Critical Role of Emx2 in the Pluripotency – Differentiation Transition in Male Gonocytes Via Regulation of FGF9/NODAL Pathway
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Spatially Heterogeneous Choroid Plexus Transcriptomes Encode Positional Identity and Contribute to Regional CSF Production
The Journal of Neuroscience, March 25, 2015 • 35(12):4903–4916 • 4903 Development/Plasticity/Repair Spatially Heterogeneous Choroid Plexus Transcriptomes Encode Positional Identity and Contribute to Regional CSF Production Melody P. Lun,1,3 XMatthew B. Johnson,2 Kevin G. Broadbelt,1 Momoko Watanabe,4 Young-jin Kang,4 Kevin F. Chau,1 Mark W. Springel,1 Alexandra Malesz,1 Andre´ M.M. Sousa,5 XMihovil Pletikos,5 XTais Adelita,1,6 Monica L. Calicchio,1 Yong Zhang,7 Michael J. Holtzman,7 Hart G.W. Lidov,1 XNenad Sestan,5 Hanno Steen,1 XEdwin S. Monuki,4 and Maria K. Lehtinen1 1Department of Pathology, and 2Division of Genetics, Boston Children’s Hospital, Boston, Massachusetts 02115, 3Department of Pathology and Laboratory Medicine, Boston University School of Medicine, Boston, Massachusetts 02118, 4Department of Pathology and Laboratory Medicine, University of California Irvine School of Medicine, Irvine, California 92697, 5Department of Neurobiology and Kavli Institute for Neuroscience, Yale School of Medicine, New Haven, Connecticut 06510, 6Department of Biochemistry, Federal University of Sa˜o Paulo, Sa˜o Paulo 04039, Brazil, and 7Pulmonary and Critical Care Medicine, Department of Medicine, Washington University, St Louis, Missouri 63110 A sheet of choroid plexus epithelial cells extends into each cerebral ventricle and secretes signaling factors into the CSF. To evaluate whether differences in the CSF proteome across ventricles arise, in part, from regional differences in choroid plexus gene expression, we defined the transcriptome of lateral ventricle (telencephalic) versus fourth ventricle (hindbrain) choroid plexus. We find that positional identitiesofmouse,macaque,andhumanchoroidplexiderivefromgeneexpressiondomainsthatparalleltheiraxialtissuesoforigin.We thenshowthatmolecularheterogeneitybetweentelencephalicandhindbrainchoroidplexicontributestoregion-specific,age-dependent protein secretion in vitro. -

Emx2 Is Required for Growth of the Hippocampus but Not for Hippocampal Field Specification
The Journal of Neuroscience, April 1, 2000, 20(7):2618–2625 Emx2 Is Required for Growth of the Hippocampus But Not for Hippocampal Field Specification Shubha Tole,1 Guy Goudreau,2 Stavroula Assimacopoulos,1 and Elizabeth A. Grove1 1Department of Neurobiology, Pharmacology, and Physiology, University of Chicago, Chicago, Illinois 60637, and 2Max Planck Institute of Biophysical Chemistry, D-37077 Goettingen, Germany The vertebrate Emx genes are expressed in a nested pattern in positioned in the Emx2 mutant. In particular, a dentate cell early embryonic cerebral cortex, such that a medial strip of population is generated, although it fails to form a morpholog- cortex expresses Emx2 but not Emx1. This pattern suggests ical gyrus. This failure may be part of a more widespread medial that Emx genes could play a role in specifying different areas or cortical defect in the mutant. Examination of cortical cell pro- fields of the cortex along the mediolateral axis. Such a role has liferation and differentiation indicates a disruption of the matu- been supported by the observation that in mice lacking func- ration of the medial cortex in the absence of Emx2. Thus, Emx2 tional Emx2 the hippocampus is shrunken and the most medial is required for normal growth and maturation of the hippocam- field of the cortex, the hippocampal dentate gyrus, appears by pus but not for the specification of cells to particular hippocam- cytoarchitecture to be missing (Pellegrini et al., 1996; Yoshida et pal field identities. al., 1997). Use of region-specific molecular markers shows, Key words: Emx2; hippocampus; patterning; specification; however, that hippocampal fields are specified and correctly cortical maturation; cortical hem The hippocampus, like the rest of the cerebral cortex, is divided Reports that specific mutations lead to morphological defects in into cytoarchitectonic areas or fields (Nauta and Feirtag, 1986). -

The Role of Emx1 and Emx2 in the Developing Chick Telencephalon
The Role of Emx1 and Emx2 in the developing chick telencephalon Dissertation Zur Erlangung des Doktorgrades der Naturwissenschaften (Dr.rer.nat.) der Fakultät für Biologie der Ludwig-Maximilian-Universität München Angefertigt am Max-Planck-Institut für Neurobiologie in der Arbeitsgruppe Neuronale Spezifizierung und in der GSF am Institut für Stammzellforschung Julia von Frowein München, Dezember 2004 1. Gutachter: Prof.Dr. Magdalena Götz 2. Gutachter: Prof.Dr. George Boyan eingereicht am 20.12.2004 Tag der mündlichen Prüfung: 26.4.2005 If the brain were so simple we could understand it, we would be so simple we couldn't. Lyall Watson Table of content 1 Table of content 1 Table of content...........................................................................................1 2 Abstract ........................................................................................................5 3 Zusammenfassung......................................................................................6 4 Introduction..................................................................................................8 4.1 General development of the regions of the central nervous system ....................................8 4.2 Patterning and regionalization .............................................................................................8 4.3 Regions of the forebrain.....................................................................................................10 4.4 Migration............................................................................................................................12 -
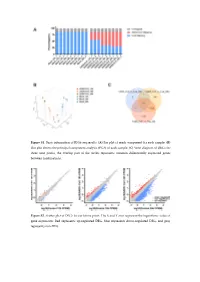
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Conversion of Cerebral Cortex Into Basal Ganglia in Emx2 Pax6 Double
articles Conversion of cerebral cortex into basal ganglia in Emx2 –/– Pax6Sey/Sey double-mutant mice Luca Muzio1, Barbara DiBenedetto1, Anastassia Stoykova2, Edoardo Boncinelli3, Peter Gruss2 and Antonello Mallamaci1 1 Department of Biological and Technological Research (DIBIT), Istituto Scientifico H. San Raffaele, via Olgettina 58, 20132 Milan, Italy 2 Max-Planck Institute of Biophysical Chemistry, Am Fassberg, 37018 Goettingen, Germany 3 International School for Advanced Studies, via Beirut 2/4, 34014 Trieste, Italy Correspondence should be addressed to A.M. ([email protected]) Published online: 15 July 2002, doi:10.1038/nn892 The molecular mechanisms that activate morphogenesis of cerebral cortex are currently the subject of intensive experimental analysis. Transcription factor genes of the homeobox, basic helix-loop- helix (bHLH) and zinc-finger families have recently been shown to have essential roles in this process. However, the actual selector genes activating corticogenesis have not yet been identified. Here we show that high-level expression of at least one functional allele of either of the homeobox genes Emx2 or Pax6 in the dorsal telencephalon is necessary and sufficient to stably activate morphogenesis of cerebral cortex and to repress that of adjacent structures, such as striatum. The molecular mechanisms that trigger morphogenesis of cere- morphologically and molecularly distinguishable from adjacent © http://neurosci.nature.com Group 2002 Nature Publishing bral cortex instead of adjacent telencephalic structures, -

The Doublesex-Related Dmrta2 Safeguards Neural Progenitor
The doublesex-related Dmrta2 safeguards neural PNAS PLUS progenitor maintenance involving transcriptional regulation of Hes1 Fraser I. Younga, Marc Keruzoreb,1, Xinsheng Nanc,1, Nicole Genneta, Eric J. Bellefroidb,2, and Meng Lia,c,2 aNeuroscience and Mental Health Research Institute, School of Medicine, Cardiff University, Cardiff CF24 4HQ, United Kingdom; bInstitute of Neuroscience, Université Libre de Bruxelles, B-6041 Gosselies, Belgium; and cSchool of Bioscience, Cardiff University, Cardiff CF24 4HQ, United Kingdom Edited by Anders Bjorklund, Lund University, Lund, Sweden, and approved May 16, 2017 (received for review March 29, 2017) The mechanisms that determine whether a neural progenitor cell onic brain, however (9, 10). Dmrta2 loss of function in zebrafish (NPC) reenters the cell cycle or exits and differentiates are pivotal for leads to significant reductions in cortical size, coupled with re- generating cells in the correct numbers and diverse types, and thus duced neuronal numbers (10, 11). Likewise, a smaller neocortex, dictate proper brain development. Combining gain-of-function and particularly the dorsomedial neocortex, has been observed in mice loss-of-function approaches in an embryonic stem cell-derived cortical carrying null deletions of Dmrta2 (12–14). Together with the as- differentiation model, we report that doublesex- and mab-3–related sociation of DMRTA2 mutation and microlissencephaly in humans, transcription factor a2 (Dmrta2, also known as Dmrt5) plays an im- these findings implicate Dmrta2 as an important regulator for cortical portant role in maintaining NPCs in the cell cycle. Temporally controlled neurogenesis. expression of transgenic Dmrta2 in NPCs suppresses differentiation Dmrta2-null mice also exhibit agenesis of the embryonic cor- Dmrta2 without affecting their neurogenic competence. -

Perkinelmer Genomics to Request the Saliva Swab Collection Kit for Patients That Cannot Provide a Blood Sample As Whole Blood Is the Preferred Sample
Autism and Intellectual Disability TRIO Panel Test Code TR002 Test Summary This test analyzes 2429 genes that have been associated with Autism and Intellectual Disability and/or disorders associated with Autism and Intellectual Disability with the analysis being performed as a TRIO Turn-Around-Time (TAT)* 3 - 5 weeks Acceptable Sample Types Whole Blood (EDTA) (Preferred sample type) DNA, Isolated Dried Blood Spots Saliva Acceptable Billing Types Self (patient) Payment Institutional Billing Commercial Insurance Indications for Testing Comprehensive test for patients with intellectual disability or global developmental delays (Moeschler et al 2014 PMID: 25157020). Comprehensive test for individuals with multiple congenital anomalies (Miller et al. 2010 PMID 20466091). Patients with autism/autism spectrum disorders (ASDs). Suspected autosomal recessive condition due to close familial relations Previously negative karyotyping and/or chromosomal microarray results. Test Description This panel analyzes 2429 genes that have been associated with Autism and ID and/or disorders associated with Autism and ID. Both sequencing and deletion/duplication (CNV) analysis will be performed on the coding regions of all genes included (unless otherwise marked). All analysis is performed utilizing Next Generation Sequencing (NGS) technology. CNV analysis is designed to detect the majority of deletions and duplications of three exons or greater in size. Smaller CNV events may also be detected and reported, but additional follow-up testing is recommended if a smaller CNV is suspected. All variants are classified according to ACMG guidelines. Condition Description Autism Spectrum Disorder (ASD) refers to a group of developmental disabilities that are typically associated with challenges of varying severity in the areas of social interaction, communication, and repetitive/restricted behaviors. -

Supplementary File Table S1
Supplementary file Table S1 Grouping of Homeobox genes according to their main known function. Anatomical Structure Morphogenesis EN1, HOXC10, HOXC13, HOXD3, LBX1, SIX2, SIX4 Organ Morphogenesis CDX1, CDX2, HOXA11, HOXA13, ISL1, LHX1, PAX3, PDHX, PITX2, PITX3, PROX1, SIX6 Body Pattern Formation ALX3, EMX2, HHEX, HOXA11, HOXA2, HOXA4, HOXA5, HOXA6, HOXB1, HOXB5, HOXB6, HOXC5, HOXD10, HOXD8, LMX1B, PITX2 Ectoderm Development PROX1, VAX2 Endoderm Development HOXC11 Brain & Nervous System Development Brain Development ALX1, DLX2, EMX2 Nervous System Development: ARX, DLX5, DLX6, HOXD10, LBX1, LHX1, OTP, PAX3, PHOX2A, PHOX2B Skeletal Development: ALX3, ALX4, DLX3, DLX5, DLX6, EN1, HOXA11, HOXA13, HOXA2, HOXB6, HOXD10, HOXD13, MSX2 Muscle Development: BARX2, MKX, SIRT1, SIRT2, SIX1 Other Homeobox Genes Involved In BARX1, CDX4, CUX1, DLX1, EMX1, EN2, Multicellular Organismal HOXA1, HOXA7, HOXA9, HOXB13, HOXB2, Development: HOXB3, HOXB4, HOXB7, HOXB8, HOXB9, HOXC12, HOXC8, HOXC9, HOXD1, HOXD11, HOXD12, HOXD9, ISL2, LBX2, LMX1A, MEIS1, NKX3-1, OTX1, TLX1, VAX1, VSX1, VSX2 Homeobox Genes Involved In Cell ARX, EMX2, HHEX, HLX, HOPX, LBX1, LHX1, Differentiation: LMX1B, MIXL1, OTP, PHOX2A, SIRT1, VSX2 Other Genes: PHTF1, SIRT3, SIRT6, SIRT7, ZHX1, ZHX2 Homeobox genes include two subsets of genes coding for transcription factors involved in multiple functions. The clustered HOX genes are indicated in bold. Supplementary file Figure S2 5’ Spatial collinearity 3’ HOXA Chr. 7p15.3 HOXB Chr. 17q21.3 HOXC Chr. 12q13.3 HOXD Chr. 2q31 13 12 11 10 9 8 7 6 5 4 3 2 1 Paralogous HOX groups Distribution of the 39 human HOX genes in four clusters located in different chromosomal regions*. Blue indicates anterior HOX genes. Yellow, paralogy group 3 Hox genes, green and purple indicatete central HOX genes and Red the posterior HOX genes. -

Mouse Otx2 Functions in the Formation and Patterning of Rostral Head
Downloaded from genesdev.cshlp.org on October 3, 2021 - Published by Cold Spring Harbor Laboratory Press Mouse Otx2 functions in the formation and patterning of rostral head Isao Matsuo/ Shigem Kuratani/ Chiham Kimura/'^ Naoki Takeda/ and Shinichi Aizawa^'^ ^Department of Morphogenesis, Institute of Molecular Embryology and Genetics (IMEG), Kumamoto University School of Medicine, Kumamoto 860, Japan; ^Department of Molecular and Cell Genetics, School of Life Sciences, Tottori University, Yonago, Tottori 683, Japan The anterior part of the vertebrate head expresses a group of homeo box genes in segmentally restricted patterns during embryogenesis. Among these, Otx2 expression covers the entire fore- and midbrains and takes place earliest. To examine its role in development of the rostral head, a mutation was introduced into this locus. The homozygous mutants did not develop structures anterior to rhombomere 3, indicating an essential role of Otx2 in the formation of the rostral head. In contrast, heterozygous mutants displayed craniofacial malformations designated as otocephaly; affected structures appeared to correspond to the most posterior and most anterior domains of Otx expression where Otxl is not expressed. The homo- and heterozygous mutant phenotypes suggest Otx2 functions as a gap-like gene in the rostral head where Hox code is not present. The evolutionary significance of Otx2 mutant phenotypes was discussed for the innovation of the neurocranium and the jaw. [Key Words: Otx-, head development; segmental patterning; homeo box; Otocephaly, cranial neural crest] Received May 1, 1995; revised version accepted September 7, 1995. The vertebrate neuraxis can be divided into three re gesting the presence of a genetic code for the brain re gions: the spinal cord, hindbrain, and rostral brain. -

Molecular Mechanisms of Gene Regulation in Response to Environmental and Developmental Stimuli
Molecular Mechanisms of Gene Regulation in Response to Environmental and Developmental Stimuli Dissertation submitted to attain the academic degree “Doctor of Natural Sciences” at the Department of Biology of the Johannes Gutenberg University Mainz by Sandra Schick born on 19.01.1987 in Mainz Mainz, 2016 Dekan: 1. Berichterstatter: 2. Berichterstatter: Tag der mündlichen Prüfung: Table of Contents Table of Contents Table of Contents…………………………………………………………………………………………………………….II List of Abbreviations………………………………………………………………………………………………………IV Abstract…………………………………………………………………………………………………………………………...X Zusammenfassung…………………………………………………………………………………………………………XI Acknowledgements…………………………………………………………………………………………..…………..XII 1. Introduction…………………………………………………………………………………………………………………1 1.1 Gene regulatory mechanisms…………………………………………………………………………….....1 1.1.1 The organization of the mammalian genome…………………………………………..…1 1.1.2 The regulation of gene expression…………………………………………………………...…3 1.1.3 Epigenetic mechanisms regulate gene expression…………………………………...…6 1.1.3.1 DNA modifications……………………………………………………………………..…7 1.1.3.2 Histone variants…………………………………………………………………………....9 1.1.3.3 Post-translational histone modifications………………………………….…11 1.1.3.4 Chromatin remodeling………………….………………………………………….…15 1.1.3.5 Non-coding RNAs……………………………………………………………………….17 1.1.3.6 Distal regulatory regions…………………………………………………………….19 1.1.3.7 Chromatin organization……………………………………………………………...21 1.2 The effect of ultraviolet light on cells…………………………………………………………….……23 1.2.1 UV light induces -

Single-Cell Transcriptomics of the Early Developing Mouse Cerebral Cortex Disentangles the Spatial and Temporal Components of Ne
bioRxiv preprint doi: https://doi.org/10.1101/2020.11.27.401398; this version posted November 27, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. 1 Single-cell transcriptomics of the early developing mouse cerebral cortex 2 disentangles the spatial and temporal components of neuronal fate acquisition 3 4 Matthieu X. MOREAU1,2, Yoann SAILLOUR1,2, Andrzej W. CWETSCH1,2, Alessandra PIERANI1,2,* 5 & Frédéric CAUSERET1,2,* 6 7 1- Université de Paris, Imagine Institute, Team Genetics and Development of the Cerebral Cortex, F- 8 75015, Paris, France 9 2- Université de Paris, Institute of Psychiatry and Neuroscience of Paris, INSERM U1266, F-75014, 10 Paris, France 11 12 * Corresponding authors: 13 [email protected] 14 [email protected] 15 16 Abstract 17 In the developing cerebral cortex, how progenitors that seemingly display limited diversity end up in 18 producing a vast array of neurons remains a puzzling question. The prevailing model that recently 19 emerged suggests that temporal maturation of these progenitors is a key driver in the diversification 20 of the neuronal output. However, temporal constrains are unlikely to account for all diversity across 21 cortical regions, especially in the ventral and lateral domains where neuronal types significantly differ 22 from their dorsal neocortical counterparts born at the same time. In this study, we implemented single- 23 cell RNAseq to sample the diversity of progenitors and neurons along the dorso-ventral axis of the 24 early developing pallium. -

EMX2 Is Epigenetically Silenced and Suppresses Growth in Human Lung Cancer
Oncogene (2010) 29, 5969–5975 & 2010 Macmillan Publishers Limited All rights reserved 0950-9232/10 www.nature.com/onc SHORT COMMUNICATION EMX2 is epigenetically silenced and suppresses growth in human lung cancer J Okamoto1,2,7, T Hirata1,2,7, Z Chen1,3, H-M Zhou3, I Mikami1,2,HLi1, A Beltran1, M Johansson4,5, LM Coussens4,5, G Clement1, Y Shi1,6, F Zhang1, K Koizumi2, K Shimizu2, D Jablons1,5 and B He1,5 1Thoracic Oncology Program, Department of Surgery, University of California, San Francisco, CA, USA; 2Department of Surgery, Division of Thoracic Surgery, Nippon Medical School, Tokyo, Japan; 3Department of Biological Sciences and Biotechnology, Tsinghua University, Beijing, China; 4Department of Pathology, University of California, San Francisco, CA, USA; 5Helen Diller Family Comprehensive Cancer Center, University of California, San Francisco, CA, USA and 6Biosciences Division, SRI International, Menlo Park, CA, USA Lung cancer is a common cancer and the leading cause of than 80% of lung cancers are non-small cell lung cancer, cancer-related death worldwide. Aberrant activation of which includes distinct histological subtypes: adenocarci- WNT signaling is implicated in lung carcinogenesis. noma, squamous cell carcinoma and large cell carcinoma EMX2, a human homologue of the Drosophila empty (Travis, 2002). The mainstays of conventional treatments spiracles gene is a homeodomain-containing transcription have offered patients only a limited and generally short- factor. The function of EMX2 has been linked to the WNT term benefit. Overall, 5-year survival has remained at a signaling pathway during embryonic patterning in mice. dismal B15% for over two decades (Travis, 2002; Jemal However, little is known about the role of EMX2 in human et al., 2008).