of aluminum or magnesium phosphide are commonly used (Garry et al. 1993). Phosphine, a highly toxic gas, is generated from phosphide. The rate of formation of phosphine (permissible exposure limit [PEL], 0.4 mg/m3) is dependent on the ambient temperature and humidity. Its release is rapid, and it is extremely fatal to the unprotected person (Garry et al. -

Phosphorous Acid
Phosphorous acid sc-286667 Material Safety Data Sheet Hazard Alert Code EXTREME HIGH MODERATE LOW Key: Section 1 - CHEMICAL PRODUCT AND COMPANY IDENTIFICATION PRODUCT NAME Phosphorous acid STATEMENT OF HAZARDOUS NATURE CONSIDERED A HAZARDOUS SUBSTANCE ACCORDING TO OSHA 29 CFR 1910.1200. NFPA FLAMMABILITY0 HEALTH3 HAZARD INSTABILITY1 SUPPLIER Santa Cruz Biotechnology, Inc. 2145 Delaware Avenue Santa Cruz, California 95060 800.457.3801 or 831.457.3800 EMERGENCY ChemWatch Within the US & Canada: 877-715-9305 Outside the US & Canada: +800 2436 2255 (1-800-CHEMCALL) or call +613 9573 3112 SYNONYMS H3-O3-P, H3PO3, "phosphonic acid", "orthophosphorous acid", "phosphorus trihydroxide", trihydroxyphosphine, "phosphorus acid (sic)", "o-phosphorous acid", "o-phosphorus acid (sic)" Section 2 - HAZARDS IDENTIFICATION CHEMWATCH HAZARD RATINGS Min Max Flammability: 0 Toxicity: 2 Body Contact: 4 Min/Nil=0 Low=1 Reactivity: 1 Moderate=2 High=3 Chronic: 2 Extreme=4 CANADIAN WHMIS SYMBOLS 1 of 9 EMERGENCY OVERVIEW RISK Harmful if swallowed. Causes severe burns. Risk of serious damage to eyes. May cause long-term adverse effects in the aquatic environment. Cumulative effects may result following exposure*. * (limited evidence). POTENTIAL HEALTH EFFECTS ACUTE HEALTH EFFECTS SWALLOWED ■ Accidental ingestion of the material may be harmful; animal experiments indicate that ingestion of less than 150 gram may be fatal or may produce serious damage to the health of the individual. ■ Ingestion of acidic corrosives may produce burns around and in the mouth, the throat and oesophagus. Immediate pain and difficulties in swallowing and speaking may also be evident. ■ As absorption of phosphates from the bowel is poor, poisoning this way is less likely. -
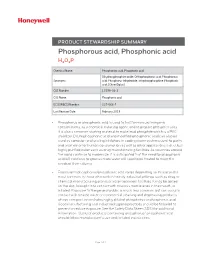
Phosphorous Acid, Phosphonic Acid H3O3P
PRODUCT STEWARDSHIP SUMMARY Phosphorous acid, Phosphonic acid H3O3P Chemical Name: Phosphorous acid, Phosphonic acid Dihydroxyphosphine oxide; Orthophosphorus acid; Phosphorous Synonyms: acid; Phosphorus trihydroxide; Trihydroxyphosphine; Phosphonic acid; [ChemIDplus] CAS Number: 13598-36-2 CAS Name: Phosphonic acid EC (EINECS) Number: 237-066-7 Last Revision Date: February 2019 • Phosphorus or phosphonic acid is used to test for mercury inorganic contaminants, as a chemical reducing agent, and to prepare phosphite salts. It is also a common starting material to make lead phosphite which is a PVC stabilizer. Dilute phosphonic acid and modified phosphonic acid can also be used as corrosion and scaling inhibitors in cooling water systems used to purify and treat water for human consumption as well as other applications that utilize highly purified water such as drug manufacturing facilities. As countries around the world continue to modernize, it is anticipated that the need for phosphonic acid will continue to grow as more water will need to be treated to meet the needs of their citizens. • Exposure to phosphorus/phosphonic acid varies depending on its use and is most common in those who work in heavily industrial settings such as drug or chemical manufacturing plants or water treatment facilities. It may be spilled on the skin, brought into contact with mucous membranes in the mouth, or inhaled. Exposure to the general public is much less common but can occur in contact with treated water or commercial cleaning and degreasing products whose composition includes highly diluted phosphorus or phosphonic acid. Good manufacturing and industrial hygiene practices should be followed to prevent or reduce exposure. -

Phosphorus-Containing Catalyst and Catalytic Cracking Process Utilizing the Same
Patentamt 0 095 364 JEuropâischesê European Patent Office (fî) Publication number: B1 Office européen des brevets ® EUROPEAN PATENT SPECIFICATION (§) Date of publication of patent spécification: 22.10.86 (§) Int.CI.4: B 01 J 29/08, C 10 G 11/05 (â) Application number: 83302938.2 ® Date offiling: 23.05.83 (54) Phosphorus-containing catalyst and catalytic cracking process utilizing the same. (§) Priority: 24.05.82 US 381388 (73) Proprietor: Exxon Research and Engineering Company P.O.Box 390 180 Park Avenue (§) Date of publication of application: Florham Park New Jersey 07932 (US) 30.11.83 Bulletin 83/48 @ Inventor: Pine, Lloyd Albert . (§) Publication of the grant of the patent: 5858 Berkshire Drive 22.10.86 Bulletin 86/43 Bâton Rouge Louisiana (US) Inventor: Cull, Neville Leverne 6260 Rolling Acres Drive (84) Designated Contracting States: Baker Louisiana (US) BE DE FR GB IT NL (74) Representative: Somers, Harold Arnold et al References cited : ESSO Engineering (Europe) Ltd. Patents & EP-A-0 045 61 8 Licences Apex Tower High Street GB-A-1 524 123 New Maiden Surrey KT3 4DJ (GB) US-A-3 595 611 US-A-3 657154 CÛ US-A-3 781 199 US-A-3 867 279 US-A-4036 739 US-A-4044065 US-A-4 182 923 US-A-4198 319 m US-A-4 356 338 o> o o Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. Notice of opposition shall ou be filed in a written reasoned statement. -

Modelling Accidental Releases of Phosphorus in Air
Modelling accidental releases of phosphorus in air Internal report for the RIVM - Centrum Veiligheid Diplomarbeit an der Universität Trier im Studiengang Angewandte Umweltwissenschaften Jan Bremer Prüfer: Prof. Dr.-Ing. Markus Casper, Physische Geographie, Universität Trier Paul Uijt de Haag, Ph.D., Centrum Veiligheid, RIVM Page 1 of 114 Eidesstattliche Erklärung Ich, Jan Bremer, versichere, diese Arbeit selbständig verfasst zu haben und keine anderen Quellen und Hilfsmittel benutzt zu haben. Alle Stellen, die wörtlich oder sinngemäß aus Veröffentlichungen entnommen sind, habe ich als solche kenntlich gemacht. Bilthoven, den ___________________ Unterschrift:_______________________ Page 2 of 114 Colofon © RIVM 2013 Parts of this publication may be reproduced, provided acknowledgement is given to the 'National Institute for Public Health and the Environment', along with the title and year of publication. Jan Sven Bremer, Centrum Veiligheid (CV) Contact: Jan Sven Bremer University of Trier, Faculty of Geography and Geosciences [email protected] This investigation has been performed by order and for the account of the Centrum Veiligheid, within the framework of a diploma thesis in cooperation with the University of Trier under the supervision of Paul Uijt de Haag, Ph.D., and Prof. Dr.-Ing. Markus Casper. Page 3 of 114 Abstract In the Netherlands, the computer model SAFETI-NL has to be applied by companies handling dangerous chemicals to calculated the individual risk (risk at specific location) and societal risk (risk to overall population) of accidental releases of toxic or flammable substances to the atmosphere. One of these dangerous substances is elemental phosphorus (P4). Some characteristics of P4 combustion are not covered by the current model and essential information has to be added in order to improve the modelling of accidental releases of P4. -

Electrical Isolation of Ingaasp Devices by Ion Implantation
VIII-th INTERNATIONAL CONFERENCE on ION IMPLANTATION AND OTHER APPLICATIONS OF IONS AND ELECTRONS June 14-17, 2010 Kazimierz Dolny, Poland Edited by Jerzy Żuk and Janusz Filiks The Market Square in Kazimierz Dolny drawn by Józef Tarłowski Published by Lublin University of Technology Press 20-109 Lublin, 13 Bernardyńska Str. e-mail: [email protected] ISBN 978-83-7497-092-1 Printed by Printing House Ex-libris 20-484 Lublin, 3 Inżynierska Str. 2 CHAIRPERSONS Dariusz Mączka – honorary chairman Jerzy Żuk – chairman, Maria Curie-Skłodowska University Paweł Żukowski – co-chairman, Lublin University of Technology Jerzy Zdanowski – co-chairman, Wrocław University of Technology INTERNATIONAL SCIENTIFIC COMMITTEE V.M. Anishchik (Belarus) A.D. Pogrebnjak (Ukraine) A.V. Belushkin (Russia) L. Pranevičius (Lithuania) R.L. Boxman (Israel) B. Rajchel (Poland) K.P. Homewood (United Kingdom) M. Rubel (Sweden) R. Hurley (Northern Ireland) J. Sielanko (Poland) J. Jagielski (Poland) W. Skorupa (Germany) F.F. Komarov (Belarus) B. Słowiński (Poland) Z.W. Kowalski (Poland) L. Thomé (France) A. Kozanecki (Poland) A. Turos (Poland) D. Machajdík (Slovakia) Z. Werner (Poland) F. Nickel (Germany) K. Wieteska (Poland) J. Piekoszewski (Poland) LOCAL ORGANIZING COMMITTEE Maria Curie-Skłodowska University Lublin University of Technology J. Filiks (Secretary) Cz. Karwat M. Kulik M. Kozak A. Markowski J. Partyka M. Turek P. Węgierek K. Pyszniak A. Droździel 3 4 Monday 14 June Tuesday 15 June Wednesday 16 June Thursday 17 June 7:30 – 8:30 Breakfast 8:30 K. Potzger – Tu-1 Young Scientist Session 8:30 F.F. Komarov – Th-1 9:00 A. Maziewski – Tu-2 Oral presentations 9:00 W. -

Page 1 of 37 Pleasepolymer Do Not Chemistryadjust Margins
Polymer Chemistry Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/polymers Page 1 of 37 PleasePolymer do not Chemistryadjust margins Polymer Chemistry Review Phosphorylation of bio-based compounds: state of the art Nicolas Illy,a,b,* Maxence Fache,c Raphaël Ménard,c Claire Negrell,c Sylvain Caillol,c and Ghislain Received 00th January 20xx, Davidc Accepted 00th January 20xx Over the last few years more and more papers have been devoted to phosphorus-containing DOI: 10.1039/x0xx00000x polymers, mainly due to their fire resistance, excellent chelating and metal-adhesion properties. www.rsc.org/ Nevertheless, sustainability, reduction of environmental impacts and green chemistry are increasingly guiding the development of the next generation of materials. -

Chemistry of Phosphorylated Formaldehyde Derivatives. Part I
Molecules 2014, 19, 12949-13009; doi:10.3390/molecules190912949 OPEN ACCESS molecules ISSN 1420-3049 www.mdpi.com/journal/molecules Review Chemistry of Phosphorylated Formaldehyde Derivatives. Part I Vasily P. Morgalyuk Laboratory of Organophosphorus Compounds, Nesmeyanov Institute of Organoelement Compounds, Russian Academy of Sciences, Vavilova str., 28, Moscow 119991, Russia; E-Mail: [email protected]; Tel.: +7-499-135-92-50; Fax: +7-495-135-65-49 Received: 8 July 2014; in revised form: 8 August 2014 / Accepted: 15 August 2014 / Published: 25 August 2014 Abstract: The underinvestigated derivatives of unstable phosphorylated formaldehyde acetals and some of the structurally related compounds, such as thioacetals, aminonitriles, aminomethylphosphinoyl compounds, are considered. Separately considered are halogen aminals of phosphorylated formaldehyde, acetals of phosphorylated formaldehyde of H-phosphinate-type and a phosphorylated gem-diol of formaldehyde. Synthetic methods, chemical properties and examples of practical applications are given. Keywords: phosphorylated acetals; -thioacetals; -aminonitriles; -aminomethylphosphinoyl compounds; -chloro(or bromo)aminals; -gem-dioles; -ketene acetale; H-phosphinate 1. Introduction Among organophosphorus compounds, α-phosphorylated carbonyl compounds stand out by the capacity of cleavage of phosphorus–carbon bond under mild conditions when reacted with nucleophiles [1–5]. The cleavage of phosphorus–carbon bond may proceed spontaneously as well. At the same time, α-oxoalkylphosphinoyl compounds also retain properties inherent in carbonyl compounds, for example, they undergo cross aldol condensation. The least stable compounds among them are phosphorylated formaldehyde derivatives, dialkyl formylphosphonates (1) [6–11] and N,N,N',N'-tetraalkyl formylphosphondiamides (2) [12] (Figure 1), whose existence was even disputed in the first half of the 1970s [13,14]. -

(12) United States Patent (10) Patent No.: US 6,824,584 B2 Young (45) Date of Patent: Nov.30, 2004
USOO6824584B2 (12) United States Patent (10) Patent No.: US 6,824,584 B2 Young (45) Date of Patent: Nov.30, 2004 (54) AMMONIUM PHOSPHATE/PHOSPHITE 5,549,729 A 8/1996 Yamashita FERTILIZER COMPOUND 5,707,418 A 1/1998 Hsu 5,797.976 A 8/1998 Yamashita (76) Inventor: Donald C. Young, 245 Altura Dr., 5. A 3. E. Fullerton, CA (US) 92835 5,865,8702 2 A 2/1999 HsuOW3 (*) Notice: Subject to any disclaimer, the term of this 6,113,665 A : 9/2000 Lovatt patent is extended or adjusted under 35 2002/0129632 A1 9/2002 Sheppardson et al. ......... 7 1/27 U.S.C. 154(b) by 189 days. FOREIGN PATENT DOCUMENTS (21) Appl. No.: 09/898,424 RU 2121990 * 11/1998 WO WO 98.38863 A 11/1998 (22) Filed: Jul. 3, 2001 OTHER PUBLICATIONS (65) Prior Publication Data A.W. Frazier, K.R. Waerstad, Fertilizer Research, vol. 32, US 2003/0061850 A1 Apr. 3, 2003 No. 2, pp. 161-168, Aug. 1992. (51) Int. Cl." .................................................. C05B 9/00 * cited by examiner (52) U.S. Cl. ..................... 71/32; 71/33; 71/34; 71/64.1 (58) Field of Search ................................ 71/32, 33, 34, Primary Examiner-C. Sayala 71/64.1 (74) Attorney, Agent, or Firm-Banner & Witcoff, Ltd. (56) References Cited (57) ABSTRACT U.S. PATENT DOCUMENTS A high concentration, Stable, neutral liquid fertilizer Solution containing ammonium phosphite with a preferred composi 4,491,464 A 1/1985 Ashmead et al. tion of 9.8-34-0 for use as a fertilizer and a method for 4,588.431 A 5/1986 Nakamura et al. -

United States Patent Office Patiented Oct
3,281,502 United States Patent Office Patiented Oct. 25, 1966 2 acid (about 6 mols per mol of acid) to produce a neu PHOSPHORUS AC DiAEOFYDRIN3,281,502 COMPOUNDS tral product sufficiently reactable with polyisocyanates taud E. Peietier, Elmwood Park, and Floy Pelletier, for polyurethane foam production, the foam product is Lockport, E., assignors, by mesne assignments, to Wy not Self-extinguishing or non-inflammable. Using epi andotte Chemicals Corporation, a corporation of Michi chlorohydrin in accordance with the invention to provide gara a halohydrin compound containing both hydroxyl groups No Drawing. Filled May 22, 1959, Ser. No. 814,967 and chlorine Substitutents enables the provision of at 5 Claims. (C. 260-920) least Substantially neutral products which are reactive with polyisocyanates to form non-inflammable and self The present invention relates to halohydrin compounds 10 extinguishing polyurethane foams. produced by reaction of a phosphorus acid with a halogen At least 1 mol of halogen-substituted monoepoxide is substituted aliphatic monoepoxide, these halohydrin com reacted with each mol of phosphorous acid and, in ac pounds possessing various utility, particularly as inter cordance with the preferred practice of the invention, mediates for the production of synthetic resins possess Sufficient total aliphatic monoepoxide (halogen-substitut ing improved resistance to burning and flame propaga 15 ed or not) is reacted with the phosphorus acid to pro tion. The halohydrin compounds of the invention are vide an at least substantially -
Are Phosphorous and Phosphoric Acids Equal Phosphorous Sources for Plant Growth?1 Asha M
HS1010 Are Phosphorous and Phosphoric Acids Equal Phosphorous Sources for Plant Growth?1 Asha M. Brunings, Guodong Liu, Eric H. Simonne, Shouan Zhang, Yuncong Li, and Lawrence E. Datnoff2 Phosphorus (P) is one of the 17 nutrient elements essential represents potassium percentage as K2O. The 2P O5 unit for plant growth and development (nickel is the 17th) used to represent P content in fertilizer is a conventional (Association of American Plant Food Control Officials unit (in reality, there is little or no P in the form of P2O5 in 2005; Bai, Reilly, and Wood 2006). Phosphorus is also a key fertilizer). component in some agrochemicals, such as phosphorous As a nutrient element essential for normal plant growth and acid (H3PO3). Thus, there are two types of P closely as- sociated with crop production. While growers are familiar development, phosphorus is utilized in the fully oxidized with P-containing fertilizers, the abundance of similar and hydrated form, orthophosphate (H3PO4). Plants absorb and utilize either hydrogen phosphate (HPO 2-) and/or di- terms (such as phosphoric acid, H3PO4, and phosphorous 4 hydrogen phosphate (H PO -), depending on the pH of the acid, H3PO3) may create some confusion about the actual 2 4 - 2- content and efficacy of these products. Table 1 lists some growing medium. At pH7.2, both H2PO4 and HPO4 are P-containing compounds used for crop production. Some approximately equal in amount. In fertilizers, P is normally claims found in commercial literature and product descrip- not found in the form of phosphoric acid (H3PO4) unless tions refer to phosphorous acid as a “supplemental fertil- the growth medium is very acidic. -
Nucleophilic Acyl Substitution
Nucleophilic acyl substitution Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products. Contents Reaction mechanism Acidic conditions Basic conditions Reactivity trends Reactions of acyl derivatives Acid halides Thioesters Anhydrides Esters Amides Carboxylic acids See also References External links Reaction mechanism Carbonyl compounds react with nucleophiles via an addition mechanism: the nucleophile attacks the carbonyl carbon, forming a tetrahedral intermediate. This reaction can be accelerated by acidic conditions, which make the carbonyl more electrophilic, or basic conditions, which provide a more anionic and therefore more reactive nucleophile. The tetrahedral intermediate itself can be an alcohol or alkoxide, depending on the pH of the reaction. The tetrahedral intermediate of an acyl compound contains a substituent attached to the central carbon that can act as a leaving group. After the tetrahedral intermediate forms, it collapses, recreating the carbonyl C=O bond and ejecting the leaving group in an elimination reaction. As a result of this two-step addition/elimination process, the nucleophile takes the place of the leaving group on the carbonyl compound by way of an intermediate state that does not contain a carbonyl.