Five Types of Basket Cell in the Hippocampal Dentate Gyrus: a Combined Golgi and Electron Microscopic Study
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Plp-Positive Progenitor Cells Give Rise to Bergmann Glia in the Cerebellum
Citation: Cell Death and Disease (2013) 4, e546; doi:10.1038/cddis.2013.74 OPEN & 2013 Macmillan Publishers Limited All rights reserved 2041-4889/13 www.nature.com/cddis Olig2/Plp-positive progenitor cells give rise to Bergmann glia in the cerebellum S-H Chung1, F Guo2, P Jiang1, DE Pleasure2,3 and W Deng*,1,3,4 NG2 (nerve/glial antigen2)-expressing cells represent the largest population of postnatal progenitors in the central nervous system and have been classified as oligodendroglial progenitor cells, but the fate and function of these cells remain incompletely characterized. Previous studies have focused on characterizing these progenitors in the postnatal and adult subventricular zone and on analyzing the cellular and physiological properties of these cells in white and gray matter regions in the forebrain. In the present study, we examine the types of neural progeny generated by NG2 progenitors in the cerebellum by employing genetic fate mapping techniques using inducible Cre–Lox systems in vivo with two different mouse lines, the Plp-Cre-ERT2/Rosa26-EYFP and Olig2-Cre-ERT2/Rosa26-EYFP double-transgenic mice. Our data indicate that Olig2/Plp-positive NG2 cells display multipotential properties, primarily give rise to oligodendroglia but, surprisingly, also generate Bergmann glia, which are specialized glial cells in the cerebellum. The NG2 þ cells also give rise to astrocytes, but not neurons. In addition, we show that glutamate signaling is involved in distinct NG2 þ cell-fate/differentiation pathways and plays a role in the normal development of Bergmann glia. We also show an increase of cerebellar oligodendroglial lineage cells in response to hypoxic–ischemic injury, but the ability of NG2 þ cells to give rise to Bergmann glia and astrocytes remains unchanged. -

A Model of Long-Term Memory Storage in the Cerebellar Cortex: a Possible Role for Plasticity at Parallel Fiber Synapses Onto Stellate͞basket Interneurons
Proc. Natl. Acad. Sci. USA Vol. 94, pp. 14200–14205, December 1997 Psychology A model of long-term memory storage in the cerebellar cortex: A possible role for plasticity at parallel fiber synapses onto stellateybasket interneurons GARRETT T. KENYON† Department of Neurobiology and Anatomy, University of Texas Medical School at Houston, 6431 Fannin, Houston, TX 77030 Edited by Richard F. Thompson, University of Southern California, Los Angeles, CA, and approved October 3, 1997 (received for review April 14, 1997) ABSTRACT By evoking changes in climbing fiber activity, Despite evidence supporting their postulated role in motor movement errors are thought to modify synapses from parallel learning, theoretical arguments suggest that memories stored fibers onto Purkinje cells (pf*Pkj) so as to improve subse- at pf*Pkj synapses would be susceptible to long-term degra- quent motor performance. Theoretical arguments suggest dation as a result of ongoing motor adaptation. First, motor there is an intrinsic tradeoff, however, between motor adap- memories are likely to be distributed across overlapping sets of tation and long-term storage. Assuming a baseline rate of pf*Pkj synapses. If motor memories did not overlap, the motor errors is always present, then repeated performance of storage capacity of pf*Pkj synapses would be greatly reduced any learned movement will generate a series of climbing (12, 17). Second, it is reasonable to assume that the pattern of fiber-mediated corrections. By reshuffling the synaptic synaptic weights necessary to effect any given movement is weights responsible for any given movement, such corrections non-unique. If the activity of a Purkinje cell depends only on will degrade the memories for other learned movements stored the total sum of the synaptic input at any given moment, then in overlapping sets of synapses. -

Cerebellar Granule Cells in Culture
Proc. Nati. Acad. Sci. USA Vol. 83, pp. 4957-4961, July 1986 Neurobiology Cerebellar granule cells in culture: Monosynaptic connections with Purkinje cells and ionic currents (excitatory postsynaptic potential/patch-clamp) ToMoo HIRANO, YOSHIHIRo KUBO, AND MICHAEL M. WU Department of Neurobiology, Institute of Brain Research, School of Medicine, University of Tokyo, Tokyo, Japan Communicated by S. Hagiwara, March 6, 1986 ABSTRACT Electrophysiological properties of cerebellar tissue was dissociated by triturating with a fire-polished granule cells and synapses between granule and Purkinje cells Pasteur pipette in Ca-free Hanks' balanced salt solution were studied in dissociated cultures. Electrophysiological prop- containing 0.05% DNase and 12 mM MgSO4. The cells were erties of neurons and synapses in the mammalian central centrifuged at 150 x g at 40C and the pelleted cells were nervous system are best studied in dissociated cell cultures resuspended at a concentration of about 106 cells per ml in a because of good target cell visibility, control over the contents defined medium (9). One milliliter ofthe cell suspension from of the extracellular solution, and the feasibility of whole-cell newborn rats was plated first in a Petri dish (3.5 cm in patch electrode recording, which has been a powerful tech- diameter) containing several pieces ofheat-sterilized, poly(L- nique in analyzing biophysical properties of ionic channels in lysine)-coated coverslips, and then 1 ml of fetal cell suspen- small cells. We have applied this whole-cell recording technique sion was added. One-half of the culture medium was ex- to cultured cerebellar granule cells whose electrophysiological changed with fresh medium once a week. -

FIRST PROOF Cerebellum
Article Number : EONS : 0736 GROSS ANATOMY Cerebellum Cortex The cerebellar cortex is an extensive three-layered sheet with a surface approximately 15 cm laterally THE HUMAN CEREBELLUM (‘‘little brain’’) is a and 180 cm rostrocaudally but densely folded around significant part of the central nervous system both three pairs of nuclei. The cortex is divided into three in size and in neural structure. It occupies approxi- transverse lobes: Anterior and posterior lobes are mately one-tenth of the cranial cavity, sitting astride separated by the primary fissure, and the smaller the brainstem, beneath the occipital cortex, and flocculonodular lobe is separated by the poster- contains more neurons than the whole of the cerebral olateral fissure (Fig. 1). The anterior and posterior cortex. It consists of an extensive cortical sheet, lobes are folded into a number of lobules and further densely folded around three pairs of nuclei. The folded into a series of folia. This transverse organiza- cortex contains only five main neural cell types and is tion is then divided at right angles into broad one of the most regular and uniform structures in the longitudinal regions. The central vermis, named for central nervous system (CNS), with an orthogonal its worm-like appearance, is most obvious in the ‘‘crystalline’’ organization. Major connections are posterior lobe. On either side is the paravermal or made to and from the spinal cord, brainstem, and intermediate cortex, which merges into the lateral sensorimotor areas of the cerebral cortex. hemispheres. The most common causes of damage to the cerebellum are stroke, tumors, or multiple sclerosis. -

NEUROGENESIS in the ADULT BRAIN: New Strategies for Central Nervous System Diseases
7 Jan 2004 14:25 AR AR204-PA44-17.tex AR204-PA44-17.sgm LaTeX2e(2002/01/18) P1: GCE 10.1146/annurev.pharmtox.44.101802.121631 Annu. Rev. Pharmacol. Toxicol. 2004. 44:399–421 doi: 10.1146/annurev.pharmtox.44.101802.121631 Copyright c 2004 by Annual Reviews. All rights reserved First published online as a Review in Advance on August 28, 2003 NEUROGENESIS IN THE ADULT BRAIN: New Strategies for Central Nervous System Diseases ,1 ,2 D. Chichung Lie, Hongjun Song, Sophia A. Colamarino,1 Guo-li Ming,2 and Fred H. Gage1 1Laboratory of Genetics, The Salk Institute, La Jolla, California 92037; email: [email protected], [email protected], [email protected] 2Institute for Cell Engineering, Department of Neurology, Johns Hopkins University School of Medicine, Baltimore, Maryland 21287; email: [email protected], [email protected] Key Words adult neural stem cells, regeneration, recruitment, cell replacement, therapy ■ Abstract New cells are continuously generated from immature proliferating cells throughout adulthood in many organs, thereby contributing to the integrity of the tissue under physiological conditions and to repair following injury. In contrast, repair mechanisms in the adult central nervous system (CNS) have long been thought to be very limited. However, recent findings have clearly demonstrated that in restricted areas of the mammalian brain, new functional neurons are constantly generated from neural stem cells throughout life. Moreover, stem cells with the potential to give rise to new neurons reside in many different regions of the adult CNS. These findings raise the possibility that endogenous neural stem cells can be mobilized to replace dying neurons in neurodegenerative diseases. -

The Stellate Cells of the Rat's Cerebellar Cortex*
Z. Anat. Entwickl.-Gesch. 136, 224--248 (1972) by Springer-Verlag 1972 The Stellate Cells of the Rat's Cerebellar Cortex* Victoria Chan-Palay and Sanford L. Palay Departments of Neurobiology and Anatomy, Harvard Medical School, Boston, Massachusetts Received February 18, 1972 Summary. Stellate cells were studied in rapid Golgi preparations and in electron micro- graphs. These small neurons can be classified on the basis of their position in the molecular layer and the patterns of their dendritic and axonal arborizations as follows: (1) superficial cells with short, contorted dendrites and a circumscribed axonal arbor (upper third of the molecular layer) ; (2) deep stellate cells with radiating, twisted dendrites and with long axons giving rise to thin, varicose collaterals (middle third of the molecular layer); (3) deep stellate cells with similar dendrites and long axons giving collaterals to the basket around the Purkinje cell bodies (middle third of the molecular layer). An important characteristic of the stellate cell axon is that it generates most of its collaterals close to its origin. Even in long axon cells, only a few collaterals issue from the more distant parts of the axon. These forms contrast with the basket cell, which sends out long, straighter dendrites, and an extended axon that first emits branches at some distance from its origin. Furthermore, basket cell axon collaterals are usually stout in contrast to the frail, beaded collaterals of the stellate cell axon. The two cell types are considered to be distinct. In electron micrographs stellate cells display folded nuclei and sparse cytoplasm with the characteristics usual for small neurons. -

Differences in the Developmental Patterns of Three Microtubule-Associated Proteins in the Rat Cerebellum’
0270.8474/85/0504-0977$02.00/O The Journal of Neuroscience Copyright 0 Society for Neuroscience Vol. 5, No. 4, pp. 977-991 Printed in U.S.A. April 1985 Differences in the Developmental Patterns of Three Microtubule-associated Proteins in the Rat Cerebellum’ ROBERT BERNHARDT,2 GERDA HUBER,3 AND ANDREW MATUS Friedrich Miescher-lnstitut, P. 0. Box 2543, CH-4002 Base/, Switzerland Abstract MAPS are potentially important physiological regulators of neuronal differentiation. The developmental distribution patternS of microtubule- It has become clear that microtubules of different tissues and cell associated proteins (MAPS) 1, 2, and 3 were studied using types are characterized by different MAPS. Thus, a category of high three monoclonal antibodies. lmmunochemical staining at the molecular weight MAPS, later identified as MAP2, is exclusively light and electron microscopic levels demonstrated the spe- expressed in adult neurons where it is most concentrated in den- cific localization of each MAP in different cellular and sub- drites (Matus et al., 1981, 1983; Caceres et al., 1983, 1984; Wiche cellular compartments. (i) MAPS, which is specifically asso- et al., 1983; Bernhardt and Matus, 1984; De Camilli et al., 1984). ciated with dendritic microtubules in the adult brain, is strictly This suggested the possibility that MAP2 could be specifically associated with growing dendrites from the onset of their involved in the formation of dendritic microtubules and hence could formation. (ii) MAP3, a recently described MAP of M, = play a significant role in the regulation of dendrite growth. This idea 180,000, which in the adult brain is associated with neurofi- found further support from immunohistochemical data of developing lament-rich axons and glial processes, is associated with brain (Bernhardt and Matus, 1982; Matus et al., 1983; Burgoyne and axons from the beginning of outgrowth. -

The Developmental Genetics of the Cerebellum and the Genetic Bases of Known Cerebellar Disorders
THE DEVELOPMENTAL GENETICS OF THE CEREBELLUM AND THE GENETIC BASES OF KNOWN CEREBELLAR DISORDERS A Review of Developmental Genetics of the Cerebellum and the Genetic Bases of known Cerebellar Disorders Olakada Favour Adamba 17/MHS01/254 Department of Medicine and Health Sciences College of Medicine and Health Sciences Afe Babalola University ANA 303: Neuroanatomy July, 2020 The Developmental Genetics of Cerebellum and the Genetic Bases of known Cerebellar Disorders: A Literature Review Olakada Favour Adamba1 Abstract The internal structure of the cerebellum is an intriguing paradox; its cytoarchitecture is relatively simple compared to the connections between its neurons, which are wired into a complex array of gene expression domains and functional circuits. The genetic research of cerebellar development has provided a great deal of information about the molecular events directing the formation of the cerebellum. The developmental mechanisms that coordinate the establishment of cerebellar structure and circuitry provide a powerful model for understanding how a functional brain develops and its significance in cerebellar disorders and diseases. The cellular makeup of the cerebellum is derived from two primary germinal matrices (the ventricular zone and a specialized germinal matrix called the rhombic lip). Each matrix/zone expresses a specific set of genes that establish the cell lineages within the cerebellar anlage. Then, cohorts of differentiated projection neurons and interneuron progenitors migrate into the developing cerebellum. thereafter, a number of remarkable patterning events occur. Altogether, structural and molecular organisations are thought to support the proper connectivity between incoming afferent projections and their target cells. Key words: Cerebellum, circuitry, genetic, development, disorders. I. Introduction The cerebellum (‘little brain’) resides at the anterior end of the hindbrain and is classically defined by its role in sensory-motor processing (Buckner, 2013). -

Neurogenesis in the Adult Human Hippocampus
1998 Nature America Inc. • http://medicine.nature.com ARTICLES Neurogenesis in the adult human hippocampus PETER S. ERIKSSON1,4, EKATERINA PERFILIEVA1, THOMAS BJÖRK-ERIKSSON2, ANN-MARIE ALBORN1, CLAES NORDBORG3, DANIEL A. PETERSON4 & FRED H. GAGE4 Department of Clinical Neuroscience, Institute of Neurology1, Department of Oncology2, Department of Pathology3, Sahlgrenska University Hospital, 41345 Göteborg, Sweden 4Laboratory of Genetics, The Salk Institute for Biological Studies, 10010 North Torrey Pines Road, La Jolla, California 92037, USA Correspondence should be addressed to F.H.G. The genesis of new cells, including neurons, in the adult human brain has not yet been demon- strated. This study was undertaken to investigate whether neurogenesis occurs in the adult human brain, in regions previously identified as neurogenic in adult rodents and monkeys. Human brain tissue was obtained postmortem from patients who had been treated with the thymidine analog, bromodeoxyuridine (BrdU), that labels DNA during the S phase. Using im- munofluorescent labeling for BrdU and for one of the neuronal markers, NeuN, calbindin or neu- ron specific enolase (NSE), we demonstrate that new neurons, as defined by these markers, are generated from dividing progenitor cells in the dentate gyrus of adult humans. Our results further indicate that the human hippocampus retains its ability to generate neurons throughout life. Loss of neurons is thought to be irreversible in the adult human one intravenous infusion (250 mg; 2.5 mg/ml, 100 ml) of bro- brain, because dying neurons cannot be replaced. This inability modeoxyuridine (BrdU) for diagnostic purposes11. One patient to generate replacement cells is thought to be an important diagnosed with a similar type and location of cancer, but with- http://medicine.nature.com cause of neurological disease and impairment. -
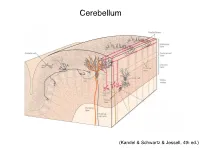
Cerebellum and Activation of the Cerebellum IO ST During Nonmotor Tasks
Cerebellum (Kandel & Schwartz & Jessell, 4th ed.) Granule186 cells are irreducibly smallChapter (6-8 7 μm) stellate cell basket cell outer synaptic layer PC rs cap PC layer grc Go inner 6 μm synaptic layer Figure 7.15 Largest cerebellar neuron occupies more than a 1,000-fold greater volume than smallest neuron. Thin section (~1 μ m) through monkey cerebellar cortex. Purkinje cell body (PC) and nucleus are far larger than those of granule cell (grc). The latter cluster to leave space for mossy fiber terminals to form glomeruli with grc dendritic claws and space for Golgi cells (Go). Note rich network of capillaries (cap). Fine, scat- tered dots are mitochondria. Courtesy of E. Mugnaini. brain ’ s most numerous neuron, this small cost grows large (see also chapter 13). Much of the inner synaptic layer is occupied by the large axon terminals of mossy fibers (figures 7.1 and 7.16). A terminal interlaces with multiple (~15) dendritic claws, each from a different but neighboring granule cell, and forms a complex knot (glomerulus ), nearly as large as a granule cell body (figures 7.1 and 7.16). The mossy fiber axon fires at an unusually high mean rate (up to 200 Hz) and is therefore among the brain’ s thickest (figure 4.6). To match the axon’ s high rate, a terminal expresses 150 active zones, 10 per postsynaptic granule cell (figure 7.16). These sites are capable of driving … and most expensive. 190 Chapter 7 10 4 8 3 9 19 10 10 × 6 × 2 2 4 ATP/s/cell ATP/s/cell ATP/s/m 1 2 0 0 astrocyte astrocyte Golgi cell Golgi cell basket cell basket cell stellate cell stellate cell granule cell granule cell mossy fiber mossy fiber Purkinje cell Purkinje Purkinje cell Purkinje Bergman glia Bergman glia climbing fiber climbing fiber Figure 7.18 Energy costs by cell type. -

Monoclonal Antibodies to Specific Astroglial and Neuronal Antigens Reveal the Cytoarchitecture of the Bergmann Glia Fibers in the Cerebellum’
0270.6474/84/0401-0265$02.00/O The Journal of Neuroscience Copyright 0 Society for Neuroscience Vol. 4, No. 1, pp. 265-273 Printed in U.S.A. January 1984 MONOCLONAL ANTIBODIES TO SPECIFIC ASTROGLIAL AND NEURONAL ANTIGENS REVEAL THE CYTOARCHITECTURE OF THE BERGMANN GLIA FIBERS IN THE CEREBELLUM’ ANGEL L. DE BLAS Department of Neurobiology and Behavior, State University of New York at Stony Brook, Stony Brook, New York 11794 Received May 11, 1983; Revised August 22, 1983; Accepted August 22, 1983 Abstract The cytoarchitecture of the cerebellar Bergmann fibers in the adult rat was investigated. Two monoclonal antibodies, one specific for the Bergmann fibers and astrocyte processes and the other specific for the cell bodies and dendrites of the Purkinje cells as well as an antiserum to the glial fibrillary acidic protein, were used in immunocytochemical peroxidase-antiperoxidase assays. The Bergmann fibers are revealed as columns organized in long vertical palisades parallel to the longitudinal plane of the folium. The palisades are not continuous; instead they are formed by sets of two to six aligned Bergmann fibers. Each of these sets of Bergmann fibers is separated from its longitudinally aligned neighbors by gaps. Each Bergmann fiber is formed by a bundle of two to four Bergmann glia processes which frequently show a helical organization. These results help to reconcile the different views on the organization of the .Bergmann fibers derived from the’studies done with the light microscope versus those done with the electron microscope. The Bergmann glia may play a fundamental role in directing the geometrical organi- zation of the cerebellar constituents. -

Signal Transmission in the Parallel Fiber-Purkinje Cell System Visualized
Proc. Natl. Acad. Sci. USA Vol. 91, pp. 13014-13017, December 1994 Neurobiology Signal transmission in the parallel fiber-Purkinje cell system visualized by high-resolution imaging (rat/brain slice/cerebelium/optical recordings/voltage-sensitive dyes) IVO VRANESIC*, ToSHIO IIJIMA*, MICHINORI ICHIKAWA*, GEN MATSUMOTO*, AND THOMAS KNOPFELtt *Electrotechnical Laboratory, Supermolecular Science Division, 1-1-4 Umezono, Tsukuba, Ibaraki 305, Japan; and tCentral Nervous System Research, K-125.6.12, Ciba, CH-4002 Basel, Switzerland Communicated by Richard F. Thompson, September 19, 1994 ABSTRACT We investigated the synaptic transmission in MATERIALS AND METHODS the parallel fiber-PurkinJe cell system at high spatio-temporal Cerebellar Slices and Recording Setup. Wistar rats (either resolution by using voltage-sensitive dyes and an imaging sex, aged between 16 days and 5 weeks) were anesthetized by system. In rat cerebellar slices, cut in the frontal plane or in a exposure to diethyl ether and then decapitated. Slices were cut plane of the cerebellar surface, local electrical stimulation from the cerebellar vermis in parafrontal planes ("frontal" induced volleys of action potentials in the parallel fibers; slices, 200 ,um) or in parallel to the cerebellar surface ("su- subsequent postsynaptic responses from Purkiaje cells were perficial" slices, 400 ,um) with a Vibroslice (Campden Instru- observed along the volleys' entire trajectories. Furthermore, ments) and left to recover for 30-60 min at 32°C in continu- the formation of an ordered spatial gradient in parallel fiber ously oxygenated (95%02/5% C02) artificial cerebrospinal conduction velocity across the depth of the molecular layer fluid (ACSF, 147 mm Na+/5 mM K+/2 mM Ca2+/2 mM during postnatal development was observed.