Variants of the Adiponectin and Adiponectin Receptor 1 Genes and Breast Cancer Risk
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Multifaceted Physiological Roles of Adiponectin in Inflammation And
International Journal of Molecular Sciences Review Multifaceted Physiological Roles of Adiponectin in Inflammation and Diseases Hyung Muk Choi 1, Hari Madhuri Doss 1,2 and Kyoung Soo Kim 1,2,* 1 Department of Clinical Pharmacology and Therapeutics, Kyung Hee University School of Medicine, Seoul 02447, Korea; [email protected] (H.M.C.); [email protected] (H.M.D.) 2 East-West Bone & Joint Disease Research Institute, Kyung Hee University Hospital at Gangdong, Gandong-gu, Seoul 02447, Korea * Correspondence: [email protected]; Tel.: +82-2-961-9619 Received: 3 January 2020; Accepted: 10 February 2020; Published: 12 February 2020 Abstract: Adiponectin is the richest adipokine in human plasma, and it is mainly secreted from white adipose tissue. Adiponectin circulates in blood as high-molecular, middle-molecular, and low-molecular weight isoforms. Numerous studies have demonstrated its insulin-sensitizing, anti-atherogenic, and anti-inflammatory effects. Additionally, decreased serum levels of adiponectin is associated with chronic inflammation of metabolic disorders including Type 2 diabetes, obesity, and atherosclerosis. However, recent studies showed that adiponectin could have pro-inflammatory roles in patients with autoimmune diseases. In particular, its high serum level was positively associated with inflammation severity and pathological progression in rheumatoid arthritis, chronic kidney disease, and inflammatory bowel disease. Thus, adiponectin seems to have both pro-inflammatory and anti-inflammatory effects. This indirectly indicates that adiponectin has different physiological roles according to an isoform and effector tissue. Knowledge on the specific functions of isoforms would help develop potential anti-inflammatory therapeutics to target specific adiponectin isoforms against metabolic disorders and autoimmune diseases. -

Adiporon, an Orally Active, Synthetic Agonist of Adipor1 and Adipor2 Receptors Has Gastroprotective Effect in Experimentally Induced Gastric Ulcers in Mice
molecules Article AdipoRon, an Orally Active, Synthetic Agonist of AdipoR1 and AdipoR2 Receptors Has Gastroprotective Effect in Experimentally Induced Gastric Ulcers in Mice Hubert Zatorski 1,2, Maciej Salaga 1 , Marta Zieli ´nska 1, Kinga Majchrzak 1, Agata Binienda 1 , Radzisław Kordek 3, Ewa Małecka-Panas 2 and Jakub Fichna 1,4,* 1 Department of Biochemistry, Medical University of Lodz, 92-215 Lodz, Poland; [email protected] (H.Z.); [email protected] (M.S.); [email protected] (M.Z.); [email protected] (K.M.); [email protected] (A.B.) 2 Department of Digestive Tract Diseases, Medical University of Lodz, 93-281 Lodz, Poland; [email protected] 3 Department of Pathology, Medical University of Lodz, 92-215 Lodz, Poland; [email protected] 4 Department of Biochemistry, Faculty of Medicine, Medical University of Lodz, Mazowiecka 6/8, 92-215 Lodz, Poland * Correspondence: jakub.fi[email protected]; Tel.: +48-42-272-57-07 Citation: Zatorski, H.; Salaga, M.; Abstract: Introduction: Adiponectin is a hormone secreted by adipocytes, which exhibits insulin- Zieli´nska,M.; Majchrzak, K.; sensitizing and anti-inflammatory properties and acts through adiponectin receptors: AdipoR1 and Binienda, A.; Kordek, R.; AdipoR2. The aim of the study was to evaluate whether activation of adiponectin receptors AdipoR1 Małecka-Panas, E.; Fichna, J. and AdipoR2 with an orally active agonist AdipoRon has gastroprotective effect and to investigate AdipoRon, an Orally Active, the possible underlying mechanism. Methods: We used two well-established mouse models of Synthetic Agonist of AdipoR1 and gastric ulcer (GU) induced by oral administration of EtOH (80% solution in water) or diclofenac AdipoR2 Receptors Has (30 mg/kg, p.o.). -

In the Porcine Pituitary During the Oestrous Cycle Marta Kiezun, Anna Maleszka, Nina Smolinska, Anna Nitkiewicz and Tadeusz Kaminski*
Kiezun et al. Reproductive Biology and Endocrinology 2013, 11:18 http://www.rbej.com/content/11/1/18 RESEARCH Open Access Expression of adiponectin receptors 1 (AdipoR1) and 2 (AdipoR2) in the porcine pituitary during the oestrous cycle Marta Kiezun, Anna Maleszka, Nina Smolinska, Anna Nitkiewicz and Tadeusz Kaminski* Abstract Background: Adiponectin, protein secreted mainly by white adipose tissue, is an important factor linking the regulation of metabolic homeostasis and reproductive processes. The biological activity of the hormone is mediated via two distinct receptors, termed adiponectin receptor 1(AdipoR1) and adiponectin receptor 2 (AdipoR2). The present study analyzed mRNA and protein expression of AdipoR1 and AdipoR2 in the anterior (AP) and posterior (NP) pituitary of cyclic pigs. Methods: The total of 20 animals was assigned to one of four experimental groups (n = 5 per group) as follows: days 2–3 (early-luteal phase), 10–12 (mid-luteal phase), 14–16 (late-luteal phase), 17–19 (follicular phase) of the oestrous cycle. mRNA and protein expression were analyzed using real-time PCR and Western Blot methods, respectively. Results: The lowest AdipoR1 gene expression was detected in AP on days 10–12 relative to days 2–3and14–16 (p < 0.05). In NP, AdipoR1 mRNA levels were elevated on days 10–12 and 14–16 (p < 0.05). AdipoR2 gene expression in AP was the lowest on days 10–12, and an expression peak occurred on days 2–3 (p < 0.05). In NP, the lowest (p < 0.05) expression of AdipoR2 mRNA was noted on days 17–19. The AdipoR1 protein content in AP was the lowest on days 17–19 (p < 0.05), while in NP the variations in protein expression levels during the oestrous cycle were negligible. -

(Title of the Thesis)*
THE PHYSIOLOGICAL ACTIONS OF ADIPONECTIN IN CENTRAL AUTONOMIC NUCLEI: IMPLICATIONS FOR THE INTEGRATIVE CONTROL OF ENERGY HOMEOSTASIS by Ted Donald Hoyda A thesis submitted to the Department of Physiology In conformity with the requirements for the degree of Doctor of Philosophy Queen‟s University Kingston, Ontario, Canada (September, 2009) Copyright © Ted Donald Hoyda, 2009 ABSTRACT Adiponectin regulates feeding behavior, energy expenditure and autonomic function through the activation of two receptors present in nuclei throughout the central nervous system, however much remains unknown about the mechanisms mediating these effects. Here I investigate the actions of adiponectin in autonomic centers of the hypothalamus (the paraventricular nucleus) and brainstem (the nucleus of the solitary tract) through examining molecular, electrical, hormonal and physiological consequences of peptidergic signalling. RT-PCR and in situ hybridization experiments demonstrate the presence of AdipoR1 and AdipoR2 mRNA in the paraventricular nucleus. Investigation of the electrical consequences following receptor activation in the paraventricular nucleus indicates that magnocellular-oxytocin cells are homogeneously inhibited while magnocellular-vasopressin neurons display mixed responses. Single cell RT-PCR analysis shows oxytocin neurons express both receptors while vasopressin neurons express either both receptors or one receptor. Co-expressing oxytocin and vasopressin neurons express neither receptor and are not affected by adiponectin. Median eminence projecting corticotropin releasing hormone neurons, brainstem projecting oxytocin neurons, and thyrotropin releasing hormone neurons are all depolarized by adiponectin. Plasma adrenocorticotropin hormone concentration is increased following intracerebroventricular injections of adiponectin. I demonstrate that the nucleus of the solitary tract, the primary cardiovascular regulation site of the medulla, expresses mRNA for AdipoR1 and AdipoR2 and mediates adiponectin induced hypotension. -

Metabolic Syndrome: Is It Time to Add the Central Nervous System?
nutrients Review Metabolic Syndrome: Is It Time to Add the Central Nervous System? Milagros Rojas 1, Mervin Chávez-Castillo 2, Daniela Pirela 1, Heliana Parra 1 , Manuel Nava 1, Maricarmen Chacín 3, Lissé Angarita 4 , Roberto Añez 5, Juan Salazar 1 , Rina Ortiz 6, Samuel Durán Agüero 7, Marbel Gravini-Donado 8, Valmore Bermúdez 9 and Edgar Díaz-Camargo 9,* 1 Endocrine and Metabolic Diseases Research Center, School of Medicine, University of Zulia, Maracaibo 4004, Venezuela; migarocafi@gmail.com (M.R.); [email protected] (D.P.); [email protected] (H.P.); [email protected] (M.N.); [email protected] (J.S.) 2 Psychiatric Hospital of Maracaibo, Maracaibo 4004, Venezuela; [email protected] 3 Facultad de Ciencias de la Salud, Universidad Simón Bolívar, Barranquilla 08002, Colombia; [email protected] 4 Escuela de Nutrición y Dietética, Facultad de Medicina, Universidad Andrés Bello, Sede Concepción 4260000, Chile; [email protected] 5 Departamento de Endocrinología y Nutrición, Hospital General Universitario Gregorio Marañón, 28007 Madrid, Spain; [email protected] 6 Posgrado, Carrera de Medicina, Universidad Católica de Cuenca, Cantón de Cuenca 010101, Ecuador; [email protected] 7 Facultad de Ciencias Para el Cuidado de la Salud, Universidad San Sebastián, Los Leones 8420524, Chile; [email protected] 8 Facultad de Ciencias Jurídicas y Sociales, Universidad Simón Bolívar, Barranquilla 080002, Colombia; [email protected] 9 Facultad de Ciencias Jurídicas y Sociales, Universidad Simón Bolívar, Cúcuta 540006, Colombia; [email protected] * Correspondence: [email protected] Citation: Rojas, M.; Chávez-Castillo, M.; Pirela, D.; Parra, H.; Nava, M.; Abstract: Metabolic syndrome (MS) is a set of cardio-metabolic risk factors that includes central Chacín, M.; Angarita, L.; Añez, R.; obesity, hyperglycemia, hypertension, and dyslipidemias. -

ADIPOQ) and the Type 1 Receptor (ADIPOR1), Obesity and Prostate Cancer in African Americans
Prostate Cancer and Prostatic Diseases (2010) 13, 362–368 & 2010 Macmillan Publishers Limited All rights reserved 1365-7852/10 www.nature.com/pcan ORIGINAL ARTICLE Genetic variation in adiponectin (ADIPOQ) and the type 1 receptor (ADIPOR1), obesity and prostate cancer in African Americans JL Beebe-Dimmer1, KA Zuhlke2, AM Ray2, EM Lange3 and KA Cooney4 1Department of Population Studies and Prevention, Karmanos Cancer Institute, Department of Internal Medicine, Wayne State University, Detroit, MI, USA; 2Departments of Internal Medicine and Urology, University of Michigan Medical School, Ann Arbor, MI, USA; 3Departments of Genetics and Biostatistics, University of North Carolina, Chapel Hill, NC, USA and 4Departments of Internal Medicine and Urology, University of Michigan Medical School, University of Michigan Comprehensive Cancer Center, Ann Arbor, MI, USA Adiponectin is a protein derived from adipose tissue suspected to have an important role in prostate carcinogenesis. Variants in the adiponectin gene (ADIPOQ) and its type 1 receptor (ADIPOR1) have been recently linked to risk of both breast and colorectal cancer. Therefore, we set out to examine the relationship between polymorphisms in these genes, obesity and prostate cancer in study of African-American men. Ten single-nucleotide polymorphisms (SNPs) in ADIPOQ and ADIPOR1 were genotyped in DNA samples from 131 African-American prostate cancer cases and 344 controls participating in the Flint Men’s Health Study. Logistic regression was then used to estimate their association with prostate cancer and obesity. While no significant associations were detected between any of the tested SNPs and prostate cancer, the rs1501299 SNP in ADIPOQ was significantly associated with body mass (P ¼ 0.03). -
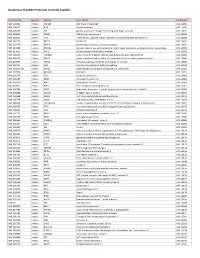
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Brain Neuropeptide Y and CCK and Peripheral Adipokine Receptors: Temporal Response
Brain neuropeptide Y and CCK and peripheral adipokine receptors: temporal response in obesity induced by palatable diet Margaret J Morris 1, Hui Chen 1, Rani Watts 2, Arthur Shulkes 3, David Cameron-Smith 2 1. Department of Pharmacology, School of Medical Sciences, University of New South Wales, 5 NSW 2052, Australia 2. School of Nutrition & Public Health, Deakin University, VIC 3125, Australia 3. Department of Surgery, Austin Health, University of Melbourne, VIC 3084, Australia Running head: NPY, CCK and adipokines in dietary obesity 10 Corresponding author: Professor Margaret J Morris Department of Pharmacology School of Medical Sciences 15 University of New South Wales NSW 2052, Australia. Telephone: 61 2 9385 1560. Fax: 61 2 9385 1059. Email: [email protected] . 20 Abstract Objective: Palatable food disrupts normal appetite regulation, which may contribute to the etiology of obesity. Neuropeptide Y (NPY) and cholecystokinin play critical roles in the regulation of food intake and energy homeostasis, while adiponectin and carnitine palmitoyl- 5 transferase (CPT) are important for insulin sensitivity and fatty acid oxidation. This study examined the impact of short and long term consumption of palatable-high-fat-diet (HFD) on these critical metabolic regulators. Methods: Male C57BL/6 mice were exposed to laboratory chow (12% fat), or cafeteria-style palatable-HFD (32% fat) for 2 or 10 weeks. Body weight and food intake were monitored 10 throughout. Plasma leptin, hypothalamic NPY and cholecystokinin, and mRNA expression of leptin, adiponectin, their receptors, and CPT-1, in fat and muscles were measured. Results: Caloric intake of the palatable-HFD group was 2-3 times greater than control, resulting in a 37% higher body weight. -

Suppression of Adiponectin Receptor 1 Promotes Memory Dysfunction And
www.nature.com/scientificreports OPEN Suppression of adiponectin receptor 1 promotes memory dysfunction and Alzheimer’s Received: 23 May 2017 Accepted: 8 September 2017 disease-like pathologies Published: xx xx xxxx Min Woo Kim, Noman bin Abid, Myeong Hoon Jo, Min Gi Jo, Gwang Ho Yoon & Myeong Ok Kim Recent studies on neurodegeneration have focused on dysfunction of CNS energy metabolism as well as proteinopathies. Adiponectin (ADPN), an adipocyte-derived hormone, plays a major role in the regulation of insulin sensitivity and glucose homeostasis in peripheral organs via adiponectin receptors. In spite of accumulating evidence that adiponectin has neuroprotective properties, the underlying role of adiponectin receptors has not been illuminated. Here, using gene therapy-mediated suppression with shRNA, we found that adiponectin receptor 1 (AdipoR1) suppression induces neurodegeneration as well as metabolic dysfunction. AdipoR1 knockdown mice exhibited increased body weight and abnormal plasma chemistry and also showed spatial learning and memory impairment in behavioural studies. Moreover, AdipoR1 suppression resulted in neurodegenerative phenotypes, diminished expression of the neuronal marker NeuN, and increased expression and activity of caspase 3. Furthermore, AD-like pathologies including insulin signalling dysfunction, abnormal protein aggregation and neuroinfammatory responses were highly exhibited in AdipoR1 knockdown groups, consistent with brain pathologies in ADPN knockout mice. Together, these results suggest that ADPN- AdipoR1 signalling has the potential to alleviate neurodegenerative diseases such as Alzheimer’s diseases. Neurodegeneration is a term describing a pathological phenotype observed in the central nervous system, espe- cially the brain1. Many etiological models of neurodegeneration, such as that in Alzheimer’s disease (AD) and Parkinson’s disease (PD), are based on abnormal protein aggregation and sequentially entail chronic infamma- tion, generation of reactive oxygen species (ROS) and apoptosis2–4. -

Role of Adipose Secreted Factors and Kisspeptin in the Metabolic Control of Gonadotropin Secretion and Puberty1
Chapter 2 Role of Adipose Secreted Factors and Kisspeptin in the Metabolic Control 1 of Gonadotropin Secretion and Puberty Clay A. Lents, C. Richard Barb and Gary J. Hausman Additional information is available at the end of the chapter http://dx.doi.org/10.5772/48802 1. Introduction 1.1. Adipose tissue as an endocrine organ Recent investigations from many species continue to reinforce and validate adipose tissue as an endocrine organ that impacts physiological mechanisms and whole-body homeostasis. Factors secreted by adipose tissue or “adipokines” continue to be discovered and are linked to important physiological roles (Ahima, 2006) including the innate immune response (Schäffler & Schöolmerich, 2010). In a number of recent experiments transcriptional profiling demonstrated that 5,000 to 8,000 adipose tissue genes were differentially expressed during central stimulation of the melanocortin 4 receptor (Barb et al., 2010a) and several conditions such as fasting (Lkhagvadorj et al., 2009) and feed restriction (Lkhagvadorj et al., 2010). In contrast, 300 to 1,800 genes were differentially expressed in livers in these three studies (Barb et al., 2010a; Lkhagvadorj et al., 2009, 2010). This degree of differential gene expression in adipose depots reflects the potential influence of adipose tissue as a secretory organ on multiple systems in the body. Furthermore, advances in the study of adipose tissue gene expression include high throughput technologies in transcriptome profiling and deep sequencing of the adipose tissue microRNA transcriptome (review, Basu et al., 2012). Recent proteomic studies of human and rat adipocytes have revealed the true scope of the adipose tissue secretome (Chen et al., 2005; Kheterpal et al., 2011; Lehr et al., 2012; Lim et al., 2008; Zhong et al., 2010). -

Hepatic Adiponectin Receptor R2 Expression Is Up-Regulated in Normal Adult Male Mice by Chronic Exogenous Growth Hormone Levels
MOLECULAR MEDICINE REPORTS 3: 525-530, 2010 Hepatic adiponectin receptor R2 expression is up-regulated in normal adult male mice by chronic exogenous growth hormone levels YING QIN and YA-PING TIAN Department of Clinical Biochemistry, Chinese PLA General Hospital, Beijing 100853, P.R. China Received February 2, 2010; Accepted March 15, 2010 DOI: 10.3892/mmr_00000292 Abstract. Previous studies investigating the effects of exogenous Introduction growth hormone (GH) on the expression of adiponectin and the adiponectin receptors have generated seemingly conflicting Adiponectin, an adipokine that is secreted exclusively by results. Here, to determine the effects of chronic exogenous GH adipocytes, plays an important role in regulating systemic levels, we investigated the expression of adiponectin receptor energy metabolism and insulin sensitivity in vivo (1,2). The R1 (adipoR1) in skeletal muscle and adiponectin receptor R2 serum adiponectin level is inversely associated with body (adipoR2) in the liver of normal C57BL/6 adult male mice 24 mass index (BMI), which is relevant to metabolic syndrome. weeks after a single injection of rAAV2/1-CMV-GH1 viral Hypoadiponectinemia is associated with insulin resistance particles. AdipoR1 and adipoR2 mRNA expression levels were and hyperinsulinemia (3-7). Adiponectin is present as a determined by quantitative real-time reverse transcription- full-length protein of 30 kDa (fAd), which circulates in polymerase chain reaction. AdipoR1 and adipoR2 proteins trimeric, hexameric or high molecular weight (HMW) forms. were analyzed by Western blotting. Serum total and high A proteolytic cleavage product of adiponectin, known as molecular weight (HMW) adiponectin concentrations were globular adiponectin (gAd), has also been shown to exhibit measured by enzyme-linked immunosorbent assay (ELISA) potent metabolic effects in various tissues (8-14). -

Adiponectin Receptor As a Key Player in Healthy Longevity and Obesity-Related Diseases
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Elsevier - Publisher Connector Cell Metabolism Review Adiponectin Receptor as a Key Player in Healthy Longevity and Obesity-Related Diseases Toshimasa Yamauchi1,* and Takashi Kadowaki1,* 1Department of Diabetes and Metabolic Diseases, Graduate School of Medicine, The University of Tokyo, Tokyo 113-0033, Japan *Correspondence: [email protected] (T.Y.), [email protected] (T.K.) http://dx.doi.org/10.1016/j.cmet.2013.01.001 Adiponectin is a fat-derived hormone whose reduction plays central roles in obesity-linked diseases including insulin resistance/type 2 diabetes and atherosclerosis. The cloning of Adiponectin receptors AdipoR1 and AdipoR2 has stimulated adiponectin research, revealing pivotal roles for AdipoRs in pleiotropic adiponectin actions, as well as some postreceptor signaling mechanisms. Adiponectin signaling has thus become one of the major research fields in metabolism and clinical medicine. Studies on AdipoRs will further our understanding of the role of adiponectin in obesity-linked diseases and shortened life span and may guide the design of antidiabetic and antiaging drugs with AdipoR as a target. Background et al., 1994; Lazar, 2006). Some of these adipokines have been Roles of Adipokines in Obesity-Related Diseases shown to directly or indirectly affect insulin sensitivity through The prevalence of obesity has increased dramatically in recent modulation of insulin signaling and the molecules involved in years (Friedman, 2000). It is commonly associated with type 2 glucose and lipid metabolism. Of these adipokines, adiponectin diabetes, dyslipidemia, and hypertension, and the coexistence has recently attracted much attention because of its antidiabetic of these diseases has been termed metabolic syndrome, which and antiatherogenic effects as well as its antiproliferative effects is related to atherosclerosis (Reaven, 1997; Matsuzawa, 1997).