WHO Pharmaceuticals Newsletters
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

“Amenalief® Tab. 200Mg” in Japan
News Release July 3, 2017 Maruho Co., Ltd. Maruho receives manufacturing and marketing approval for Anti-Herpes Virus Agent “Amenalief® Tab. 200mg” in Japan Osaka (Japan), July 3, 2017 – Maruho Co., Ltd (“Maruho”, Head Office: Osaka, Japan, President and CEO: Koichi Takagi) announces that today it has received manufacturing and marketing approval from the Japanese Ministry of Health, Labor and Welfare (MHLW), for anti-herpes virus agent “Amenalief® Tab. 200mg” (INN: amenamevir) (hereinafter referred to as “the product”) for the treatment of herpes zoster (shingles) in Japan. The product is an anti-herpes virus agent with a novel mechanism of action created by Astellas Pharma Inc. (“Astellas”; Head Office: Tokyo, President and CEO: Yoshihiko Hatanaka). The product has been observed to inhibit the proliferation of the varicella-zoster virus (hereinafter VZV) by inhibiting the activity of the helicase-primase complex, which is essential for viral DNA replication. In August 2012, Maruho and Astellas agreed on a license agreement for the development and commercialization of the product in Japan, and Maruho has been progressing its development. Herpes zoster (shingles) is a disease caused by reactivation of the chickenpox VZV in latently-infected nerve ganglia. The main treatment for shingles is anti-herpes virus agents. The product is proven to be effective against VZV when administered once a day after meals. Also, since most of the product is excreted in feces, it is not necessary to adjust the dosage and administration according to creatinine clearance, an indicator of kidney function. Maruho hopes the product will contribute to the expansion of treatment options and improvement in adherence for the treatment of shingles in Japan. -
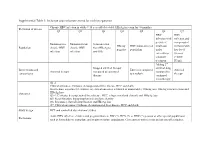
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

AHFS Pharmacologic-Therapeutic Classification System
AHFS Pharmacologic-Therapeutic Classification System Abacavir 48:24 - Mucolytic Agents - 382638 8:18.08.20 - HIV Nucleoside and Nucleotide Reverse Acitretin 84:92 - Skin and Mucous Membrane Agents, Abaloparatide 68:24.08 - Parathyroid Agents - 317036 Aclidinium Abatacept 12:08.08 - Antimuscarinics/Antispasmodics - 313022 92:36 - Disease-modifying Antirheumatic Drugs - Acrivastine 92:20 - Immunomodulatory Agents - 306003 4:08 - Second Generation Antihistamines - 394040 Abciximab 48:04.08 - Second Generation Antihistamines - 394040 20:12.18 - Platelet-aggregation Inhibitors - 395014 Acyclovir Abemaciclib 8:18.32 - Nucleosides and Nucleotides - 381045 10:00 - Antineoplastic Agents - 317058 84:04.06 - Antivirals - 381036 Abiraterone Adalimumab; -adaz 10:00 - Antineoplastic Agents - 311027 92:36 - Disease-modifying Antirheumatic Drugs - AbobotulinumtoxinA 56:92 - GI Drugs, Miscellaneous - 302046 92:20 - Immunomodulatory Agents - 302046 92:92 - Other Miscellaneous Therapeutic Agents - 12:20.92 - Skeletal Muscle Relaxants, Miscellaneous - Adapalene 84:92 - Skin and Mucous Membrane Agents, Acalabrutinib 10:00 - Antineoplastic Agents - 317059 Adefovir Acamprosate 8:18.32 - Nucleosides and Nucleotides - 302036 28:92 - Central Nervous System Agents, Adenosine 24:04.04.24 - Class IV Antiarrhythmics - 304010 Acarbose Adenovirus Vaccine Live Oral 68:20.02 - alpha-Glucosidase Inhibitors - 396015 80:12 - Vaccines - 315016 Acebutolol Ado-Trastuzumab 24:24 - beta-Adrenergic Blocking Agents - 387003 10:00 - Antineoplastic Agents - 313041 12:16.08.08 - Selective -

Herpes Simplex Virus
HSV Herpes simplex virus HSV (Herpes simplex virus) can be spread when an infected person is producing and shedding the virus. Herpes simplex can be spread through contact with saliva, such as sharing drinks. Symptoms of herpes simplex virus infection include watery blisters in the skin or mucous membranes of the mouth, lips or genitals. Lesions heal with ascab characteristic of herpetic disease. As neurotropic and neuroinvasive viruses, HSV-1 and -2 persist in the body by becoming latent and hiding from the immune system in the cell bodies of neurons. After the initial or primary infection, some infected people experience sporadic episodes of viral reactivation or outbreaks. www.MedChemExpress.com 1 HSV Inhibitors (Z)-Capsaicin 1-Docosanol (Zucapsaicin; Civamide; cis-Capsaicin) Cat. No.: HY-B1583 (Behenyl alcohol) Cat. No.: HY-B0222 (Z)-Capsaicin is the cis isomer of capsaicin, acts 1-Docosanol is a saturated fatty alcohol used as an orally active TRPV1 agonist, and is used in traditionally as an emollient, emulsifier, and the research of neuropathic pain. thickener in cosmetics, and nutritional supplement; inhibitor of lipid-enveloped viruses including herpes simplex. Purity: 99.96% Purity: ≥98.0% Clinical Data: Launched Clinical Data: Launched Size: 10 mM × 1 mL, 10 mg, 50 mg Size: 500 mg 2-Deoxy-D-glucose 20(R)-Ginsenoside Rh2 (2-DG; 2-Deoxy-D-arabino-hexose; D-Arabino-2-deoxyhexose) Cat. No.: HY-13966 Cat. No.: HY-N1401 2-Deoxy-D-glucose is a glucose analog that acts as 20(R)-Ginsenoside Rh2, a matrix a competitive inhibitor of glucose metabolism, metalloproteinase (MMP) inhibitor, acts as a inhibiting glycolysis via its actions on hexokinase. -

WHO Drug Information Vol
WHO Drug Information Vol. 33, No. 2, 2019 WHO Drug Information Contents Publication News 135 53rd report of the World Health Organization (WHO) Expert Committee on Specifications for Pharmaceutical Preparations (ECSPP) Consultation Documents 139 Concept Note: A Framework for Evaluating and Publicly Designating Regulatory Authorities as WHO-Listed Authorities 159 Quality Management System Requirements for National Inspectorates 174 Procedure for the Elaboration, Revision and Omission of Monographs and Other Texts for The International Pharmacopoeia 179 Good Chromatography Practices 194 Good Storage and Distribution Practices for Medical Products ATC/DD Classification 226 ATC/DDD Classification (Temporary) 229 ATC/DDD Classification (Final) International Nonproprietary Names (INN) 233 List No. 121 of Proposed International Nonproprietary Names (INN) for Pharmaceutical Substances Abbreviations and websites CHMP Committee for Medicinal Products for Human Use (EMA) EMA European Medicines Agency (www.ema.europa.eu) EU European Union FDA U.S. Food and Drug Administration (www.fda.gov) Health Canada Federal department responsible for health product regulation in Canada (www.hc-sc.gc.ca) HPRA Health Products Regulatory Authority, Ireland (www.hpra.ie) HSA Health Sciences Authority, Singapore (www.hsa.gov.sg) ICDRA International Conference of Drug Regulatory Authorities ICH International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (www.ich.org) IGDRP International Generic Drug Regulators Programme (https://www.igdrp.com) -

S41598-020-80397-9.Pdf
www.nature.com/scientificreports OPEN Activation of PKC supports the anticancer activity of tigilanol tiglate and related epoxytiglianes Jason K. Cullen1, Glen M. Boyle1,2,3,7*, Pei‑Yi Yap1, Stefan Elmlinger1, Jacinta L. Simmons1, Natasa Broit1, Jenny Johns1, Blake Ferguson1, Lidia A. Maslovskaya1,4, Andrei I. Savchenko4, Paul Malek Mirzayans4, Achim Porzelle4, Paul V. Bernhardt4, Victoria A. Gordon5, Paul W. Reddell5, Alberto Pagani6, Giovanni Appendino6, Peter G. Parsons1,3 & Craig M. Williams4* The long‑standing perception of Protein Kinase C (PKC) as a family of oncoproteins has increasingly been challenged by evidence that some PKC isoforms may act as tumor suppressors. To explore the hypothesis that activation, rather than inhibition, of these isoforms is critical for anticancer activity, we isolated and characterized a family of 16 novel phorboids closely‑related to tigilanol tiglate (EBC‑46), a PKC‑activating epoxytigliane showing promising clinical safety and efcacy for intratumoral treatment of cancers. While alkyl branching features of the C12‑ester infuenced potency, the 6,7‑epoxide structural motif and position was critical to PKC activation in vitro. A subset of the 6,7‑epoxytiglianes were efcacious against established tumors in mice; which generally correlated with in vitro activation of PKC. Importantly, epoxytiglianes without evidence of PKC activation showed limited antitumor efcacy. Taken together, these fndings provide a strong rationale to reassess the role of PKC isoforms in cancer, and suggest in some situations their activation can be a promising strategy for anticancer drug discovery. Te Protein Kinase C (PKC) family of serine/threonine kinases was frst identifed almost 40 years ago1. -

The Effect of Wool Hydrolysates on Squamous Cell Carcinoma Cells in Vitro
RESEARCH ARTICLE The effect of wool hydrolysates on squamous cell carcinoma cells in vitro. Possible implications for cancer treatment Tatsiana Damps1*, Anna Katarzyna Laskowska1, Tomasz Kowalkowski2,3, Monika Prokopowicz4, Anna Katarzyna Puszko5, Piotr Sosnowski1, Joanna Czuwara6, Marek Konop6, Krzysztof RoÂżycki4, Joanna Karolina Borkowska7, Aleksandra Misicka1, Lidia Rudnicka1 1 Department of Neuropeptides, Mossakowski Medical Research Centre, Polish Academy of Sciences, Warsaw, Poland, 2 Chair of Environmental Chemistry and Bioanalytics, Faculty of Chemistry, Nicolaus a1111111111 Copernicus University, Toruń, Poland, 3 Interdisciplinary Centre of Modern Technology, Nicolaus Copernicus a1111111111 University, Toruń, Poland, 4 Laboratory of Chemical Synthesis, CePT, Mossakowski Medical Research a1111111111 Centre, Polish Academy of Sciences, Warsaw, Poland, 5 Faculty of Chemistry, University of Warsaw, a1111111111 Warsaw, Poland, 6 Department of Dermatology, Medical University of Warsaw, Warsaw, Poland, 7 Department of Human Epigenetics, Mossakowski Medical Research Centre, Polish Academy of Sciences, a1111111111 Warsaw, Poland * [email protected] OPEN ACCESS Abstract Citation: Damps T, Laskowska AK, Kowalkowski T, Prokopowicz M, Puszko AK, Sosnowski P, et al. Squamous cell carcinoma of the skin is the second most common cutaneous malignancy. (2017) The effect of wool hydrolysates on Despite various available treatment methods and advances in noninvasive diagnostic tech- squamous cell carcinoma cells in vitro. Possible niques, the incidence of metastatic cutaneous squamous cell carcinoma is rising. Deficiency implications for cancer treatment. PLoS ONE 12 (8): e0184034. https://doi.org/10.1371/journal. in effective preventive or treatment methods of transformed keratinocytes leads to necessity pone.0184034 of searching for new anticancer agents. The present study aims to evaluate the possibility of Editor: Andrzej T Slominski, University of Alabama using wool hydrolysates as such agents. -
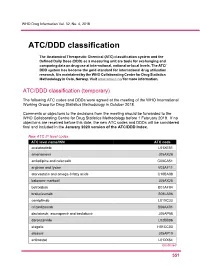
ATC/DDD Classification
WHO Drug Information Vol. 32, No. 4, 2018 ATC/DDD classification The Anatomical Therapeutic Chemical (ATC) classification system and the Defined Daily Dose (DDD) as a measuring unit are tools for exchanging and comparing data on drug use at international, national or local levels. The ATC/ DDD system has become the gold standard for international drug utilization research. It is maintained by the WHO Collaborating Centre for Drug Statistics Methodology in Oslo, Norway. Visit www.whocc.no/ for more information. ATC/DDD classification (temporary) The following ATC codes and DDDs were agreed at the meeting of the WHO International Working Group for Drug Statistics Methodology in October 2018. Comments or objections to the decisions from the meeting should be forwarded to the WHO Collaborating Centre for Drug Statistics Methodology before 1 February 2019. If no objections are received before this date, the new ATC codes and DDDs will be considered final and included in the January 2020 version of the ATC/DDD Index. New ATC 5th level codes ATC level name/INN ATC code acalabrutinib L01XE51 amenamevir J05AX26 amlodipine and celecoxib C08CA51 arginine and lysine V03AF11 atorvastatin and omega-3 fatty acids C10BA08 baloxavir marboxil J05AX25 betrixaban B01AF04 brolucizumab S01LA06 cemiplimab L01XC33 crizanlizumab B06AX01 daclatasvir, asunaprevir and beclabuvir J05AP58 darolutamide L02BB06 elagolix H01CC03 elbasvir J05AP10 entinostat L01XX64 Continued 5 51 ATC/DDD classification WHO Drug Information Vol. 32, No. 4, 2018 New ATC 5th level codes (continued) -

Report on the Deliberation Results June 7, 2017 Pharmaceutical
Report on the Deliberation Results June 7, 2017 Pharmaceutical Evaluation Division, Pharmaceutical Safety and Environmental Health Bureau Ministry of Health, Labour and Welfare Brand Name Amenalief Tab. 200 mg Non-proprietary Name Amenamevir (JAN*) Applicant Maruho Co., Ltd. Date of Application April 27, 2016 Results of Deliberation In its meeting held on May 30, 2017, the Second Committee on New Drugs concluded that the product may be approved and that this result should be presented to the Pharmaceutical Affairs Department of the Pharmaceutical Affairs and Food Sanitation Council. The product is not classified as a biological product or a specified biological product. The re- examination period is 8 years. Neither the drug product nor its drug substance is classified as a poisonous drug or a powerful drug. Condition of Approval The applicant is required to develop and appropriately implement a risk management plan. *Japanese Accepted Name (modified INN) This English translation of this Japanese review report is intended to serve as reference material made available for the convenience of users. In the event of any inconsistency between the Japanese original and this English translation, the Japanese original shall take precedence. PMDA will not be responsible for any consequence resulting from the use of this reference English translation. Review Report May 8, 2017 Pharmaceuticals and Medical Devices Agency The following are the results of the review of the following pharmaceutical product submitted for marketing approval conducted by the Pharmaceuticals and Medical Devices Agency (PMDA). Brand Name Amenalief Tab. 200 mg Non-proprietary Name Amenamevir Applicant Maruho Co., Ltd. Date of Application April 27, 2016 Dosage Form/Strength Tablets: Each tablet contains 200 mg of Amenamevir. -

2021 Formulary List of Covered Prescription Drugs
2021 Formulary List of covered prescription drugs This drug list applies to all Individual HMO products and the following Small Group HMO products: Sharp Platinum 90 Performance HMO, Sharp Platinum 90 Performance HMO AI-AN, Sharp Platinum 90 Premier HMO, Sharp Platinum 90 Premier HMO AI-AN, Sharp Gold 80 Performance HMO, Sharp Gold 80 Performance HMO AI-AN, Sharp Gold 80 Premier HMO, Sharp Gold 80 Premier HMO AI-AN, Sharp Silver 70 Performance HMO, Sharp Silver 70 Performance HMO AI-AN, Sharp Silver 70 Premier HMO, Sharp Silver 70 Premier HMO AI-AN, Sharp Silver 73 Performance HMO, Sharp Silver 73 Premier HMO, Sharp Silver 87 Performance HMO, Sharp Silver 87 Premier HMO, Sharp Silver 94 Performance HMO, Sharp Silver 94 Premier HMO, Sharp Bronze 60 Performance HMO, Sharp Bronze 60 Performance HMO AI-AN, Sharp Bronze 60 Premier HDHP HMO, Sharp Bronze 60 Premier HDHP HMO AI-AN, Sharp Minimum Coverage Performance HMO, Sharp $0 Cost Share Performance HMO AI-AN, Sharp $0 Cost Share Premier HMO AI-AN, Sharp Silver 70 Off Exchange Performance HMO, Sharp Silver 70 Off Exchange Premier HMO, Sharp Performance Platinum 90 HMO 0/15 + Child Dental, Sharp Premier Platinum 90 HMO 0/20 + Child Dental, Sharp Performance Gold 80 HMO 350 /25 + Child Dental, Sharp Premier Gold 80 HMO 250/35 + Child Dental, Sharp Performance Silver 70 HMO 2250/50 + Child Dental, Sharp Premier Silver 70 HMO 2250/55 + Child Dental, Sharp Premier Silver 70 HDHP HMO 2500/20% + Child Dental, Sharp Performance Bronze 60 HMO 6300/65 + Child Dental, Sharp Premier Bronze 60 HDHP HMO -

FOI 073-1718 Document 3
Document 3 PEP005 (ingenol mebutate) Gel 0000 2.7.4 Summary of Clinical Safety CONFIDENTIAL PEP005 (ingenol mebutate) Gel 2.7.4 Summary of Clinical Safety for Actinic Keratosis LEO Pharma A/S Clinical Development 29 May 2011 THIS DOCUMENT CONTAINS TRADE SECRETS, OR COMMERCIAL OR FINANCIAL INFORMATION, PRIVILEGED OR CONFIDENTIAL, DELIVERED IN CONFIDENCE AND RELIANCE THAT SUCH INFORMATION WILL NOT BE COPIED OR MADE AVAILABLE TO ANY THIRD PARTY WITHOUT THE WRITTEN CONSENT OF LEO PHARMA A/S - LEO PHARMACEUTICAL PRODUCTS LTD. A/S 00255345v 2.0 PEP005 (ingenol mebutate) Gel 0000 2.7.4 Summary of Clinical Safety PEP005 (ingenol mebutate) Gel Page 2 of 168 2.7.4 Summary of Clinical Safety for Actinic Keratosis 29 May 2011 Module 2 Summary of Clinical Safety TABLE OF CONTENTS TABLE OF CONTENTS ......................................................................................................... 2 TABLE OF TABLES ............................................................................................................... 5 TABLE OF FIGURES ............................................................................................................. 8 TABLE OF ATTACHMENTS ............................................................................................... 9 LIST OF ABBREVIATIONS AND DEFINITIONS OF TERMS ..................................... 10 1 EXPOSURE TO THE DRUG ............................................................................................ 12 1.1 OVERALL SAFETY EVAULATION PLAN AND NARRATIVES OF SAFETY STUDIES ......................................................................................................................... -

Current Drugs to Treat Infections with Herpes Simplex Viruses-1 and -2
viruses Review Current Drugs to Treat Infections with Herpes Simplex Viruses-1 and -2 Lauren A. Sadowski 1,†, Rista Upadhyay 1,2,†, Zachary W. Greeley 1,‡ and Barry J. Margulies 1,3,* 1 Towson University Herpes Virus Lab, Department of Biological Sciences, Towson University, Towson, MD 21252, USA; [email protected] (L.A.S.); [email protected] (R.U.); [email protected] (Z.W.G.) 2 Towson University Department of Chemistry, Towson, MD 21252, USA 3 Molecular Biology, Biochemistry, and Bioinformatics Program, Towson University, Towson, MD 21252, USA * Correspondence: [email protected] † Authors contributed equally to this manuscript. ‡ Current address: Becton-Dickinson, Sparks, MD 21152, USA. Abstract: Herpes simplex viruses-1 and -2 (HSV-1 and -2) are two of the three human alphaher- pesviruses that cause infections worldwide. Since both viruses can be acquired in the absence of visible signs and symptoms, yet still result in lifelong infection, it is imperative that we provide interventions to keep them at bay, especially in immunocompromised patients. While numerous experimental vaccines are under consideration, current intervention consists solely of antiviral chemotherapeutic agents. This review explores all of the clinically approved drugs used to prevent the worst sequelae of recurrent outbreaks by these viruses. Keywords: acyclovir; ganciclovir; cidofovir; vidarabine; foscarnet; amenamevir; docosanol; nelfi- navir; HSV-1; HSV-2 Citation: Sadowski, L.A.; Upadhyay, R.; Greeley, Z.W.; Margulies, B.J. Current Drugs to Treat Infections 1. Introduction with Herpes Simplex Viruses-1 and -2. The world of anti-herpes simplex (anti-HSV) agents took flight in 1962 with the FDA Viruses 2021, 13, 1228.