Epilepsy Syndromes in the First Year of Life and Usefulness of Genetic Testing for Precision Therapy
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

1 ILAE Classification & Definition of Epilepsy Syndromes in the Neonate
ILAE Classification & Definition of Epilepsy Syndromes in the Neonate and Infant: Position Statement by the ILAE Task Force on Nosology and Definitions Authors: Sameer M Zuberi1, Elaine Wirrell2, Elissa Yozawitz3, Jo M Wilmshurst4, Nicola Specchio5, Kate Riney6, Ronit Pressler7, Stephane Auvin8, Pauline Samia9, Edouard Hirsch10, O Carter Snead11, Samuel Wiebe12, J Helen Cross13, Paolo Tinuper14,15, Ingrid E Scheffer16, Rima Nabbout17 1. Paediatric Neurosciences Research Group, Royal Hospital for Children & Institute of Health & Wellbeing, University of Glasgow, Member of European Reference Network EpiCARE, Glasgow, UK. 2. Divisions of Child and Adolescent Neurology and Epilepsy, Department of Neurology, Mayo Clinic, Rochester MN, USA. 3. Isabelle Rapin Division of Child Neurology of the Saul R Korey Department of Neurology, Montefiore Medical Center, Bronx, NY USA. 4. Department of Paediatric Neurology, Red Cross War Memorial Children’s Hospital, Neuroscience Institute, University of Cape Town, South Africa. 5. Rare and Complex Epilepsy Unit, Department of Neuroscience, Bambino Gesu’ Children’s Hospital, IRCCS, Member of European Reference Network EpiCARE, Rome, Italy 6. Neurosciences Unit, Queensland Children's Hospital, South Brisbane, Queensland, Australia. Faculty of Medicine, University of Queensland, Queensland, Australia. 7. Clinical Neuroscience, UCL- Great Ormond Street Institute of Child Health, London, UK. Department of Clinical Neurophysiology, Great Ormond Street Hospital for Children NHS Foundation Trust, Member of European Reference Network EpiCARE London, UK 8. Université de Paris, AP-HP, Hôpital Robert-Debré, INSERM NeuroDiderot, DMU Innov-RDB, Neurologie Pédiatrique, Member of European Reference Network EpiCARE, Paris, France. 9. Department of Paediatrics and Child Health, Aga Khan University, East Africa. 1 10. Neurology Epilepsy Unit “Francis Rohmer”, INSERM 1258, FMTS, Strasbourg University, France. -

The Mineralocorticoid Receptor Leads to Increased Expression of EGFR
www.nature.com/scientificreports OPEN The mineralocorticoid receptor leads to increased expression of EGFR and T‑type calcium channels that support HL‑1 cell hypertrophy Katharina Stroedecke1,2, Sandra Meinel1,2, Fritz Markwardt1, Udo Kloeckner1, Nicole Straetz1, Katja Quarch1, Barbara Schreier1, Michael Kopf1, Michael Gekle1 & Claudia Grossmann1* The EGF receptor (EGFR) has been extensively studied in tumor biology and recently a role in cardiovascular pathophysiology was suggested. The mineralocorticoid receptor (MR) is an important efector of the renin–angiotensin–aldosterone‑system and elicits pathophysiological efects in the cardiovascular system; however, the underlying molecular mechanisms are unclear. Our aim was to investigate the importance of EGFR for MR‑mediated cardiovascular pathophysiology because MR is known to induce EGFR expression. We identifed a SNP within the EGFR promoter that modulates MR‑induced EGFR expression. In RNA‑sequencing and qPCR experiments in heart tissue of EGFR KO and WT mice, changes in EGFR abundance led to diferential expression of cardiac ion channels, especially of the T‑type calcium channel CACNA1H. Accordingly, CACNA1H expression was increased in WT mice after in vivo MR activation by aldosterone but not in respective EGFR KO mice. Aldosterone‑ and EGF‑responsiveness of CACNA1H expression was confrmed in HL‑1 cells by Western blot and by measuring peak current density of T‑type calcium channels. Aldosterone‑induced CACNA1H protein expression could be abrogated by the EGFR inhibitor AG1478. Furthermore, inhibition of T‑type calcium channels with mibefradil or ML218 reduced diameter, volume and BNP levels in HL‑1 cells. In conclusion the MR regulates EGFR and CACNA1H expression, which has an efect on HL‑1 cell diameter, and the extent of this regulation seems to depend on the SNP‑216 (G/T) genotype. -

Monogenic Causation in Chronic Kidney Disease
University of Dublin, Trinity College School of Medicine, Department of Medicine Investigation of the monogenic causes of chronic kidney disease PhD Thesis April 2020 Dervla Connaughton Supervisor: Professor Mark Little Co-Supervisors: Professor Friedhelm Hildebrandt and Professor Peter Conlon 1 DECLARATION I declare that this thesis has not been submitted as an exercise for a degree at this or any other university and it is entirely my own work. This work was funding by the Health Research Board, Ireland (HPF-206-674), the International Pediatric Research Foundation Early Investigators’ Exchange Program and the Amgen® Irish Nephrology Society Specialist Registrar Bursary. I agree to deposit this thesis in the University’s open access institutional repository or allow the Library to do so on my behalf, subject to Irish Copyright Legislation and Trinity College Library conditions of use and acknowledgement. I consent to the examiner retaining a copy of the thesis beyond the examining period, should they so wish (EU GDPR May 2018). _____________________ Dervla Connaughton 2 SUMMARY Chapter 1 provides an introduction to the topic while Chapter 2 provides details of the methods employed in this work. In Chapter 3 I provide an overview of the currently known monogenic causes for human chronic kidney disease (CKD). I also describe how next- generation sequencing can facilitate molecular genetic diagnostics in individuals with suspected genetic kidney disease. Chapter 4 details the findings of a multi-centre, cross-sectional study of patients with CKD in the Republic of Ireland. The primary aim of this study (the Irish Kidney Gene Project) was to describe the prevalence of reporting a positive family history of CKD among a representation sample of the CKD population. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Childhood Epilepsy: an Update on Diagnosis and Management
American Journal of Neuroscience Review Childhood Epilepsy: An Update on Diagnosis and Management Khaled Saad Department of Pediatrics, Faculty of Medicine, University of Assiut, Assiut 71516, Egypt Article history Abstract: Within the past few years there is a rapid expansion in our Received: 23-10-2014 understanding of epilepsy. The development of new anti-epileptic drugs and Revised: 25-11-2014 refinements to epilepsy surgery are widening the therapeutic options for Accepted: 12-01-2015 epilepsy. In addition, the classification of the epilepsies continues to evolve based on an increased understanding of the molecular genetics of the condition and this includes the recognition of possible novel epilepsy syndromes. This review considers some of these exciting developments, as well as addressing the essential features of the diagnosis, investigations, management and impact of epilepsy in childhood. Keywords: Children, Epilepsy, Seizure, Anti-Epileptic Drugs Introduction guidelines for optimal practices, rational therapy and counseling (Aylwad, 2008; Tamber and Mountz, 2012; Epilepsy is a common heterogeneous neurological Sharma, 2013). problem in children. It exerts a significant physical, psychological, economic and social toll on children and Definitions their caregivers. Fifty million people have epilepsies A seizure is defined as an excessive burst of abnormal globally; more than half of them are children. In the synchronized neuronal activity affecting small or large USA; between 25,000 and 40,000 children will have a neuronal networks that results in clinical manifestations first non-febrile seizure each year. The problem is that are sudden, transient and usually brief. Epilepsy is further compounded in developing countries as they add defined as a disorder of the brain characterized by any of about 75-80% of new cases of epilepsy (Guerrini, 2006; the following conditions: (1) At least two unprovoked (or Tamber and Mountz, 2012; Sharma, 2013). -
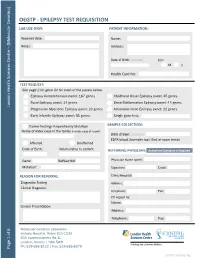
Oegtp - Epilepsy Test Requisition Lab Use Only: Patient Information
OEGTP - EPILEPSY TEST REQUISITION LAB USE ONLY: PATIENT INFORMATION: Received date: Name: Notes: Address: Date of Birth: YY/MM/DD Sex: M F Health Card No: TEST REQUEST: See page 2 for gene list for each of the panels below Epilepsy Comprehensive panel: 167 genes Childhood Onset Epilepsy panel: 45 genes Focal Epilepsy panel: 14 genes Brain Malformation Epilepsy panel: 44 genes London Health Sciences Centre – (Molecular Genetics) London Health Sciences Centre Progressive Myoclonic Epilepsy panel: 20 genes Actionable Gene Epilepsy panel: 22 genes Early Infantile Epilepsy panel: 51 genes Single gene test: Carrier Testing/ KnownFamily Mutation SAMPLE COLLECTION: Name of index case in the family (include copy of report) Date drawn: YY/MM/DD EDTA blood (lavender top) (5ml at room temp) Affected Unaffected Date of Birth: Relationship to patient: REFERRING PHYSICIAN: Authorized Signature is Required Gene: RefSeq:NM Physician Name (print): Mutation: Signature: Email: REASON FOR REFERRAL: Clinic/Hospital: Diagnostic Testing Address: Clinical Diagnosis: Telephone: Fax: CC report to: Name: Clinical Presentation: Address: Telephone: Fax: Molecular Genetics Laboratory Victoria Hospital, Room B10-123A 800 Commissioners Rd. E. London, Ontario | N6A 5W9 Pathology and Laboratory Medicine Ph: 519-685-8122 | Fax: 519-685-8279 Page 1 of 6 Page OEGTP (2021/05/28) OEGTP - EPILEPSY TEST PANELS Patient Identifier: COMPREHENSIVE EPILEPSY PANEL: 167 Genes ACTB, ACTG1, ADSL, AKT3, ALDH7A1, AMT, AP3B2, ARFGEF2, ARHGEF9, ARV1, ARX, ASAH1, ASNS, ATP1A3, ATP6V0A2, ATP7A, -

EEG in the Diagnosis, Classification, and Management of Patients With
EEG IN THE DIAGNOSIS, J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.2005.069245 on 16 June 2005. Downloaded from CLASSIFICATION, AND MANAGEMENT ii2 OF PATIENTS WITH EPILEPSY SJMSmith J Neurol Neurosurg Psychiatry 2005;76(Suppl II):ii2–ii7. doi: 10.1136/jnnp.2005.069245 he human electroencephalogram (EEG) was discovered by the German psychiatrist, Hans Berger, in 1929. Its potential applications in epilepsy rapidly became clear, when Gibbs and Tcolleagues in Boston demonstrated 3 per second spike wave discharge in what was then termed petit mal epilepsy. EEG continues to play a central role in diagnosis and management of patients with seizure disorders—in conjunction with the now remarkable variety of other diagnostic techniques developed over the last 30 or so years—because it is a convenient and relatively inexpensive way to demonstrate the physiological manifestations of abnormal cortical excitability that underlie epilepsy. However, the EEG has a number of limitations. Electrical activity recorded by electrodes placed on the scalp or surface of the brain mostly reflects summation of excitatory and inhibitory postsynaptic potentials in apical dendrites of pyramidal neurons in the more superficial layers of the cortex. Quite large areas of cortex—in the order of a few square centimetres—have to be activated synchronously to generate enough potential for changes to be registered at electrodes placed on the scalp. Propagation of electrical activity along physiological pathways or through volume conduction in extracellular spaces may give a misleading impression as to location of the source of the electrical activity. Cortical generators of the many normal and abnormal cortical activities recorded in the EEG are still largely unknown. -

Causes of Mortality in Early Infantile Epileptic Encephalopathy: a Systematic Review
Epilepsy & Behavior 85 (2018) 32–36 Contents lists available at ScienceDirect Epilepsy & Behavior journal homepage: www.elsevier.com/locate/yebeh Review Causes of mortality in early infantile epileptic encephalopathy: A systematic review Graciane Radaelli a,b, Francisco de Souza Santos b, Wyllians Vendramini Borelli b,LeonardoPisanib, Magda Lahorgue Nunes b,d, Fulvio Alexandre Scorza c,d, Jaderson Costa da Costa b,d,⁎ a Federal University of São Paulo (UNIFESP)/Paulista School of Medicine, São Paulo, Brazil b Brain Institute of Rio Grande do Sul (BraIns), Pontifical Catholic University of Rio Grande do Sul, Porto Alegre, RS, Brazil c Laboratory of Neuroscience, Department of Neurology and Neurosurgery, Federal University of São Paulo, São Paulo, SP, Brazil d CNPq, Brazil article info abstract Article history: Introduction: Early infantile epileptic encephalopathy syndrome (EIEE), also known as Ohtahara syndrome, is an Received 6 March 2018 age-dependent epileptic encephalopathy syndrome defined by clinical features and electroencephalographic Revised 25 April 2018 findings. Epileptic disorders with refractory seizures beginning in the neonatal period and/or early infancy Accepted 5 May 2018 have a potential risk of premature mortality, including sudden death. We aimed to identify the causes of death Available online xxxx in EIEE and conducted a literature survey of fatal outcomes. Methods: We performed a literature search in MEDLINE, EMBASE, and Web of Science for data from inception Keywords: “ ”“ ”“ ”“ ” “ ” Ohtahara syndrome until September 2017. The terms death sudden, unexplained death, SUDEP, lethal, and fatal and the “ ”“ ”“ ”“ Early infantile epileptic syndrome medical subject heading terms epileptic encephalopathy, mortality, death, sudden infant death syn- Suppression burst drome,” and “human” were used in the search strategy. -

Epilepsy in Childhood
Psychogenic Non-epileptic Seizures Dr Katharina Junejo Consultant Child and Adolescent Psychiatrist Aim Overview of basic principles in childhood epilepsy PNES Diagnosis of epilepsy Every child’s brain is capable of generating seizures with certain pro-convulsive drugs, acute metabolic disturbance, CNS infection, acute head trauma Risk of epilepsy after acute and provoked seizure is 3-5% Emotional stress is not considered provoking factor Henri Gastaut and colleagues proposed a classification of seizures: “all attempts at classification of seizures are hampered by our limited knowledge of the underlying pathological processes within the brain and that any classification must of necessity be a tentative one and will be subject to change with every advance in scientific understanding of epilepsy” (Gastaut et al 1964). Epilepsy has variable clinical presentations Convulsions (tonic- Staring spells clonic seizures) (absences) Period with confusion with or without Variants of automatism (complex paroxysmal events partial seizures) during sleep (common frontal lobe seizures) Limb or body jerk movements (myoclonus) Loss of speech/decline in Spasms (infantile or social, cognitive function Falls or drops epileptic spasms) (Landau-Kleffner (atonic but also syndrome) tonic seizures) ILAE Revised Terminology for Organization of Seizures and Epilepsies 2011 ‐ 2013 Classification of seizures: generalised and focal seizures Electroclinical syndromes and other epilepsies grouped by specificity of diagnosis (arranged by typical age of onset) Major conceptual changes: using aetiological classification of epilepsy-genetic, metabolic, immune, infectious and unknown causes Changes in terminology Operational diagnosis of epilepsy: occurrence of 2 or more unprovoked seizures, irrespective of seizure type (as recurrence risk is 80-90%) Careful history is only diagnostic test as many paroxysmal disorders that mimic epileptic seizures (most common vasovagal syncope or reflex anoxic seizure) Limitations of EEG: approx. -

Prognostic Utility of Hypsarrhythmia Scoring in Children with West
Clinical Neurology and Neurosurgery 184 (2019) 105402 Contents lists available at ScienceDirect Clinical Neurology and Neurosurgery journal homepage: www.elsevier.com/locate/clineuro Prognostic utility of hypsarrhythmia scoring in children with West syndrome T after ketogenic diet ⁎ Yunjian Zhang1, Lifei Yu1, Yuanfeng Zhou, Linmei Zhang, Yi Wang, Shuizhen Zhou Department of Pediatric Neurology, Children’s Hospital of Fudan University, China ARTICLE INFO ABSTRACT Keywords: Objective: The aim of this study was to evaluate the clinical efficacy and electroencephalographic (EEG) changes West syndrome of West syndrome after ketogenic diet (KD) therapy and to explore the correlation of EEG features and clinical Ketogenic diet efficacy. EEG Patients and methods: We retrospectively studied 39 patients with West syndrome who accepted KD therapy from Hypsarrhythmia May 2011 to October 2017. Outcomes including clinical efficacy and EEG features with hypsarrhythmia severity scores were analyzed. Results: After 3 months of treatment, 20 patients (51.3%) had ≥50% seizure reduction, including 4 patients (10.3%) who became seizure-free. After 6 months of treatment, 4 patients remained seizure free, 12 (30.8%) had 90–99% seizure reduction, 8 (20.5%) had a reduction of 50–89%, and 15 (38.5%) had < 50% reduction. Hypsarrhythmia scores were significantly decreased at 3 months of KD. They were associated with seizure outcomes at 6 months independent of gender, the course of disease and etiologies. Patients with a hypsar- rhythmia score ≥8 at 3 months of therapy may not be benefited from KD. Conclusion: Our findings suggest a potential benefit of KD for patients with drug-resistant West syndrome. Early change of EEG after KD may be a predictor of a patient’s response to the therapy. -

Clinicians Using the Classification Will Identify a Seizure As Focal Or Generalized Onset If There Is About an 80% Confidence Level About the Type of Onset
GENERALIZED ONSET SEIZURES Generalized onset seizures are not characterized by level of awareness, because awareness is almost always impaired. Generalized tonic-clonic: Immediate loss of Generalized epileptic spasms: Brief seizures with awareness, with stiffening of all limbs (tonic phase), flexion at the trunk and flexion or extension of the followed by sustained rhythmic jerking of limbs and limbs. Video-EEG recording may be required to face (clonic phase). Duration is typically 1 to 3 minutes. determine focal versus generalized onset. The seizure may produce a cry at the start, falling, tongue biting, and incontinence. Generalized typical absence: Sudden onset when activity stops with a brief pause and staring, Generalized clonic: Rhythmical sustained jerking of sometimes with eye fluttering and head nodding or limbs and/or head with no tonic stiffening phase. other automatic behaviors. If it lasts for more than These seizures most often occur in young children. several seconds, awareness and memory are impaired. Recovery is immediate. The EEG during these seizures Generalized tonic: Stiffening of all limbs, without always shows generalized spike-waves. clonic jerking. Generalized atypical absence: Like typical absence Generalized myoclonic: Irregular, unsustained jerking seizures, but may have slower onset and recovery and of limbs, face, eyes, or eyelids. The jerking of more pronounced changes in tone. Atypical absence generalized myoclonus may not always be left-right seizures can be difficult to distinguish from focal synchronous, but it occurs on both sides. impaired awareness seizures, but absence seizures usually recover more quickly and the EEG patterns are Generalized myoclonic-tonic-clonic: This seizure is like different. -

Title in All Caps
Epilepsy Syndromes: Where does Dravet Syndrome fit in? Scott Demarest MD Assistant Professor, Departments of Pediatrics and Neurology University of Colorado School of Medicine Children's Hospital Colorado Disclosures Scott Demarest has consulted for Upsher-Smith on an unrelated subject matter. No conflicts of interest Objectives • What is an Epilepsy Syndrome? • How do we define epilepsy syndromes? • Genetic vs Phenotype (Features) • So what? Why do we care about Epilepsy Syndromes? • How do we organize and categorize Epilepsy Syndromes? • What epilepsy syndromes are similar to Dravet Syndrome and what is different about them? Good Resource International League Against Epilepsy Epilepsydiagnosis.org https://www.epilepsydiagnosis.org/syndrome/epilepsy- syndrome-groupoverview.html What is an Epilepsy Syndrome? A syndrome is a collection of common clinical traits. For Epilepsy this is usually about: • What type of seizures occur? • Age seizure start? Electroclinical • Development? Features or • What does the EEG look like? Phenotype • Other Co-morbidities… Course of an Epilepsy Syndrome Developmental Trajectories - Theoretical Model Normal Previously Normal with Epileptic Encephalopathy Development Never Normal Gray represents Epilepsy Onset the intensity of Age Epilepsy How distinct are Epilepsy Syndromes? A B C Many features might overlap, but the hope is that the cluster of symptoms are “specific” to that epilepsy syndrome…this is often better in theory than practice. How does the individual patient fit? A B C Is this patient at type A,B or C? What about Syndromes Defined by Genes? Is SCN1A the same as Dravet Syndrome? …I don’t have a perfect answer for this… many diseases are being defined by the gene (CDKL5, SCN8A, CHD2).