Insect Egg-Killing: a New Front on the Evolutionary Arms-Race Between Brassicaceae Plants And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

(Cruciferae) – Mustard Family
BRASSICACEAE (CRUCIFERAE) – MUSTARD FAMILY Plant: herbs mostly, annual to perennial, sometimes shrubs; sap sometimes peppery Stem: Root: Leaves: mostly simple but sometimes pinnately divided; alternate, rarely opposite or whorled; no stipules Flowers: mostly perfect, mostly regular (actinomorphic); 4 sepals, 4 petals often forming a cross; 6 stamens with usually 2 outer ones shorter than the inner 4; ovary superior, mostly 2 fused carpels, 1 to many ovules, 1 pistil Fruit: seed pods, often used in classification, many are slender and long (Silique), some broad (Silicle) – see morphology slide Other: a large family, many garden plants such as turnip, radish, and cabbage, also some spices; often termed the Cruciferae family; Dicotyledons Group Genera: 350+ genera; 40+ locally WARNING – family descriptions are only a layman’s guide and should not be used as definitive Flower Morphology in the Brassicaceae (Mustard Family) - flower with 4 sepals, 4 petals (often like a cross, sometimes split or lobed), commonly small, often white or yellow, distinctive fruiting structures often important for ID 2 types of fruiting pods: in addition, fruits may be circular, flattened or angled in cross-section Silicle - (usually <2.5x long as wide), 2-valved with septum (replum) Silique - (usually >2.5x long as wide), 2- valved with septum (replum) Flowers, Many Genera BRASSICACEAE (CRUCIFERAE) – MUSTARD FAMILY Sanddune [Western] Wallflower; Erysimum capitatum (Douglas ex Hook.) Greene var. capitatum Wormseed Wallflower [Mustard]; Erysimum cheiranthoides L. (Introduced) Spreading Wallflower [Treacle Mustard]; Erysimum repandum L. (Introduced) Dame’s Rocket [Dame’s Violet]; Hesperis matronalis L. (Introduced) Purple [Violet] Rocket; Iodanthus pinnatifidus (Michx.) Steud. Michaux's Gladecress; Leavenworthia uniflora (Michx.) Britton [Cow; Field] Cress [Peppergrass]; Lepidium campestre L.) Ait. -

Planting Planner
what seeds to sow BRIZA MEDIA Planting ‘GOLDEN BEE’ I love quaking grasses and this selection has a golden tint to the planner seedheads as they age. SLP†. Height (H) 60cm. Sow (S) Best The best seeds to sown in situ in spring. Period of interest (P) sow for 2015 May – September. Winter is the perfect time for studying seed catalogues and planning what to grow. Leading designer Sarah Price gives us a sneak peek of her list WORDS SARAH PRICE PERSICARIA ORIENTALIS Definitely a plant for the back of the border, this tall, multi-branched annual has dramatic, long, deep pink, drooping plumes. Popular in the Victorian era. HA. H 1.8m. S Best sown fresh in autumn. CLOCKWISE FROM TOP: JASON INGRAM X2; STEFFEN HAUSER / GARDEN WORLD IMAGES; NICHOLAS APPLEBY / GARDEN WORLD IMAGES / GARDEN WORLD APPLEBY NICHOLAS IMAGES; / GARDEN WORLD HAUSER X2; STEFFEN INGRAM JASON TOP: FROM CLOCKWISE Can also be sown in early spring. his year my cash-strapped local authority P Late summer to autumn. Twas flooded with unlikely letters of love and appreciation after it set verges and roundabouts alive with a blaze of LATHYRUS annual colour instead of the usual regimented bedding. VERNUS ‘FILIGREE’ It’s proof that broadcasting a packet, or in this case Low-growing, bucketfuls, of mixed seed can have mood-enhancing bushy, hardy effects. It’s not all about live-fast-die-young annuals. perennial sweet pea, Many of my favourite plants are hardy biennials, which invaluable for its take two years to flower, set seed and die, while others are early, purple-blue hardy herbaceous perennials that will live for years. -

Plant Life MagillS Encyclopedia of Science
MAGILLS ENCYCLOPEDIA OF SCIENCE PLANT LIFE MAGILLS ENCYCLOPEDIA OF SCIENCE PLANT LIFE Volume 4 Sustainable Forestry–Zygomycetes Indexes Editor Bryan D. Ness, Ph.D. Pacific Union College, Department of Biology Project Editor Christina J. Moose Salem Press, Inc. Pasadena, California Hackensack, New Jersey Editor in Chief: Dawn P. Dawson Managing Editor: Christina J. Moose Photograph Editor: Philip Bader Manuscript Editor: Elizabeth Ferry Slocum Production Editor: Joyce I. Buchea Assistant Editor: Andrea E. Miller Page Design and Graphics: James Hutson Research Supervisor: Jeffry Jensen Layout: William Zimmerman Acquisitions Editor: Mark Rehn Illustrator: Kimberly L. Dawson Kurnizki Copyright © 2003, by Salem Press, Inc. All rights in this book are reserved. No part of this work may be used or reproduced in any manner what- soever or transmitted in any form or by any means, electronic or mechanical, including photocopy,recording, or any information storage and retrieval system, without written permission from the copyright owner except in the case of brief quotations embodied in critical articles and reviews. For information address the publisher, Salem Press, Inc., P.O. Box 50062, Pasadena, California 91115. Some of the updated and revised essays in this work originally appeared in Magill’s Survey of Science: Life Science (1991), Magill’s Survey of Science: Life Science, Supplement (1998), Natural Resources (1998), Encyclopedia of Genetics (1999), Encyclopedia of Environmental Issues (2000), World Geography (2001), and Earth Science (2001). ∞ The paper used in these volumes conforms to the American National Standard for Permanence of Paper for Printed Library Materials, Z39.48-1992 (R1997). Library of Congress Cataloging-in-Publication Data Magill’s encyclopedia of science : plant life / edited by Bryan D. -

Butterflies & Flowers of the Kackars
Butterflies and Botany of the Kackars in Turkey Greenwings holiday report 14-22 July 2018 Led by Martin Warren, Yiannis Christofides and Yasemin Konuralp White-bordered Grayling © Alan Woodward Greenwings Wildlife Holidays Tel: 01473 254658 Web: www.greenwings.co.uk Email: [email protected] ©Greenwings 2018 Introduction This was the second year of a tour to see the wonderful array of butterflies and plants in the Kaçkar mountains of north-east Turkey. These rugged mountains rise steeply from Turkey’s Black Sea coast and are an extension of the Caucasus mountains which are considered by the World Wide Fund for Nature to be a global biodiversity hotspot. The Kaçkars are thought to be the richest area for butterflies in this range, a hotspot in a hotspot with over 160 resident species. The valley of the River Çoruh lies at the heart of the Kaçkar and the centre of the trip explored its upper reaches at altitudes of 1,300—2,300m. The area consists of steep-sided valleys with dry Mediterranean vegetation, typically with dense woodland and trees in the valley bottoms interspersed with small hay-meadows. In the upper reaches these merge into alpine meadows with wet flushes and few trees. The highest mountain in the range is Kaçkar Dağı with an elevation of 3,937 metres The tour was centred around the two charming little villages of Barhal and Olgunlar, the latter being at the fur- thest end of the valley that you can reach by car. The area is very remote and only accessed by a narrow road that winds its way up the valley providing extraordinary views that change with every turn. -
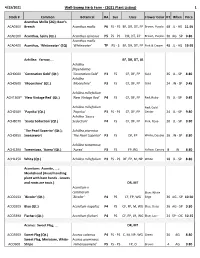
WSHF Catalog
4/23/2021 Well-Sweep Herb Farm - (2021 Plant Listing) 1 Stock # Common Botanical HA Sun Uses Flower ColorHT When Price Acanthus Mollis (2Q); Bear's ACA030X Breech Acanthus mollis P6 FS - PS BF, DR, DT, FP Brown, Purple- 48 JL - AG 11.95 ACA010X Acanthus, Spiny (Qt.) Acanthus spinosus P5 FS - PS DR, DT, FP Brown, Purple- 30 AG - SP 9.80 Acanthus mollis ACA040X Acanthus, `Whitewater' (2Q) `Whitewater' TP PS - S BF, DR, DT, FP Pink & Cream 48 JL - AG 19.95 Achillea: Yarrow, ... BF, DR, DT, LB Achillea filipendulina ACH000X `Coronation Gold' (Qt.) `Coronation Gold' P3 FS CF, DF, FP Gold 36 JL - SP 8.80 Achillea ACH050X `Moonshine' (Qt.) `Moonshine' P3 FS CF, DF, FP Gold 24 JN - SP 9.45 Achillea millefolium ACH130X* `New Vintage Red' (Qt.) `New Vintage Red' P4 FS CF, DF, FP Red, Ruby- 15 JL - SP 9.45 Achillea millefolium Red; Gold ACH250X `Paprika' (Qt.) `Paprika' P3 FS - PS CF, DF, FP Center 24 JL - SP 9.80 Achillea `Saucy ACH807X `Saucy Seduction' (Qt.) Seduction' P4 FS CF, DF, FP Pink, Rose- 20 JL - SP 9.80 `The Pearl Superior' (Qt.); Achillea ptarmica ACH095X Sneezewort `The Pearl Superior' P3 FS DF, FP White; Double 16 JN - SP 8.80 Achillea tomentosa ACH120X Tomentosa, `Aurea' (Qt.) `Aurea' P3 FS FP, RG Yellow, Canary- 8 JN 8.80 ACH125X White (Qt.) Achillea millefolium P3 FS - PS DF, FP, M, NP White 18 JL - SP 8.80 Aconitum: Aconite, ... ; Monkshood (Avoid handling plant with bare hands - Leaves and roots are toxic.) DR, MT Aconitum x cammarum Blue; White ACO015X `Bicolor' (Qt.) `Bicolor' P4 PS CF, FP, WG Edge 36 AG - SP 10.50 ACO020X Blue (Qt.) Aconitum napellus P4 PS CF, FP, M, WG Blue, Deep- 36 AG - SP 9.80 ACO339X Fischeri (Qt.) Aconitum fischeri P4 PS CF, FP, LB, WG Blue, Lav.- 24 SP - OC 10.15 Acorus: Sweet Flag, .. -

Gardenergardener®
Theh American A n GARDENERGARDENER® The Magazine of the AAmerican Horticultural Societyy January / February 2016 New Plants for 2016 Broadleaved Evergreens for Small Gardens The Dwarf Tomato Project Grow Your Own Gourmet Mushrooms contents Volume 95, Number 1 . January / February 2016 FEATURES DEPARTMENTS 5 NOTES FROM RIVER FARM 6 MEMBERS’ FORUM 8 NEWS FROM THE AHS 2016 Seed Exchange catalog now available, upcoming travel destinations, registration open for America in Bloom beautifi cation contest, 70th annual Colonial Williamsburg Garden Symposium in April. 11 AHS MEMBERS MAKING A DIFFERENCE Dale Sievert. 40 HOMEGROWN HARVEST Love those leeks! page 400 42 GARDEN SOLUTIONS Understanding mycorrhizal fungi. BOOK REVIEWS page 18 44 The Seed Garden and Rescuing Eden. Special focus: Wild 12 NEW PLANTS FOR 2016 BY CHARLOTTE GERMANE gardening. From annuals and perennials to shrubs, vines, and vegetables, see which of this year’s introductions are worth trying in your garden. 46 GARDENER’S NOTEBOOK Link discovered between soil fungi and monarch 18 THE DWARF TOMATO PROJECT BY CRAIG LEHOULLIER butterfl y health, stinky A worldwide collaborative breeds diminutive plants that produce seeds trick dung beetles into dispersal role, regular-size, fl avorful tomatoes. Mt. Cuba tickseed trial results, researchers unravel how plants can survive extreme drought, grant for nascent public garden in 24 BEST SMALL BROADLEAVED EVERGREENS Delaware, Lady Bird Johnson Wildfl ower BY ANDREW BUNTING Center selects new president and CEO. These small to mid-size selections make a big impact in modest landscapes. 50 GREEN GARAGE Seed-starting products. 30 WEESIE SMITH BY ALLEN BUSH 52 TRAVELER’S GUIDE TO GARDENS Alabama gardener Weesie Smith championed pagepage 3030 Quarryhill Botanical Garden, California. -

Fragrant Annuals Fragrant Annuals
TheThe AmericanAmerican GARDENERGARDENER® TheThe MagazineMagazine ofof thethe AAmericanmerican HorticulturalHorticultural SocietySociety JanuaryJanuary // FebruaryFebruary 20112011 New Plants for 2011 Unusual Trees with Garden Potential The AHS’s River Farm: A Center of Horticulture Fragrant Annuals Legacies assume many forms hether making estate plans, considering W year-end giving, honoring a loved one or planting a tree, the legacies of tomorrow are created today. Please remember the American Horticultural Society when making your estate and charitable giving plans. Together we can leave a legacy of a greener, healthier, more beautiful America. For more information on including the AHS in your estate planning and charitable giving, or to make a gift to honor or remember a loved one, please contact Courtney Capstack at (703) 768-5700 ext. 127. Making America a Nation of Gardeners, a Land of Gardens contents Volume 90, Number 1 . January / February 2011 FEATURES DEPARTMENTS 5 NOTES FROM RIVER FARM 6 MEMBERS’ FORUM 8 NEWS FROM THE AHS 2011 Seed Exchange catalog online for AHS members, new AHS Travel Study Program destinations, AHS forms partnership with Northeast garden symposium, registration open for 10th annual America in Bloom Contest, 2011 EPCOT International Flower & Garden Festival, Colonial Williamsburg Garden Symposium, TGOA-MGCA garden photography competition opens. 40 GARDEN SOLUTIONS Plant expert Scott Aker offers a holistic approach to solving common problems. 42 HOMEGROWN HARVEST page 28 Easy-to-grow parsley. 44 GARDENER’S NOTEBOOK Enlightened ways to NEW PLANTS FOR 2011 BY JANE BERGER 12 control powdery mildew, Edible, compact, upright, and colorful are the themes of this beating bugs with plant year’s new plant introductions. -

How Reliable Is It?
PROTECTED AREA SITE SELECTION BASED ON ABIOTIC DATA: HOW RELIABLE IS IT? A THESIS SUBMITTED TO THE GRADUATE SCHOOL OF NATURAL AND APPLIED SCIENCES OF MIDDLE EAST TECHNICAL UNIVERSITY BY BANU KAYA ÖZDEMĠREL IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF DOCTOR OF PHILOSOPHY IN BIOLOGY FEBRUARY 2011 Approval of the thesis: PROTECTED AREA SITE SELECTION BASED ON ABIOTIC DATA: HOW RELIABLE IS IT? submitted by BANU KAYA ÖZDEMİREL in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Department of Biological Sciences, Middle East Technical University by, Prof. Dr. Canan Özgen _____________ Dean, Graduate School of Natural and Applied Sciences Prof. Dr. Musa Doğan _____________ Head of Department, Biological Sciences, METU Assoc. Prof. Dr. C. Can Bilgin _____________ Supervisor, Department of Biological Sciences, METU Examining Committee Members: Prof. Dr. Aykut Kence ____________________ Department of Biological Sciences, METU. Assoc. Prof. Dr. C. Can Bilgin ____________________ Department of Biological Sciences, METU. Prof. Dr. Zeki Kaya ____________________ Department of Biological Sciences, METU. Prof. Dr. Nilgül Karadeniz ____________________ Department of Landscape Architecture. AU. Prof. Dr. ġebnem Düzgün ____________________ Department of Mining Engineering. METU. Date: 11.02.2011 I hereby declare that all information in this document has been obtained and presented in accordance with academic rules and ethical conduct. I also declare that, as required by these rules and conduct, I have fully cited and referenced all material and results that are not original to this work. Name, Last Name: Banu Kaya Özdemirel Signature : III ABSTRACT PROTECTED AREA SITE SELECTION BASED ON ABIOTIC DATA: HOW RELIABLE IS IT? Özdemirel Kaya, Banu Ph.D., Department of Biology Supervisor: Assoc. -

Light-Colored Butterflies and Dragonflies Thriving As European Climate Warms 27 May 2014
Light-colored butterflies and dragonflies thriving as European climate warms 27 May 2014 (Pieris mannii), have dispersed to Germany during the last ten years and are still continuing their northward shift. As with lizards and snakes, the colour of an insect's body plays a key role in how they absorb energy from the sun, and is crucial in fuelling their flight as well as regulating their body temperature. Credit: Heiner Blischke Butterflies and dragonflies with lighter colours are out-competing darker-coloured insects in the face of climate change. In a new study published in Nature Communications, scientists from Imperial College London, Philipps-University Marburg and University of Copenhagen have shown that as the climate warms across Europe, communities of The map shows the mean color lightness of dragonflies in Europe with assemblages dominated by light-colored butterflies and dragonflies consist of more lighter species in the south to assemblages dominated by dark- coloured species. Darker coloured species are colored species in the north. This distinct color pattern retreating northwards to cooler areas, but lighter across Europe was unknown until now. Credit: Zeuss et coloured species are also moving their al. 2014. geographical range north as Europe gets warmer. For example, several Mediterranean dragonfly species have expanded their northern range and Dark-coloured insects are able to absorb more immigrated to Germany, such as the Southern sunlight than light-coloured insects, in order to Migrant Hawker (Aeshna affinis), the Scarlet Darter increase their body temperature, and are more (Crocothemis erythraea) and the Dainty Damselfly likely to be found in cooler climates. -

Nomenclature Class Nov2017
What’s in a Name? Cindy Newlander Associate Director of Horticulture Denver Botanic Gardens November 6, 2017 What the Public Sees Family Scientific Name Family Common Name Scientific Name Common Name Special Info: logos, awards, groups Nativity/Range or Hybridizer info What the Public Sees Goals for today: Plant Classification refresher What is a scientific name? Why we use scientific names versus common names? What are some resources for more confident pronunciation? Plant Classification Hierarchy Kingdom Phylum Class Order Family Genus Species King Philip can order fish guts Sunday… Plant Classification Hierarchy Family – approx. 416 (APG IV-2016) Genus – 17,000+ (The Plant List) Species ~ 391,000 vascular plant species named known to science; 369K are Angiosperms, the flowering plants (Kew, 2016) DBG’s Living Collections Statistics 244 Plant Families 2177 Genera 9100+ Species 14,000-15,000 taxa (includes subspecies, varieties, cultivars, grexes) ~23,000 accessions (with +/- 3000 new accessions added yearly) A majority of DBG’s nomenclature data at the species, genus and family level is based on information from The Plant List. Plant Classification Family Useful level for distinguishing characteristics of plants Share obvious traits (anatomical – reproductive features – flowers) Some common families and their members: Rosaceae – rose, hawthorn, pear, apple, raspberry Apiaceae – carrot, parsley, Angelica Asteraceae – yarrow, daisy, Joe Pye, sunflower Brassicaceae – horseradish, broccoli, mustard, wallflower -

Well-Sweep Herb Farm
5/30/2019 Well-Sweep Herb Farm - (2019 Plant Locator) 1 Bed Locati on Common Botanical HA Sun Uses Flower Color HT When Price Acanthus Mollis (2Q); Bear's 18 Breech Acanthus mollis P6 FS - PS BF, DR, DT, FP Brown, Purple- 48 JL - AG 11.95 18 Acanthus, Spiny (Qt.) Acanthus spinosus P5 FS - PS DR, DT, FP Brown, Purple- 30 AG - SP 8.50 Acanthus mollis 18 Acanthus, `Whitewater' (2Q) `Whitewater' TP PS - S BF, DR, DT, FP Pink & Cream 48 JL - AG 22.95 Achillea: Yarrow, ... BF, DR, DT, LB Achillea filipendulina 40 `Coronation Gold' (Qt.) `Coronation Gold' P3 FS CF, DF, FP Gold 36 JL - SP 7.75 40 `Moonshine' (Qt.) Achillea `Moonshine' P3 FS CF, DF, FP Gold 24 JN - SP 8.25 Achillea millefolium Red; Gold 40 `Paprika' (Qt.) `Paprika' P3 FS - PS CF, DF, FP Center 24 JL - SP 7.75 Achillea millefolium 40 `Red Velvet' (Qt.) `Red Velvet' P4 FS CF, DF, FP Red, Dark Rose- 20 JL - SP 8.25 Achillea `Saucy 40 `Saucy Seduction' (Qt.) Seduction' P4 FS CF, DF, FP Pink, Rose- 20 JL - SP 8.50 Achillea `Strawberry Red, Velvet-; 40 `Strawberry Seduction' (Qt.) Seduction' P3 FS CF, DF, FP Gold Center 20 JL - SP 8.50 `The Pearl Superior' (Qt.); Achillea ptarmica 40 Sneezewort `The Pearl Superior' P3 FS DF, FP White; Double 16 JN - SP 7.75 Achillea tomentosa 40 Tomentosa, `Aurea' (Qt.) `Aurea' P3 FS FP, RG Yellow, Canary- 8 JN 7.75 `Well-Sweep Gold' (Qt.) (Best for Achillea filipendulina 40 Dried Flowers) `Well-Sweep Gold' P3 FS CF, DF, FP Gold, Yellow- 24 JL - SP 7.75 40 White (Qt.) Achillea millefolium P3 FS - PS DF, FP, M, NP White 18 JL - SP 7.75 Aconitum: Aconite, .. -

How Much Biodiversity Is in Natura 2000?
Alterra Wageningen UR Alterra Wageningen UR is the research institute for our green living environment. P.O. Box 47 We off er a combination of practical and scientifi c research in a multitude of How much Biodiversity is in Natura 2000? 6700 AA Wageningen disciplines related to the green world around us and the sustainable use of our living The Netherlands environment, such as fl ora and fauna, soil, water, the environment, geo-information The “Umbrella Eff ect” of the European Natura 2000 protected area network T +31 (0) 317 48 07 00 and remote sensing, landscape and spatial planning, man and society. www.wageningenUR.nl/en/alterra The mission of Wageningen UR (University & Research centre) is ‘To explore Technical report Alterra Report 2730B the potential of nature to improve the quality of life’. Within Wageningen UR, ISSN 1566-7197 nine specialised research institutes of the DLO Foundation have joined forces with Wageningen University to help answer the most important questions in the Theo van der Sluis, Ruud Foppen, Simon Gillings, Thomas Groen, René Henkens, Stephan Hennekens, domain of healthy food and living environment. With approximately 30 locations, 6,000 members of staff and 9,000 students, Wageningen UR is one of the leading Kim Huskens, David Noble, Fabrice Ottburg, Luca Santini, Henk Sierdsema, Andre van Kleunen, organisations in its domain worldwide. The integral approach to problems and Joop Schaminee, Chris van Swaay, Bert Toxopeus, Michiel Wallis de Vries and Lawrence Jones-Walters the cooperation between the various disciplines