Improved Regeneration and De Novo Bone Formation in a Diabetic Zebrafish Model Treated with Paricalcitol and Cinacalcet
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Prognostic Utility and Clinical Outcomes of MNX1-AS1 Expression in Cancers: a Systematic Review and Meta-Analysis
The prognostic utility and clinical outcomes of MNX1-AS1 expression in cancers: a systematic review and meta-analysis Juan Li The rst aliated hospital, college of medicine, zhejiang university https://orcid.org/0000-0002-0121-7098 Wen Jin The rst aliated hospital, college of medicine, zhejiang university Zhengyu Zhang The rst aliated hospital, college of medicine, zhejiang university Jingjing Chu The rst aliated hospital, college of medicine, zhejiang university Hui Yang The rst aliated hospital, college of meicine, zhejiang university Chang Li the rst aliated hospital, college of medicine, zhejiang university Ruiyin Dong The rst aliated hospital, college of medicine, zhejiang university Cailian Zhao ( [email protected] ) https://orcid.org/0000-0001-8337-0610 Primary research Keywords: Long non-coding RNA, MNX1-AS1, Cancer, Prognosis Posted Date: March 25th, 2020 DOI: https://doi.org/10.21203/rs.3.rs-19089/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/9 Abstract Background: Recently, emerging studies have identied that MNX1-AS1 highly expressed among variety of cancers and related with worse prognosis of cancer patients. The purpose of this study was to evaluate the relationship between MNX1-AS1 expression with clinical features and prognosis in different cancers. Methods: In this study, we searched the Web of Science, PubMed, CNKI, and Wanfang databases to nd relevant studies of MNX1-AS1. Pooled hazard ratios (HRs) and odds ratios (ORs) with 95% condence intervals (CIs) were applied to explore the prognostic and clinical signicance of MNX1-AS1. Results: A total of 9 literatures were included in this study, including 882 cancer patients. -

Watsonjn2018.Pdf (1.780Mb)
UNIVERSITY OF CENTRAL OKLAHOMA Edmond, Oklahoma Department of Biology Investigating Differential Gene Expression in vivo of Cardiac Birth Defects in an Avian Model of Maternal Phenylketonuria A THESIS SUBMITTED TO THE GRADUATE FACULTY In partial fulfillment of the requirements For the degree of MASTER OF SCIENCE IN BIOLOGY By Jamie N. Watson Edmond, OK June 5, 2018 J. Watson/Dr. Nikki Seagraves ii J. Watson/Dr. Nikki Seagraves Acknowledgements It is difficult to articulate the amount of gratitude I have for the support and encouragement I have received throughout my master’s thesis. Many people have added value and support to my life during this time. I am thankful for the education, experience, and friendships I have gained at the University of Central Oklahoma. First, I would like to thank Dr. Nikki Seagraves for her mentorship and friendship. I lucked out when I met her. I have enjoyed working on this project and I am very thankful for her support. I would like thank Thomas Crane for his support and patience throughout my master’s degree. I would like to thank Dr. Shannon Conley for her continued mentorship and support. I would like to thank Liz Bullen and Dr. Eric Howard for their training and help on this project. I would like to thank Kristy Meyer for her friendship and help throughout graduate school. I would like to thank my committee members Dr. Robert Brennan and Dr. Lilian Chooback for their advisement on this project. Also, I would like to thank the biology faculty and staff. I would like to thank the Seagraves lab members: Jailene Canales, Kayley Pate, Mckayla Muse, Grace Thetford, Kody Harvey, Jordan Guffey, and Kayle Patatanian for their hard work and support. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Htra1 Is a Novel Transcriptional Target of RUNX2 That Promotes Osteogenic Differentiation
Cellular Physiology Cell Physiol Biochem 2019;53:832-850 DOI: 10.33594/00000017610.33594/000000176 © 2019 The Author(s).© 2019 Published The Author(s) by and Biochemistry Published online: online: 9 9November November 2019 2019 Cell Physiol BiochemPublished Press GmbH&Co. by Cell Physiol KG Biochem 832 Press GmbH&Co. KG, Duesseldorf IyyanarAccepted: et 7al.: November Runx2 Regulates 2019 Htra1 During Osteogenesiswww.cellphysiolbiochem.com This article is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 Interna- tional License (CC BY-NC-ND). Usage and distribution for commercial purposes as well as any distribution of modified material requires written permission. Original Paper Htra1 is a Novel Transcriptional Target of RUNX2 That Promotes Osteogenic Differentiation Paul P.R. Iyyanara,b Merlin P. Thangaraja B. Frank Eamesc Adil J. Nazaralia aLaboratory of Molecular Cell Biology, College of Pharmacy and Nutrition and Neuroscience Research Cluster, University of Saskatchewan, Saskatoon, SK, Canada, bDivision of Developmental Biology, Cincinnati Children’s Hospital Medical Center, Cincinnati, OH, USA, cDepartment of Anatomy and Cell Biology, University of Saskatchewan, Saskatoon, SK, Canada Key Words Runx2 • Htra1 • Osteoblast differentiation • Matrix mineralization Abstract Background/Aims: Runt-related transcription factor 2 (Runx2) is a master regulator of osteogenic differentiation, but most of the direct downstream targets of RUNX2 during osteogenesis are unknown. Likewise, High-temperature requirement factor A1 (HTRA1) is a serine protease expressed in bone, yet the role of Htra1 during osteoblast differentiation remains elusive. We investigated the role of Htra1 in osteogenic differentiation and the transcriptional regulation of Htra1 by RUNX2 in primary mouse mesenchymal progenitor cells. Methods: Overexpression of Htra1 was carried out in primary mouse mesenchymal progenitor cells to evaluate the extent of osteoblast differentiation. -

SUPPLEMENTARY MATERIAL Bone Morphogenetic Protein 4 Promotes
www.intjdevbiol.com doi: 10.1387/ijdb.160040mk SUPPLEMENTARY MATERIAL corresponding to: Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells SUMIYO MIMURA, MIKA SUGA, KAORI OKADA, MASAKI KINEHARA, HIROKI NIKAWA and MIHO K. FURUE* *Address correspondence to: Miho Kusuda Furue. Laboratory of Stem Cell Cultures, National Institutes of Biomedical Innovation, Health and Nutrition, 7-6-8, Saito-Asagi, Ibaraki, Osaka 567-0085, Japan. Tel: 81-72-641-9819. Fax: 81-72-641-9812. E-mail: [email protected] Full text for this paper is available at: http://dx.doi.org/10.1387/ijdb.160040mk TABLE S1 PRIMER LIST FOR QRT-PCR Gene forward reverse AP2α AATTTCTCAACCGACAACATT ATCTGTTTTGTAGCCAGGAGC CDX2 CTGGAGCTGGAGAAGGAGTTTC ATTTTAACCTGCCTCTCAGAGAGC DLX1 AGTTTGCAGTTGCAGGCTTT CCCTGCTTCATCAGCTTCTT FOXD3 CAGCGGTTCGGCGGGAGG TGAGTGAGAGGTTGTGGCGGATG GAPDH CAAAGTTGTCATGGATGACC CCATGGAGAAGGCTGGGG MSX1 GGATCAGACTTCGGAGAGTGAACT GCCTTCCCTTTAACCCTCACA NANOG TGAACCTCAGCTACAAACAG TGGTGGTAGGAAGAGTAAAG OCT4 GACAGGGGGAGGGGAGGAGCTAGG CTTCCCTCCAACCAGTTGCCCCAAA PAX3 TTGCAATGGCCTCTCAC AGGGGAGAGCGCGTAATC PAX6 GTCCATCTTTGCTTGGGAAA TAGCCAGGTTGCGAAGAACT p75 TCATCCCTGTCTATTGCTCCA TGTTCTGCTTGCAGCTGTTC SOX9 AATGGAGCAGCGAAATCAAC CAGAGAGATTTAGCACACTGATC SOX10 GACCAGTACCCGCACCTG CGCTTGTCACTTTCGTTCAG Suppl. Fig. S1. Comparison of the gene expression profiles of the ES cells and the cells induced by NC and NC-B condition. Scatter plots compares the normalized expression of every gene on the array (refer to Table S3). The central line -
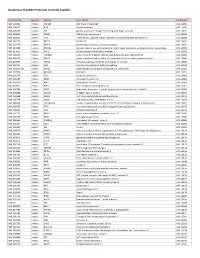
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Rabbit Anti-Osterix/FITC Conjugated Antibody-SL1110R-FITC
SunLong Biotech Co.,LTD Tel: 0086-571- 56623320 Fax:0086-571- 56623318 E-mail:[email protected] www.sunlongbiotech.com Rabbit Anti-Osterix/FITC Conjugated antibody SL1110R-FITC Product Name: Anti-Osterix/FITC Chinese Name: FITC标记的成骨相关转录因子抗体 Osterix; MGC126598; Osx; Sp 7; Sp7; Sp7 transcription factor; Transcription factor Alias: Sp7; Zinc finger protein osterix; SP7_HUMAN. Organism Species: Rabbit Clonality: Polyclonal React Species: Human,Mouse,Rat,Dog,Pig,Cow,Horse,Rabbit, Flow-Cyt=1:50-200IF=1:50-200 Applications: not yet tested in other applications. optimal dilutions/concentrations should be determined by the end user. Molecular weight: 45kDa Form: Lyophilized or Liquid Concentration: 1mg/ml immunogen: KLH conjugated synthetic peptide derived from human Osterix Lsotype: IgG Purification: affinity purified by Protein A Storage Buffer: 0.01M TBS(pH7.4) with 1% BSA, 0.03% Proclin300 and 50% Glycerol. Storewww.sunlongbiotech.com at -20 °C for one year. Avoid repeated freeze/thaw cycles. The lyophilized antibody is stable at room temperature for at least one month and for greater than a year Storage: when kept at -20°C. When reconstituted in sterile pH 7.4 0.01M PBS or diluent of antibody the antibody is stable for at least two weeks at 2-4 °C. background: This gene encodes a member of the Sp subfamily of Sp/XKLF transcription factors. Sp family proteins are sequence-specific DNA-binding proteins characterized by an amino- terminal trans-activation domain and three carboxy-terminal zinc finger motifs. This Product Detail: protein is a bone specific transcription factor and is required for osteoblast differentiation and bone formation.[provided by RefSeq, Jul 2010] Function: Transcriptional activator essential for osteoblast differentiation. -

Ingenuity Pathway Analysis of Human Facet Joint Tissues: Insight Into Facet Joint Osteoarthritis
EXPERIMENTAL AND THERAPEUTIC MEDICINE 19: 2997-3008, 2020 Ingenuity pathway analysis of human facet joint tissues: Insight into facet joint osteoarthritis CHU CHEN*, SHENGYU CUI*, WEIDONG LI, HURICHA JIN, JIANBO FAN, YUYU SUN and ZHIMING CUI Department of Spine Surgery, The Second Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China Received August 17, 2019; Accepted January 30, 2020 DOI: 10.3892/etm.2020.8555 Abstract. Facet joint osteoarthritis (FJOA) is a common the top 5 IPA networks (with a score >30). The present study degenerative joint disorder with high prevalence in the elderly. provides insight into the pathological processes of FJOA from FJOA causes lower back pain and lower extremity pain, and a genetic perspective and may thus benefit the clinical treat- thus severely impacts the quality of life of affected patients. ment of FJOA. Emerging studies have focused on the histomorphological and histomorphometric changes in FJOA. However, the dynamic Introduction genetic changes in FJOA have remained to be clearly deter- mined. In the present study, previously obtained RNA deep Facet joint osteoarthritis (FJOA) is a common degenerative sequencing data were subjected to an ingenuity pathway joint disorder that causes the degeneration and breakdown analysis (IPA) and canonical signaling pathways of differ- of cartilage and restricts the movement of joints (1). It has entially expressed genes (DEGs) in FJOA were studied. The been reported that lumbar FJOA occurs at high prevalence top 25 enriched canonical signaling pathways were identified and the presence of FJOA is highly associated with age (2,3). and canonical signaling pathways with high absolute values A community-based cross-sectional study indicated that of z-scores, specifically leukocyte extravasation signaling, in populations aged <50 years, the prevalence of FJOA was Tec kinase signaling and osteoarthritis pathway, were inves- <45%, while it was ~75% in populations aged >50 years (4). -

Microrna Mir-378 Promotes BMP2-Induced Osteogenic
Hupkes et al. BMC Molecular Biology 2014, 15:1 http://www.biomedcentral.com/1471-2199/15/1 RESEARCH ARTICLE Open Access MicroRNA miR-378 promotes BMP2-induced osteogenic differentiation of mesenchymal progenitor cells Marlinda Hupkes1*, Ana M Sotoca1, José M Hendriks1, Everardus J van Zoelen1 and Koen J Dechering1,2,3 Abstract Background: MicroRNAs (miRNAs) are a family of small, non-coding single-stranded RNA molecules involved in post-transcriptional regulation of gene expression. As such, they are believed to play a role in regulating the step-wise changes in gene expression patterns that occur during cell fate specification of multipotent stem cells. Here, we have studied whether terminal differentiation of C2C12 myoblasts is indeed controlled by lineage-specific changes in miRNA expression. Results: Using a previously generated RNA polymerase II (Pol-II) ChIP-on-chip dataset, we show differential Pol-II occupancy at the promoter regions of six miRNAs during C2C12 myogenic versus BMP2-induced osteogenic differentiation. Overexpression of one of these miRNAs, miR-378, enhances Alp activity, calcium deposition and mRNA expression of osteogenic marker genes in the presence of BMP2. Conclusions: Our results demonstrate a previously unknown role for miR-378 in promoting BMP2-induced osteogenic differentiation. Background nucleotides) involved in post-transcriptional gene silencing The generation of distinct populations of terminally dif- and as such play important roles in diverse biological pro- ferentiated, mature specialized cell types from multipo- cesses such as developmental timing [3], insulin secretion tent stem cells, via progenitor cells, is characterized by a [4], apoptosis [5], oncogenesis [6] and organ development progressive restriction of differentiation potential that [7,8]. -

Gene 599 (2017) 36–52
Gene 599 (2017) 36–52 Contents lists available at ScienceDirect Gene journal homepage: www.elsevier.com/locate/gene Research paper Old age and the associated impairment of bones' adaptation to loading are associated with transcriptomic changes in cellular metabolism, cell-matrix interactions and the cell cycle Gabriel L. Galea, PhD a,1, Lee B. Meakin, PhD a,⁎,1, Marie A. Harris, PhD b, Peter J. Delisser, BVSc(Hons) a, Lance E. Lanyon, DSc a, Stephen E. Harris, PhD b, Joanna S. Price, PhD a a School of Veterinary Sciences, University of Bristol, Bristol, UK b Department of Periodontics & Cellular and Structural Biology, University of Texas Health Science Centre, San Antonio, USA article info abstract Article history: In old animals, bone's ability to adapt its mass and architecture to functional load-bearing requirements is dimin- Received 15 October 2016 ished, resulting in bone loss characteristic of osteoporosis. Here we investigate transcriptomic changes associated Accepted 6 November 2016 with this impaired adaptive response. Young adult (19-week-old) and aged (19-month-old) female mice were Available online 10 November 2016 subjected to unilateral axial tibial loading and their cortical shells harvested for microarray analysis between 1 h and 24 h following loading (36 mice per age group, 6 mice per loading group at 6 time points). In non-loaded Keywords: aged bones, down-regulated genes are enriched for MAPK, Wnt and cell cycle components, including E2F1. E2F1 Ageing Bone is the transcription factor most closely associated with genes down-regulated by ageing and is down-regulated at Mechanical loading the protein level in osteocytes. -

Table of Contents
The Role of CHD1 during Mesenchymal Stem Cell Differentiation Dissertation for the award of the degree “Doctor rerum naturalium (Dr. rer. nat.)” Division of Mathematics and Natural Sciences of the Georg-August-Universität Göttingen submitted by Simon Baumgart born in Gießen Göttingen, 2015 Members of the Thesis Committee: Prof. Dr. Steven A. Johnsen (Reviewer) Department of General, Visceral and Pediatric Surgery University of Göttingen Medical School, Göttingen Prof. Dr. Heidi Hahn (Reviewer) Department of Human Genetics University of Göttingen Medical School, Göttingen Prof. Dr. Jürgen Wienands Department of Cellular and Molecular Immunology University of Göttingen Medical School, Göttingen Date of oral examination: 22nd of February, 2016 Affidavit I hereby declare that the PhD thesis entitled “The Role of CHD1 during Mesenchymal Stem Cell Differentiation” has been written independently and with no other sources and aids than quoted. Simon Baumgart December, 2015 Göttingen Table of Contents Table of Contents Abbreviations ............................................................................................................... I List of Figures ........................................................................................................... VII Summary ................................................................................................................... IX 1 Introduction .............................................................................................................. 1 1.1 DNA organization ............................................................................................. -

Spatial and Temporal Specification of Neural Fates by Transcription Factor Codes François Guillemot
REVIEW 3771 Development 134, 3771-3780 (2007) doi:10.1242/dev.006379 Spatial and temporal specification of neural fates by transcription factor codes François Guillemot The vertebrate central nervous system contains a great diversity Box 1. Neurons and glial cells of neurons and glial cells, which are generated in the embryonic neural tube at specific times and positions. Several classes of transcription factors have been shown to control various steps in the differentiation of progenitor cells in the neural tube and to determine the identity of the cells produced. Recent evidence indicates that combinations of transcription factors of the homeodomain and basic helix-loop-helix families establish molecular codes that determine both where and when the different kinds of neurons and glial cells are generated. Introduction Neuron Oligodendrocyte Astrocyte A multitude of neurons of different types, as well as oligodendrocytes and astrocytes (see Box 1), are generated as the vertebrate central The vertebrate central nervous system comprises three primary cell nervous system develops. These different neural cells are generated types, including neurons and two types of glial cells. Neurons are at defined times and positions by multipotent progenitors located in electrically excitable cells that process and transmit information via the walls of the embryonic neural tube. Progenitors located in the the release of neurotransmitters at synapses. Different subtypes of ventral neural tube at spinal cord level first produce motor neurons, neurons can be distinguished by the morphology of their cell body which innervate skeletal muscles and later produce oligodendrocytes and dendritic tree, the type of cells they connect with via their axon, the type of neurotransmitter used, etc.