Study of Electron and Positron Elastic Scattering from Hydrogen Sulphide Using Analytically Obtained Static Potential
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
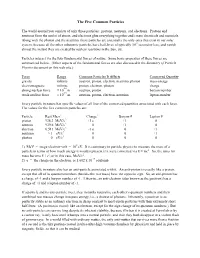
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

A Discussion on Characteristics of the Quantum Vacuum
A Discussion on Characteristics of the Quantum Vacuum Harold \Sonny" White∗ NASA/Johnson Space Center, 2101 NASA Pkwy M/C EP411, Houston, TX (Dated: September 17, 2015) This paper will begin by considering the quantum vacuum at the cosmological scale to show that the gravitational coupling constant may be viewed as an emergent phenomenon, or rather a long wavelength consequence of the quantum vacuum. This cosmological viewpoint will be reconsidered on a microscopic scale in the presence of concentrations of \ordinary" matter to determine the impact on the energy state of the quantum vacuum. The derived relationship will be used to predict a radius of the hydrogen atom which will be compared to the Bohr radius for validation. The ramifications of this equation will be explored in the context of the predicted electron mass, the electrostatic force, and the energy density of the electric field around the hydrogen nucleus. It will finally be shown that this perturbed energy state of the quan- tum vacuum can be successfully modeled as a virtual electron-positron plasma, or the Dirac vacuum. PACS numbers: 95.30.Sf, 04.60.Bc, 95.30.Qd, 95.30.Cq, 95.36.+x I. BACKGROUND ON STANDARD MODEL OF COSMOLOGY Prior to developing the central theme of the paper, it will be useful to present the reader with an executive summary of the characteristics and mathematical relationships central to what is now commonly referred to as the standard model of Big Bang cosmology, the Friedmann-Lema^ıtre-Robertson-Walker metric. The Friedmann equations are analytic solutions of the Einstein field equations using the FLRW metric, and Equation(s) (1) show some commonly used forms that include the cosmological constant[1], Λ. -

Part B. Radiation Sources Radiation Safety
Part B. Radiation sources Radiation Safety 1. Interaction of electrons-e with the matter −31 me = 9.11×10 kg ; E = m ec2 = 0.511 MeV; qe = -e 2. Interaction of photons-γ with the matter mγ = 0 kg ; E γ = 0 eV; q γ = 0 3. Interaction of neutrons-n with the matter −27 mn = 1.68 × 10 kg ; En = 939.57 MeV; qn = 0 4. Interaction of protons-p with the matter −27 mp = 1.67 × 10 kg ; Ep = 938.27 MeV; qp = +e Note: for any nucleus A: mass number – nucleons number A = Z + N Z: atomic number – proton (charge) number N: neutron number 1 / 35 1. Interaction of electrons with the matter Radiation Safety The physical processes: 1. Ionization losses inelastic collisions with orbital electrons 2. Bremsstrahlung losses inelastic collisions with atomic nuclei 3. Rutherford scattering elastic collisions with atomic nuclei Positrons at nearly rest energy: annihilation emission of two 511 keV photons 2 / 35 1. Interaction of electrons with1.E+02 the matter Radiation Safety ) 1.E+03 -1 Graphite – Z = 6 .g 2 1.E+01 ) Lead – Z = 82 -1 .g 2 1.E+02 1.E+00 1.E+01 1.E-01 1.E+00 collision 1.E-02 radiative collision 1.E-01 stopping pow er (MeV.cm total radiative 1.E-03 stopping pow er (MeV.cm total 1.E-02 1.E-01 1.E+00 1.E+01 1.E+02 1.E+03 1.E-02 energy (MeV) 1.E-02 1.E-01 1.E+00 1.E+01 1.E+02 1.E+03 Electrons – stopping power 1.E+02 energy (MeV) S 1 dE ) === -1 Copper – Z = 29 ρρρ ρρρ dl .g 2 1.E+01 S 1 dE 1 dE === +++ ρρρ ρρρ dl coll ρρρ dl rad 1 dE 1.E+00 : mass stopping power (MeV.cm 2 g. -

Lesson 1: the Single Electron Atom: Hydrogen
Lesson 1: The Single Electron Atom: Hydrogen Irene K. Metz, Joseph W. Bennett, and Sara E. Mason (Dated: July 24, 2018) Learning Objectives: 1. Utilize quantum numbers and atomic radii information to create input files and run a single-electron calculation. 2. Learn how to read the log and report files to obtain atomic orbital information. 3. Plot the all-electron wavefunction to determine where the electron is likely to be posi- tioned relative to the nucleus. Before doing this exercise, be sure to read through the Ins and Outs of Operation document. So, what do we need to build an atom? Protons, neutrons, and electrons of course! But the mass of a proton is 1800 times greater than that of an electron. Therefore, based on de Broglie’s wave equation, the wavelength of an electron is larger when compared to that of a proton. In other words, the wave-like properties of an electron are important whereas we think of protons and neutrons as particle-like. The separation of the electron from the nucleus is called the Born-Oppenheimer approximation. So now we need the wave-like description of the Hydrogen electron. Hydrogen is the simplest atom on the periodic table and the most abundant element in the universe, and therefore the perfect starting point for atomic orbitals and energies. The compu- tational tool we are going to use is called OPIUM (silly name, right?). Before we get started, we should know what’s needed to create an input file, which OPIUM calls a parameter files. Each parameter file consist of a sequence of ”keyblocks”, containing sets of related parameters. -

1.1. Introduction the Phenomenon of Positron Annihilation Spectroscopy
PRINCIPLES OF POSITRON ANNIHILATION Chapter-1 __________________________________________________________________________________________ 1.1. Introduction The phenomenon of positron annihilation spectroscopy (PAS) has been utilized as nuclear method to probe a variety of material properties as well as to research problems in solid state physics. The field of solid state investigation with positrons started in the early fifties, when it was recognized that information could be obtained about the properties of solids by studying the annihilation of a positron and an electron as given by Dumond et al. [1] and Bendetti and Roichings [2]. In particular, the discovery of the interaction of positrons with defects in crystal solids by Mckenize et al. [3] has given a strong impetus to a further elaboration of the PAS. Currently, PAS is amongst the best nuclear methods, and its most recent developments are documented in the proceedings of the latest positron annihilation conferences [4-8]. PAS is successfully applied for the investigation of electron characteristics and defect structures present in materials, magnetic structures of solids, plastic deformation at low and high temperature, and phase transformations in alloys, semiconductors, polymers, porous material, etc. Its applications extend from advanced problems of solid state physics and materials science to industrial use. It is also widely used in chemistry, biology, and medicine (e.g. locating tumors). As the process of measurement does not mostly influence the properties of the investigated sample, PAS is a non-destructive testing approach that allows the subsequent study of a sample by other methods. As experimental equipment for many applications, PAS is commercially produced and is relatively cheap, thus, increasingly more research laboratories are using PAS for basic research, diagnostics of machine parts working in hard conditions, and for characterization of high-tech materials. -

A Young Physicist's Guide to the Higgs Boson
A Young Physicist’s Guide to the Higgs Boson Tel Aviv University Future Scientists – CERN Tour Presented by Stephen Sekula Associate Professor of Experimental Particle Physics SMU, Dallas, TX Programme ● You have a problem in your theory: (why do you need the Higgs Particle?) ● How to Make a Higgs Particle (One-at-a-Time) ● How to See a Higgs Particle (Without fooling yourself too much) ● A View from the Shadows: What are the New Questions? (An Epilogue) Stephen J. Sekula - SMU 2/44 You Have a Problem in Your Theory Credit for the ideas/example in this section goes to Prof. Daniel Stolarski (Carleton University) The Usual Explanation Usual Statement: “You need the Higgs Particle to explain mass.” 2 F=ma F=G m1 m2 /r Most of the mass of matter lies in the nucleus of the atom, and most of the mass of the nucleus arises from “binding energy” - the strength of the force that holds particles together to form nuclei imparts mass-energy to the nucleus (ala E = mc2). Corrected Statement: “You need the Higgs Particle to explain fundamental mass.” (e.g. the electron’s mass) E2=m2 c4+ p2 c2→( p=0)→ E=mc2 Stephen J. Sekula - SMU 4/44 Yes, the Higgs is important for mass, but let’s try this... ● No doubt, the Higgs particle plays a role in fundamental mass (I will come back to this point) ● But, as students who’ve been exposed to introductory physics (mechanics, electricity and magnetism) and some modern physics topics (quantum mechanics and special relativity) you are more familiar with.. -

1 Solving Schrödinger Equation for Three-Electron Quantum Systems
Solving Schrödinger Equation for Three-Electron Quantum Systems by the Use of The Hyperspherical Function Method Lia Leon Margolin 1 , Shalva Tsiklauri 2 1 Marymount Manhattan College, New York, NY 2 New York City College of Technology, CUNY, Brooklyn, NY, Abstract A new model-independent approach for the description of three electron quantum dots in two dimensional space is developed. The Schrödinger equation for three electrons interacting by the logarithmic potential is solved by the use of the Hyperspherical Function Method (HFM). Wave functions are expanded in a complete set of three body hyperspherical functions. The center of mass of the system and relative motion of electrons are separated. Good convergence for the ground state energy in the number of included harmonics is obtained. 1. Introduction One of the outstanding achievements of nanotechnology is construction of artificial atoms- a few-electron quantum dots in semiconductor materials. Quantum dots [1], artificial electron systems realizable in modern semiconductor structures, are ideal physical objects for studying effects of electron-electron correlations. Quantum dots may contain a few two-dimensional (2D) electrons moving in the plane z=0 in a lateral confinement potential V(x, y). Detailed theoretical study of physical properties of quantum-dot atoms, including the Fermi-liquid-Wigner molecule crossover in the ground state with growing strength of intra-dot Coulomb interaction attracted increasing interest [2-4]. Three and four electron dots have been studied by different variational methods [4-6] and some important results for the energy of states have been reported. In [7] quantum-dot Beryllium (N=4) as four Coulomb-interacting two dimensional electrons in a parabolic confinement was investigated. -

One-Electron Atom
One-electron Atom The atomic orbitals of hydrogen-like atoms are solutions to the Schrödinger equation in a spherically symmetric potential. In this case, the potential term is the potential given by Coulomb's law: where •ε0 is the permittivity of the vacuum, •Z is the atomic number (number of protons in the nucleus), •e is the elementary charge (charge of an electron), •r is the distance of the electron from the nucleus. After writing the wave function as a product of functions: (in spherical coordinates), where Ylm are spherical harmonics, we arrive at the following Schrödinger equation: 2010年10月11日星期一 where µ is, approximately, the mass of the electron. More accurately, it is the reduced mass of the system consisting of the electron and the nucleus. Different values of l give solutions with different angular momentum, where l (a non-negative integer) is the quantum number of the orbital angular momentum. The magnetic quantum number m (satisfying ) is the (quantized) projection of the orbital angular momentum on the z-axis. 2010年10月11日星期一 Wave function In addition to l and m, a third integer n > 0, emerges from the boundary conditions placed on R. The functions R and Y that solve the equations above depend on the values of these integers, called quantum numbers. It is customary to subscript the wave functions with the values of the quantum numbers they depend on. The final expression for the normalized wave function is: where: • are the generalized Laguerre polynomials in the definition given here. • Here, µ is the reduced mass of the nucleus-electron system, where mN is the mass of the nucleus. -

1 the Principle of Wave–Particle Duality: an Overview
3 1 The Principle of Wave–Particle Duality: An Overview 1.1 Introduction In the year 1900, physics entered a period of deep crisis as a number of peculiar phenomena, for which no classical explanation was possible, began to appear one after the other, starting with the famous problem of blackbody radiation. By 1923, when the “dust had settled,” it became apparent that these peculiarities had a common explanation. They revealed a novel fundamental principle of nature that wascompletelyatoddswiththeframeworkofclassicalphysics:thecelebrated principle of wave–particle duality, which can be phrased as follows. The principle of wave–particle duality: All physical entities have a dual character; they are waves and particles at the same time. Everything we used to regard as being exclusively a wave has, at the same time, a corpuscular character, while everything we thought of as strictly a particle behaves also as a wave. The relations between these two classically irreconcilable points of view—particle versus wave—are , h, E = hf p = (1.1) or, equivalently, E h f = ,= . (1.2) h p In expressions (1.1) we start off with what we traditionally considered to be solely a wave—an electromagnetic (EM) wave, for example—and we associate its wave characteristics f and (frequency and wavelength) with the corpuscular charac- teristics E and p (energy and momentum) of the corresponding particle. Conversely, in expressions (1.2), we begin with what we once regarded as purely a particle—say, an electron—and we associate its corpuscular characteristics E and p with the wave characteristics f and of the corresponding wave. -

Photon–Photon and Electron–Photon Colliders with Energies Below a Tev Mayda M
Photon–Photon and Electron–Photon Colliders with Energies Below a TeV Mayda M. Velasco∗ and Michael Schmitt Northwestern University, Evanston, Illinois 60201, USA Gabriela Barenboim and Heather E. Logan Fermilab, PO Box 500, Batavia, IL 60510-0500, USA David Atwood Dept. of Physics and Astronomy, Iowa State University, Ames, Iowa 50011, USA Stephen Godfrey and Pat Kalyniak Ottawa-Carleton Institute for Physics Department of Physics, Carleton University, Ottawa, Canada K1S 5B6 Michael A. Doncheski Department of Physics, Pennsylvania State University, Mont Alto, PA 17237 USA Helmut Burkhardt, Albert de Roeck, John Ellis, Daniel Schulte, and Frank Zimmermann CERN, CH-1211 Geneva 23, Switzerland John F. Gunion Davis Institute for High Energy Physics, University of California, Davis, CA 95616, USA David M. Asner, Jeff B. Gronberg,† Tony S. Hill, and Karl Van Bibber Lawrence Livermore National Laboratory, Livermore, CA 94550, USA JoAnne L. Hewett, Frank J. Petriello, and Thomas Rizzo Stanford Linear Accelerator Center, Stanford University, Stanford, California 94309 USA We investigate the potential for detecting and studying Higgs bosons in γγ and eγ collisions at future linear colliders with energies below a TeV. Our study incorporates realistic γγ spectra based on available laser technology, and NLC and CLIC acceleration techniques. Results include detector simulations.√ We study the cases of: a) a SM-like Higgs boson based on a devoted low energy machine with see ≤ 200 GeV; b) the heavy MSSM Higgs bosons; and c) charged Higgs bosons in eγ collisions. 1. Introduction The option of pursuing frontier physics with real photon beams is often overlooked, despite many interesting and informative studies [1]. -

The Basic Interactions Between Photons and Charged Particles With
Outline Chapter 6 The Basic Interactions between • Photon interactions Photons and Charged Particles – Photoelectric effect – Compton scattering with Matter – Pair productions Radiation Dosimetry I – Coherent scattering • Charged particle interactions – Stopping power and range Text: H.E Johns and J.R. Cunningham, The – Bremsstrahlung interaction th physics of radiology, 4 ed. – Bragg peak http://www.utoledo.edu/med/depts/radther Photon interactions Photoelectric effect • Collision between a photon and an • With energy deposition atom results in ejection of a bound – Photoelectric effect electron – Compton scattering • The photon disappears and is replaced by an electron ejected from the atom • No energy deposition in classical Thomson treatment with kinetic energy KE = hν − Eb – Pair production (above the threshold of 1.02 MeV) • Highest probability if the photon – Photo-nuclear interactions for higher energies energy is just above the binding energy (above 10 MeV) of the electron (absorption edge) • Additional energy may be deposited • Without energy deposition locally by Auger electrons and/or – Coherent scattering Photoelectric mass attenuation coefficients fluorescence photons of lead and soft tissue as a function of photon energy. K and L-absorption edges are shown for lead Thomson scattering Photoelectric effect (classical treatment) • Electron tends to be ejected • Elastic scattering of photon (EM wave) on free electron o at 90 for low energy • Electron is accelerated by EM wave and radiates a wave photons, and approaching • No -

Particles and Deep Inelastic Scattering
Heidi Schellman Northwestern Particles and Deep Inelastic Scattering Heidi Schellman Northwestern University HUGS - JLab - June 2010 June 2010 HUGS 1 Heidi Schellman Northwestern k’ k q P P’ A generic scatter of a lepton off of some target. kµ and k0µ are the 4-momenta of the lepton and P µ and P 0µ indicate the target and the final state of the target, which may consist of many particles. qµ = kµ − k0µ is the 4-momentum transfer to the target. June 2010 HUGS 2 Heidi Schellman Northwestern Lorentz invariants k’ k q P P’ 2 2 02 2 2 2 02 2 2 2 The 5 invariant masses k = m` , k = m`0, P = M , P ≡ W , q ≡ −Q are invariants. In addition you can define 3 Mandelstam variables: s = (k + P )2, t = (k − k0)2 and u = (P − k0)2. 2 2 2 2 s + t + u = m` + M + m`0 + W . There are also handy variables ν = (p · q)=M , x = Q2=2Mµ and y = (p · q)=(p · k). June 2010 HUGS 3 Heidi Schellman Northwestern In the lab frame k’ k θ q M P’ The beam k is going in the z direction. Confine the scatter to the x − z plane. µ k = (Ek; 0; 0; k) P µ = (M; 0; 0; 0) 0µ 0 0 0 k = (Ek; k sin θ; 0; k cos θ) qµ = kµ − k0µ June 2010 HUGS 4 Heidi Schellman Northwestern In the lab frame k’ k θ q M P’ 2 2 2 s = ECM = 2EkM + M − m ! 2EkM 2 0 0 2 02 0 t = −Q = −2EkEk + 2kk cos θ + mk + mk ! −2kk (1 − cos θ) 0 ν = (p · q)=M = Ek − Ek energy transfer to target 0 y = (p · q)=(p · k) = (Ek − Ek)=Ek the inelasticity P 02 = W 2 = 2Mν + M 2 − Q2 invariant mass of P 0µ June 2010 HUGS 5 Heidi Schellman Northwestern In the CM frame k’ k q P P’ The beam k is going in the z direction.