Tetrabromobisphenol A
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Petition to Order Testing of Tetrabromobisphenol a (“TBBPA”)
PETITION TO ORDER TESTING OF TETRABROMOBISPHENOL A (CAS NO. 79-94-7) UNDER SECTION 4(a) OF THE TOXIC SUBSTANCES CONTROL ACT (DECEMBER 13, 2016) Via Federal Express & Electronic Mail Gina McCarthy, Administrator U.S. Environmental Protection Agency 1200 Pennsylvania Avenue, N.W. Mail Code: 1101A Washington D.C. 20460 [email protected] Dear Administrator McCarthy: Earthjustice,1 Natural Resources Defense Council (NRDC), 2 Toxic-Free Future (TFF),3 Safer Chemicals, Healthy Families (SCHF),4 BlueGreen Alliance (BGA),5 and Environmental Health Strategy Center (EHSC),6 submit this Petition to the U.S. Environmental Protection Agency (“EPA”), pursuant to section 21 of the Toxic Substances Control Act (“TSCA”),7 to 1 Earthjustice is the nation’s largest environmental law organization. Protecting people and the environment from exposure to toxic substances is a key part of its mission. Earthjustice submits this petition on behalf of NRDC, SCHF, TFF, BGA, and EHSC. 2 NRDC is an international nonprofit environmental organization with more than 2 million members and online activists. Since 1970, our lawyers, scientists, and other environmental specialists have worked to protect the world's natural resources, public health, and the environment. Protecting families and communities from toxic chemicals is a key NRDC goal. 3 TFF advocates for the use of safer products, chemicals, and practices through advanced research, advocacy, grassroots organizing, and consumer engagement to ensure a healthier tomorrow. 4 SCHF is a coalition representing over 450 organizations and businesses united by a common concern about toxic chemicals in our homes, places of work, and products we use every day. -

Uncovering Evidence for Endocrine-Disrupting Chemicals That Elicit Differential Susceptibility Through Gene-Environment Interactions
toxics Review Uncovering Evidence for Endocrine-Disrupting Chemicals That Elicit Differential Susceptibility through Gene-Environment Interactions Dylan J. Wallis 1 , Lisa Truong 2 , Jane La Du 2, Robyn L. Tanguay 2 and David M. Reif 1,* 1 Department of Biological Sciences, North Carolina State University, Raleigh, NC 27695, USA; [email protected] 2 Department of Environmental and Molecular Toxicology, Oregon State University, Corvallis, OR 97331, USA; [email protected] (L.T.); [email protected] (J.L.D.); [email protected] (R.L.T.) * Correspondence: [email protected] Abstract: Exposure to endocrine-disrupting chemicals (EDCs) is linked to myriad disorders, charac- terized by the disruption of the complex endocrine signaling pathways that govern development, physiology, and even behavior across the entire body. The mechanisms of endocrine disruption in- volve a complex system of pathways that communicate across the body to stimulate specific receptors that bind DNA and regulate the expression of a suite of genes. These mechanisms, including gene regulation, DNA binding, and protein binding, can be tied to differences in individual susceptibility across a genetically diverse population. In this review, we posit that EDCs causing such differential responses may be identified by looking for a signal of population variability after exposure. We begin Citation: Wallis, D.J.; Truong, L.; La by summarizing how the biology of EDCs has implications for genetically diverse populations. We Du, J.; Tanguay, R.L.; Reif, D.M. then describe how gene-environment interactions (GxE) across the complex pathways of endocrine Uncovering Evidence for Endocrine- signaling could lead to differences in susceptibility. We survey examples in the literature of individual Disrupting Chemicals That Elicit susceptibility differences to EDCs, pointing to a need for research in this area, especially regarding Differential Susceptibility through the exceedingly complex thyroid pathway. -

OFR Staff Plan
Staff Briefing Package Project Plan: Organohalogen Flame Retardant Chemicals Assessment July 1, 2020 CPSC Consumer Hotline and General Information: 1-800-638-CPSC (2772) CPSC's Web Site: http://www.cpsc.gov THIS DOCUMENT HAS NOT BEEN REVIEWED CLEARED FOR PUBLIC RELEASE OR ACCEPTED BY THE COMMISSION UNDER CPSA 6(b)(1) Acknowledgments The preparation, writing, and review of this report was supported by a team of staff. We acknowledge and thank team members for their significant contributions. Michael Babich, Ph.D., Directorate for Health Sciences Charles Bevington, M.P.H., Directorate for Health Sciences Xinrong Chen, Ph.D., D.A.B.T., Directorate for Health Sciences Eric Hooker, M.S., D.A.B.T., Directorate for Health Sciences Cynthia Gillham, M.S., Directorate for Economic Analysis John Gordon, Ph.D., Directorate for Health Sciences Kristina Hatlelid, Ph.D., M.P.H., Directorate for Health Sciences Barbara Little, Attorney, Office of the General Counsel Joanna Matheson, Ph.D., Directorate for Health Sciences ii THIS DOCUMENT HAS NOT BEEN REVIEWED CLEARED FOR PUBLIC RELEASE OR ACCEPTED BY THE COMMISSION UNDER CPSA 6(b)(1) Table of Contents Briefing Memo ............................................................................................................................... iv 1. Executive summary .............................................................................................................. 5 2. Introduction ......................................................................................................................... -

FLAME RETARDANTS in PRINTED CIRCUIT BOARDS Chapter 5
FLAME RETARDANTS IN PRINTED CIRCUIT BOARDS Chapter 5 FINAL REPORT August 2015 EPA Publication 744-R-15-001 5 Potential Exposure to Flame Retardants and Other Life- Cycle Considerations Many factors must be considered to evaluate the risk to human health and the environment posed by any flame-retardant chemical. Risk is a function of two parameters, hazard and exposure. The hazard associated with a particular substance or chemical is its potential to impair human health, safety, or ecological health. While some degree of hazard can be assigned to most substances, the toxicity and harmful effects of other substances are not fully understood. The exposure potential of a given substance is a function of the exposure route (inhalation, ingestion, and dermal), the concentration of the substance in the contact media, and the frequency and duration of the exposure. The purpose of this chapter is to identify the highest priority routes of exposure to flame- retardant chemicals used in printed circuit boards (PCBs). Section 5.1 through Section 5.4 provide general background regarding potential exposure pathways that can occur during different life-cycle stages, discuss factors that affect exposure potential in an industrial setting, provide process descriptions for the industrial operations involved in the PCB manufacturing supply chain (identifying the potential primary release points and exposure pathways), and discuss potential consumer and environmental exposures. Following this general discussion, Section 5.5 highlights life-cycle considerations for the ten flame retardants evaluated by this partnership. The chapter is intended to help the reader identify and characterize the exposure potential of flame-retardant chemicals based on factors including physical and chemical properties and reactive versus additive incorporation into the epoxy resin. -

June 19, 2019 Transmitted Electronically
June 19, 2019 Transmitted Electronically Document Control Manager Office of Pollution Prevention and Toxics (OPPT) US EPA 1200 Pennsylvania Avenue, NW Washington, DC 20460-0001 Dear Sir/Maddam, The Center for Environmental Health (CEH) hereby submits these comments on the proposed Toxic Substances Control Act high-priority candidates (EPA-HQ-OPPT-2019-0131- 0001). As required by the Toxic Substances Control Act (TSCA), the United States Environmental Protection Agency (USEPA) must regularly release a list of chemicals that are under review for prioritization for risk evaluation. This list consists of 20 high-priority and 20 low-priority candidates. These comments identify data on the 2019 list of the 20 high-priority candidates (Table 1) in the literature that support the notion that these chemicals may be endocrine disrupting compounds (EDCs). EDCs are a class of chemicals that can interfere with the production, action, transport, or metabolism of endogenous hormones in the body.1 For the purposes of these comments, an EDC will be considered any compound that showed an effect on an endocrine system at any dose or concentration, thus accounting for any potential dose responses that may be non-monotonic. This is supported in the literature wherein disruption of upstream signaling, even at low doses and within “normal” levels of hormone variability, adverse outcomes can be observed.2,3 These comments will describe various studies to determine whether the compound of interest could be considered an EDC. This was accomplished by searching PubMed as well as The Endocrine Disruption Exchange’s List of Potential Endocrine Disruptors for peer- reviewed articles by searching with the CAS Registry Number for each chemical; USEPA’s Computational Toxicology Dashboard was also searched. -

TBBPA Clh Report
CLH REPORT FOR TBBPA CLH report Proposal for Harmonised Classification and Labelling Based on Regulation (EC) No 1272/2008 (CLP Regulation), Annex VI, Part 2 International Chemical Identification: 2,2',6,6'-tetrabromo-4,4'-isopropylidenediphenol; tetrabromobisphenol-A (TBBPA) EC Number: 201-236-9 CAS Number: 79-94-7 Index Number: 604-074-00-0 Contact details for dossier submitter: Norwegian Environment Agency P.O. Box 5672 Torgarden, 7485 Trondheim, Norway [email protected] +47 73 58 05 00 Co-submitter: The Danish Environmental Protection Agency Version number: 2.0 Date: 18.09.2020 [04.01-MF-003.01] CLH REPORT FOR TBBPA CONTENTS 1 IDENTITY OF THE SUBSTANCE........................................................................................................................1 1.1 NAME AND OTHER IDENTIFIERS OF THE SUBSTANCE...............................................................................................1 1.2 COMPOSITION OF THE SUBSTANCE..........................................................................................................................2 2 PROPOSED HARMONISED CLASSIFICATION AND LABELLING............................................................3 2.1 PROPOSED HARMONISED CLASSIFICATION AND LABELLING ACCORDING TO THE CLP CRITERIA............................3 3 HISTORY OF THE PREVIOUS CLASSIFICATION AND LABELLING ......................................................5 4 JUSTIFICATION THAT ACTION IS NEEDED AT COMMUNITY LEVEL.................................................5 5 IDENTIFIED USES..................................................................................................................................................5 -
![Tetrabromobisphenol a [79-94-7]](https://docslib.b-cdn.net/cover/1113/tetrabromobisphenol-a-79-94-7-581113.webp)
Tetrabromobisphenol a [79-94-7]
Tetrabromobisphenol A [79-94-7] Review of Toxicological Literature June 2002 Tetrabromobisphenol A [79-94-7] Review of Toxicological Literature Prepared for Scott Masten, Ph.D. National Institute of Environmental Health Sciences P.O. Box 12233 Research Triangle Park, North Carolina 27709 Contract No. N01-ES-65402 Submitted by Karen E. Haneke, M.S. Integrated Laboratory Systems P.O. Box 13501 Research Triangle Park, North Carolina 27709 June 2002 Toxicological Summary for Tetrabromobisphenol A [79-94-7] 06/2002 Executive Summary Basis for Nomination Tetrabromobisphenol A (TBBPA) was nominated by the NIEHS for toxicological characterization including neurodevelopmental toxicity studies and carcinogenicity. The nomination is based on its high production volume, widespread human exposures, and suspicion to cause thyroid toxicity and thyroid tumors. Nontoxicological Information TBBPA contains 58.7% bromine. When pyrolyzed in open quartz tubes for ten minutes, TBBPA produces polybrominated dibenzo-p-dioxins (PBDDs) and -furans (PBDFs) at 700 °C. When used as a flame retardant, TBBPA contains <0.01 to 0.05 µg/kg PBDDs and <0.01 to 0.02 µg/kg PBDFs. TBBPA can be detected by chromatographic methods coupled with mass spectrometry (MS) or other detectors. Gas chromatography (GC) and GC/MS are used after conversion of TBBPA to the diethyl derivative by ethylation. The Occupational Safety and Health Administration (OSHA) air sampling method for TBBPA is high-performance liquid chromatography (HPLC) with ultraviolet (UV) detection. TBBPA has been determined in human plasma using solid-phase extraction (SPE) and GC with electron-capture MS (ECMS) detection in the concentration range of 4-200 pg/g plasma; the estimated detection limit was 0.8 pg/g plasma. -

ROHS Annex II Dossier for TBBP-A Restriction Proposal for Substances in Electrical and Electronic Equipment Under Rohs
www.oeko.de ROHS Annex II Dossier for TBBP-A Restriction proposal for substances in electrical and electronic equipment under RoHS Report No. 2 Version 2 Substance Name: Tetrabromobisphenol A 04/12/2019 (TBBP-A, flame retardant) (2,2',6,6'-tetrabromo-4,4'-isopropylidenediphenol) EC Number: 201-236-9 CAS Number: 79-94-7 Head Office Freiburg P.O. Box 17 71 79017 Freiburg Street address Merzhauser Strasse 173 79100 Freiburg Tel. +49 761 45295-0 Office Berlin Schicklerstrasse 5-7 10179 Berlin Tel. +49 30 405085-0 Office Darmstadt Rheinstrasse 95 64295 Darmstadt Tel. +49 6151 8191-0 [email protected] www.oeko.de RoHS Annex II Dossier, V. 2 TBBP-A (flame retardant) Table of Contents List of Tables 4 Abbreviations 5 CONTEXT and SCOPE of the Substance Assessment 7 1. IDENTIFICATION, CLASSIFICATION AND LABELLING, LEGAL STATUS AND USE RESTRICTIONS 9 1.1.1. Name, other identifiers, and composition of the substance 9 1.1.2. Physico-chemical properties 10 1.2. Classification and labelling status 10 1.3. Legal status and use restrictions 11 1.3.1. Regulation of the substance under REACH 11 1.3.2. Other legislative measures 12 1.3.3. Non-governmental initiatives 12 2. USE IN ELECTRICAL AND ELECTRONIC EQUIPMENT 14 2.1. Function of the substance 14 2.2. Types of applications / types of materials 14 2.3. Quantities of the substance used 17 3. HUMAN HEALTH HAZARD PROFILE 19 3.1. Endpoints of concern 19 3.2. Existing Guidance values (DNELs, OELs) 20 3.3. Non-testing information opposing existing DNELs 21 4. -

An Overview of Alternatives to Tetrabromobisphenol a (TBBPA) and Hexabromocyclododecane (HBCD)
An Overview of Alternatives to Tetrabromobisphenol A (TBBPA) and Hexabromocyclododecane (HBCD) By Gregory Morose A Publication Of the Lowell Center For Sustainable Production Prepared for The Jennifer University of Massachusetts Altman Foundation Lowell March 2006 Purpose of this Study The Jennifer Altman Foundation has commissioned the Lowell Center for Sustainable Production (LCSP) of the University of Massachusetts Lowell to conduct a study of potential alternatives to TBBPA and HBCD. The Lowell Center for Sustainable Production The Lowell Center for Sustainable Production develops, studies, and promotes environmentally sound systems of production, healthy work environments, and economically viable work organizations. The Center operates on the premise that environmental quality, safe and healthy workplaces, and social accountability can be achieved while at the same time enhancing the economic life of firms and communities. This is accomplished by broadening the fundamental design criteria for all productive activities to include an explicit and comprehensive commitment to sustainability. The Center is composed of faculty and staff at the University of Massachusetts Lowell who work directly with industrial firms, social services institutions, citizen organizations, and government agencies to promote sustainable production. The LCSP is based at the University of Massachusetts Lowell, where it works closely with the Massachusetts Toxics Use Reduction Institute (TURI) and the Department of Work Environment. An Overview of Alternatives to Tetrabromobisphenol A (TBBPA) and Hexabromocyclododecane (HBCD) March, 2006 Prepared for: The Jennifer Altman Foundation By Gregory Morose A Publication of the Lowell Center for Sustainable Production University of Massachusetts Lowell One University Avenue Lowell, MA 01854 978-934-2980 Table of Contents INTRODUCTION........................................................................................ -

Environmental Pollutants and the Reproductive System in Birds
Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 565 _____________________________ _____________________________ Environmental Pollutants and the Reproductive System in Birds Developmental Effects of Estrogenic Compounds BY CECILIA BERG ACTA UNIVERSITATIS UPSALIENSIS UPPSALA 2000 Dissertation for the Degree of Doctor of Philosophy in Environmental Toxicology presented at Uppsala University in 2000 ABSTRACT Berg, C. 2000. Environmental Pollutants and the Reproductive System in Birds. Developmental effects of estrogenic compounds. Acta Universitatis Upsaliensis. Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 565, 63 pp. Uppsala. ISBN 91-554-4798-8. A number of environmental pollutants have been shown to mimick the action of the female sex hormone estrogen and are, therefore, suspected to be responsible for reproductive abnormalities seen in wildlife. Test systems which can be used in hazard and risk assessment of chemicals with estrogenic effects are consequently needed. In this thesis, I propose the avian egg as an in vivo test system for estrogenic compounds. I conclude that malformation of the left testis and the Müllerian ducts (MDs: embryonic oviducts) in avian embryos can be used as endpoints to examine estrogenic activity of chemicals. MD malformation is more easily determined and thereby faster to use as an endpoint than histologically observed feminization of the testis. The usefulness of MD/oviduct malformations as biomarkers for estrogenic effects in wild birds should be considered. The environmental pollutants bisphenol A (BPA) and o,p´-DDT induced similar effects as the synthetic estrogens, ethynylestradiol and diethylstilbestrol. BPA caused MD malformations in quail embryos and ovotestis formation in chicken embryos. -

Download PDF Fact Sheet
PollutionTracker Fact Sheet Tetrabromobisphenol A (TBBPA) What is it? Tetrabromobisphenol A (TBBPA) is a brominated flame retardant used in flame- retarded epoxy and polycarbonate resins and is currently the highest selling brominated flame retardant globally. Resins containing TBBPA have wide- ranging applications, including communications and electronics equipment, automotive parts, and appliances.1 TBBPA is not known to have been manufactured in Canada. How does it get into the ocean? The major pathways by which brominated flame retardants enter the marine environment are likely via leaching from finished products into wastewater systems and via landfill runoff.1,2 TBBPA has relatively low solubility in water, and therefore, is likely to bind to organic matter in sediments. TBBPA may also accumulate in the lipids of marine organisms.1 ) 1 PollutionTracker Fact Sheet: Tetrabromobisphenol A (TBBPA) Is it a problem? Like other brominated flame retardants, TBBPA is persistent in the environment and can accumulate in organisms (bioaccumulative). It has even been detected in the remote Arctic in sediments, plants, fish, and birds.3 FACT: Bisphenol A, a hormone mimic that has received much public attention due to its presence in plastic water bottles, is a breakdown product of TBBPA. As a group, brominated flame retardants are known to have toxic effects in terrestrial and aquatic organisms, with TBBPA known to have adverse effects on survival, reproduction, and development.1 In bluegill sunfish and rainbow trout, exposure to TBBPA resulted in irritation, twitching, and erratic swimming, while in fathead minnow, exposure resulted in reduced growth.4 Zebrafish embryos exposed to TBBPA experienced swelling, hemorrhage, decreased heart rate, tail malformations, and death.5 One of the degradation products of TBBPA in the environment is bisphenol A. -
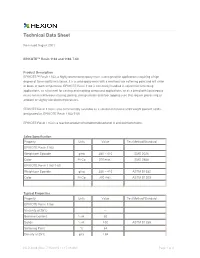
Technical Data Sheet
Technical Data Sheet Re-issued August 2001 EPIKOTE™ Resin 1163 and 1163-T-60 Product Description EPIKOTE™ Resin 1163, a highly brominated epoxy resin, is designed for applications requiring a high degree of flammability resistance. It is a solid epoxy resin with a relatively low softening point and will sinter or block at room temperature. EPIKOTE Resin 1163 is commonly handled in solvent for laminating applications, as a hot melt for casting and molding compound applications, or as a blend with liquid epoxy resins for miscellaneous casting, potting, encapsulation and floor topping uses that require processing at ambient or slightly elevated temperatures. EPIKOTE Resin 1163 is also commercially available as a solution in toluene at 60 weight percent solids, designated as EPIKOTE Resin 1163-T-60. EPIKOTE Resin 1163 is a reaction product of tetrabromobisphenol A and epichlorohydrin. Sales Specification Property Units Value Test Method/Standard EPIKOTE Resin 1163 Weight per Epoxide g/eq 380 – 410 SMS 2026 Color Pt-Co 200 max.1 SMS 2858 EPIKOTE Resin 1163-T-60 Weight per Epoxide g/eq 380 – 410 ASTM D1652 Color Pt-Co 300 max. ASTM D1209 Typical Properties Property Units Value Test Method/Standard EPIKOTE Resin 1163 Viscosity at 25°C --- Bromine Content % wt. 50 Solids % wt. 100 ASTM D1259 Softening Point °C 64 Density at 25°C g/cc 1.84 HCD-4038 (Rev. 7/15/2015 11:17:39 AM) Page 1 of 4 EPIKOTE Resin 1163 and 1163-T-60 Appearance Off white, coarse powder 2 EPIKOTE Resin 1163-T-60 Viscosity at 25°C cP 10 Bromine Content % wt.