Overview of Current Application of Artemisinin Derivatives for Malaria Control
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Trypanocidal Activity of Ethanolic Leaf Extract of Andrographis Paniculata
Science World Journal Vol. 15(No 4) 2020 www.scienceworldjournal.org ISSN: 1597-6343 (Online), ISSN: 2756-391X (Print) Published by Faculty of Science, Kaduna State University TRYPANOCIDAL ACTIVITY OF ETHANOLIC LEAF EXTRACT OF ANDROGRAPHIS PANICULATA 1* 2 3 4 Ladan Z., Olanrewaju T. O., Maikaje D.B., and Waziri P. N. Full Length Research Article 1Department of Chemistry, Kaduna State University, Kaduna, Nigeria 2Human African Trypanosomiasis Research Department, Nigerian Institute for Trypanosomiasis Research, Kaduna, Nigeria 3Department of Microbiology, Kaduna State University, Kaduna, Nigeria 4Department of Biochemistry, Kaduna State University, Kaduna, Nigeria *Corresponding Author’s Email Address: [email protected] Phone: 2347030475898 ABSTRACT animal Trypanosomiasis (Achenef and Bekele, 2013; Giordani et Andrographis paniculata used in this study belongs to Acanthacae al., 2016). Various industries are now on the search for sources of family and is commonly known as king of bitters. This study aimed alternative which include synthetic and natural products. Medicinal at evaluating effect of A. paniculata leaf extract on experimental plants remain a major source of alternative medicine and scientific rats infected with Trypanosoma brucei brucei. Cold maceration was investigations have shown that various plants indicate promising used for extraction and the biomass was fractionated in open results for the development of cheaper less toxic drugs for column chromatography. One of the ethanolic fractions obtained treatment and management of trypanosomiasis (Simoben et al., from A. paniculata was evaluated for in vitro and in vivo activities 2018). Andrographis paniculata evaluated in this study belong to using RPMI 1640 cell culture media and experimental wistar rats Acanthacae family of the plant kingdom and commonly known as respectively. -

Folic Acid Antagonists: Antimicrobial and Immunomodulating Mechanisms and Applications
International Journal of Molecular Sciences Review Folic Acid Antagonists: Antimicrobial and Immunomodulating Mechanisms and Applications Daniel Fernández-Villa 1, Maria Rosa Aguilar 1,2 and Luis Rojo 1,2,* 1 Instituto de Ciencia y Tecnología de Polímeros, Consejo Superior de Investigaciones Científicas, CSIC, 28006 Madrid, Spain; [email protected] (D.F.-V.); [email protected] (M.R.A.) 2 Consorcio Centro de Investigación Biomédica en Red de Bioingeniería, Biomateriales y Nanomedicina, 28029 Madrid, Spain * Correspondence: [email protected]; Tel.: +34-915-622-900 Received: 18 September 2019; Accepted: 7 October 2019; Published: 9 October 2019 Abstract: Bacterial, protozoan and other microbial infections share an accelerated metabolic rate. In order to ensure a proper functioning of cell replication and proteins and nucleic acids synthesis processes, folate metabolism rate is also increased in these cases. For this reason, folic acid antagonists have been used since their discovery to treat different kinds of microbial infections, taking advantage of this metabolic difference when compared with human cells. However, resistances to these compounds have emerged since then and only combined therapies are currently used in clinic. In addition, some of these compounds have been found to have an immunomodulatory behavior that allows clinicians using them as anti-inflammatory or immunosuppressive drugs. Therefore, the aim of this review is to provide an updated state-of-the-art on the use of antifolates as antibacterial and immunomodulating agents in the clinical setting, as well as to present their action mechanisms and currently investigated biomedical applications. Keywords: folic acid antagonists; antifolates; antibiotics; antibacterials; immunomodulation; sulfonamides; antimalarial 1. -

Annual Review 2004 Contents Contentsforeword
Annual Review 2004 Contents ContentsForeword ........................................................................................................................... 3 Editor’s Note ..................................................................................................................... 4 Organization Chart ............................................................................................................ 5 Administrative Board .......................................................................................................... 6 Special Events..................................................................................................................... 8 Consultants ...................................................................................................................... 13 Visiting Professors ............................................................................................................ 13 Faculty Board ...................................................................................................................13 Faculty Senate ..................................................................................................................14 Department of Clinical Tropical Medicine ........................................................................ 15 Department of Helminthology ......................................................................................... 22 Department of Medical Entomology ............................................................................... -

Pharmaceutical Composition Comprising Artesunate
(19) TZZ ¥ 4B_T (11) EP 2 322 244 B1 (12) EUROPEAN PATENT SPECIFICATION (45) Date of publication and mention (51) Int Cl.: of the grant of the patent: A61P 33/06 (2006.01) A61K 9/20 (2006.01) 21.11.2012 Bulletin 2012/47 A61K 31/357 (2006.01) A61K 31/505 (2006.01) A61K 31/4965 (2006.01) (21) Application number: 10192206.0 (22) Date of filing: 09.09.2005 (54) Pharmaceutical composition comprising artesunate, sulfamethoxypyrazine and pyrimethamine suitable for the treatment of malaria within one day, in the form of a tablet Pharmazeutische Zusammensetzung enthaltend Artesunat, Sulfamethoxypyrazin und Pyrimethamin, die zur Behandlung von Malaria innerhalb eines Tages geeignet ist, in Form einer Tablette Composition pharmaceutique à base d’artesunate, de sulfaméthoxypyrazine et de pyriméthamine permettant le traitement du palludisme en un jour, sous forme de comprimé (84) Designated Contracting States: (56) References cited: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR • LUXEMBURGER C ET AL: "Single day HU IE IS IT LI LT LU LV MC NL PL PT RO SE SI mefloquine-artesunate combination in the SK TR treatment of multi-drug resistant falciparum malaria", TRANSACTIONS OF THE ROYAL (43) Date of publication of application: SOCIETY OF TROPICAL MEDICINE AND 18.05.2011 Bulletin 2011/20 HYGIENE, ELSEVIER, GB, vol. 88, no. 2, 1 March 1994 (1994-03-01), pages 213-217, XP023097868, (62) Document number(s) of the earlier application(s) in ELSEVIER, GB ISSN: 0035-9203, DOI: accordance with Art. 76 EPC: 10.1016/0035-9203(94)90303-4 [retrieved on 05795966.0 / 1 940 519 1994-03-01] • DAO ET AL: "Fatty food does not alter blood (73) Proprietor: Dafra Pharma N.V. -

Artemether-Lumefantrine (Six-Dose Regimen) for Treating Uncomplicated Falciparum Malaria (Review)
Artemether-lumefantrine (six-dose regimen) for treating uncomplicated falciparum malaria (Review) Omari AAA, Gamble CL, Garner P This is a reprint of a Cochrane review, prepared and maintained by The Cochrane Collaboration and published in The Cochrane Library 2009, Issue 1 http://www.thecochranelibrary.com Artemether-lumefantrine (six-dose regimen) for treating uncomplicated falciparum malaria (Review) Copyright © 2009 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd. TABLE OF CONTENTS HEADER....................................... 1 ABSTRACT ...................................... 1 PLAINLANGUAGESUMMARY . 2 BACKGROUND .................................... 2 OBJECTIVES ..................................... 3 METHODS ...................................... 3 RESULTS....................................... 5 DISCUSSION ..................................... 9 AUTHORS’CONCLUSIONS . 9 ACKNOWLEDGEMENTS . 10 REFERENCES ..................................... 10 CHARACTERISTICSOFSTUDIES . 13 DATAANDANALYSES. 20 Analysis 1.1. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 1 Total failure by day 28. 22 Analysis 1.2. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 2 Total failure by day 14. 23 Analysis 1.3. Comparison 1 Artemether-lumefantrine vs amodiaquine, Outcome 3 Gametocyte carriage on day 14. 23 Analysis 2.1. Comparison 2 Artemether-lumefantrine vs chloroquine plus sulfadoxine-pyrimethamine, Outcome 1 Total failurebyday28. ................................ 24 Analysis 2.2. Comparison 2 Artemether-lumefantrine -

How to Protect Yourself Against Malaria 1 Fig
From our Whitepaper Files: How to > See companion document Protect Yourself Against Malaria World Malaria Risk Chart 2015 Edition Canada 67 Mowat Avenue, Suite 036 Toronto, Ontario M6K 3E3 (416) 652-0137 USA 1623 Military Road, #279 Niagara Falls, New York 14304-1745 (716) 754-4883 New Zealand 206 Papanui Road Christchurch 5 www.iamat.org | [email protected] | Twitter @IAMAT_Travel | Facebook IAMATHealth THE ENEMY area. Of the 460 Anopheles species, approximately 100 can transmit malaria Sunset — the hunt for human blood begins. parasites. From dusk to dawn the female Anopheles, Mosquitoes prey on a variety of hosts — the malaria-carrying mosquito searches for a host humans, monkeys, lizards, birds — carrying to supply her with blood. Blood is an absolute different species of malaria parasites which in necessity for her because it provides the protein turn infect only specific hosts. Of the approxi- needed for the development of her eggs which mately 50 different species of malaria parasites she later deposits in her breeding place. sharing the genetic name Plasmodium, only She has a tiny, elegant body, measuring 5 infect humans: Plasmodium falciparum, from 8 mm to 1 cm. She has dark spots on the killer; Plasmodium vivax; Plasmodium ovale, her wings, three pairs of long, slender legs and Plasmodium malariae and Plasmodium knowlesi. a prominent tubular proboscis with which The latter, a malaria parasite of Old World she draws blood. monkeys, has been identified to infect humans Fig. 1 Female Anopheles mosquito. The Anopheles enters your room at night. in Southeast Asia. In the past this parasite has Image source: World Health Organization You may recognize her by the way she rests been misdiagnosed as Plasmodium malariae. -

Hydroxychloroquine Or Chloroquine for Treating Coronavirus Disease 2019 (COVID-19) – a PROTOCOL for a Systematic Review of Individual Participant Data
Hydroxychloroquine or Chloroquine for treating Coronavirus Disease 2019 (COVID-19) – a PROTOCOL for a systematic review of Individual Participant Data Authors Fontes LE, Riera R, Miranda E, Oke J, Heneghan CJ, Aronson JK, Pacheco RL, Martimbianco ALC, Nunan D BACKGROUND In the face of the pandemic of SARS CoV2, urgent research is needed to test potential therapeutic agents against the disease. Reliable research shall inform clinical decision makers. Currently, there are several studies testing the efficacy and safety profiles of different pharmacological interventions. Among these drugs, we can cite antimalarial, antivirals, biological drugs, interferon, etc. As of 6 April 2020 there are three published reportsand 100 ongoing trials testing hydroxychloroquine/chloroquine alone or in association with other drugs for COVID-19. This prospective systematic review with Individual Participant data aims to assess the rigour of the best-available evidence for hydroxychloroquine or chloroquine as treatment for COVID-19 infection. The PICO framework is: P: adults with COVID-19 infection I: chloroquine or hydroxychloroquine (alone or in association) C: placebo, other active treatments, usual standard care without antimalarials O: efficacy and safety outcomes OBJECTIVES To assess the effects (benefits and harms) of chloroquine or hydroxychloroquine for the treatment of COVID-19 infection. METHODS Criteria for considering studies for this review Types of studies We shall include randomized controlled trials (RCTs) with a parallel design. We intend to include even small trials (<50 participants), facing the urgent need for evidence to respond to the current pandemic. Quasi-randomized, non-randomized, or observational studies will be excluded due to a higher risk of confounding and selection bias (1). -

Quinoline-Based Hybrid Compounds with Antimalarial Activity
molecules Review Quinoline-Based Hybrid Compounds with Antimalarial Activity Xhamla Nqoro, Naki Tobeka and Blessing A. Aderibigbe * Department of Chemistry, University of Fort Hare, Alice Campus, Alice 5700, Eastern Cape, South Africa; [email protected] (X.N.); [email protected] (N.T.) * Correspondence: [email protected] Received: 7 November 2017; Accepted: 11 December 2017; Published: 19 December 2017 Abstract: The application of quinoline-based compounds for the treatment of malaria infections is hampered by drug resistance. Drug resistance has led to the combination of quinolines with other classes of antimalarials resulting in enhanced therapeutic outcomes. However, the combination of antimalarials is limited by drug-drug interactions. In order to overcome the aforementioned factors, several researchers have reported hybrid compounds prepared by reacting quinoline-based compounds with other compounds via selected functionalities. This review will focus on the currently reported quinoline-based hybrid compounds and their preclinical studies. Keywords: 4-aminoquinoline; 8-aminoquinoline; malaria; infectious disease; hybrid compound 1. Introduction Malaria is a parasitic infectious disease that is a threat to approximately half of the world’s population. This parasitic disease is transmitted to humans by a female Anopheles mosquito when it takes a blood meal. Pregnant women and young children in the sub-tropical regions are more prone to malaria infection. In 2015, 214 million infections were reported, with approximately 438,000 deaths and the African region accounted for 90% of the deaths [1–3]. Malaria is caused by five parasites of the genus Plasmodium, but the majority of deaths are caused by Plasmodium falciparum and Plasmodium vivax [1,4–6]. -

Part 4 May 2016 Sulfadoxine/Pyrimethamine 500Mg/25Mg, Tablets, (Guilin Pharmaceutical Co., Ltd), MA066
Artesunate 50mg Tablets + WHOPAR part 4 May 2016 Sulfadoxine/Pyrimethamine 500mg/25mg, Tablets, (Guilin Pharmaceutical Co., Ltd), MA066 SUMMARY OF PRODUCT CHARACTERISTICS 1 Artesunate 50mg Tablets + WHOPAR part 4 May 2016 Sulfadoxine/Pyrimethamine 500mg/25mg, Tablets, (Guilin Pharmaceutical Co., Ltd), MA066 1. NAME OF THE MEDICINAL PRODUCT ARTECOSPE ® 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each artesunate tablet contains 50 mg artesunate, each sulfadoxine/pyrimethamine tablet contains 500 mg sulfadoxine and 25 mg pyrimethamine. Each artesunate tablet contains 15 mg of sucrose. Each sulfadoxine/pyrimethamine tablet contains 20 mg of lactose. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM The artesunate 50 mg tablets are white round tablets debossed with “AS ” on one side and a score line on the other side. The sulfadoxine/pyrimethamine tablets (500/25 mg) are white, round, and debossed with “SP” on both sides, with a score line on one side. 4. CLINICAL PARTICULARS 4.1 Therapeutic indication ARTECOSPE® (artesunate + sulfadoxine/pyrimethamine) is indicated for the treatment of uncomplicated cases of malaria due to Plasmodium falciparum strains which are susceptible to artesunate as well as to sulfadoxine /pyrimethamine. The most recent official guidelines on the appropriate use of antimalarial agents and local information on the prevalence of resistance to antimalarial drugs must be taken into consideration for deciding on the appropriateness of therapy with artesunate tablets and sulfadoxine/pyrimethamine tablets. Official guidance will normally include WHO (http://apps.who.int/iris/bitstream/10665/162441/1/9789241549127_eng.pdf?ua=1&ua=1) and public health authorities’ guidelines (see also sections 4.4 and 5.1). -

Review of Mass Drug Administration and Primaquine
Contents Acknowledgements ...................................................................................................................................... 2 Acronyms ...................................................................................................................................................... 3 Introduction .................................................................................................................................................. 4 Methods ....................................................................................................................................................... 4 Findings ......................................................................................................................................................... 6 Study objectives and design ............................................................................................................ 9 Contextual parameters - endemicity, seasonality, target population .......................................... 10 Outcome measures ....................................................................................................................... 13 Drug regimens ............................................................................................................................... 13 Co-Interventions ............................................................................................................................ 15 Delivery methods and community engagement .......................................................................... -
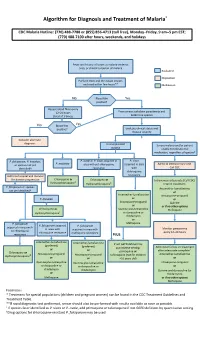
Algorithm for Diagnosis and Treatment of Malaria*
Algorithm for Diagnosis and Treatment of Malaria* CDC Malaria Hotline: (770) 488-7788 or (855) 856-4713 (toll free), Monday–Friday, 9 am–5 pm EST; (770) 488-7100 after hours, weekends, and holidays Fever and history of travel to malaria-endemic area, or clinical suspicion of malaria Evaluation Disposition Perform thick and thin blood smears and read within few hours** Medication No Blood film Yes positive? Repeat blood films every 12–24 hours From smear: calculate parasitemia and (total of 3 times) determine species No Blood film Yes positive? Evaluate clinical status and disease severity Consider alternate diagnosis Uncomplicated Severe malaria and/or patient malaria unable to tolerate oral medication, regardless of species† P. falciparum, P. knowlesi, P. ovale or P. vivax acquired in P. vivax P. malariae or species not yet area without chloroquine acquired in area Admit to intensive care unit. identified† resistance with Call CDC chloroquine Admit to hospital and monitor resistance for disease progression Chloroquine or Chloroquine or ‡ Intravenous artesunate (Call CDC) Hydroxychloroquine Hydroxychloroquine‡ Interim treatment: P. falciparum or species Artemether-lumefantrine not yet identified† or Artemether-lumefantrine Atovaquone-proguanil P. knowlesi or or Atovaquone-proguanil Quinine or or if no other options Chloroquine or Quinine plus tetracycline Mefloquine Hydroxychloroquine‡ or doxycycline or clindamycin or P. falciparum Mefloquine P. falciparum acquired P. falciparum acquired in area with in area with acquired in area with -

1 Antimalarial Activity of the 8 - Aminoquinolines Edward A
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln US Army Research U.S. Department of Defense 1991 1 Antimalarial Activity of the 8 - Aminoquinolines Edward A. Nodiff Franklin Research Center, Arvin Calspan Corporation, Valley Forge Corporate Center, Norristown, PA Sankar Chatterjee Department of Medicinal Chemistry, Division of Experimental Therapeutics, Walter Reed Army Institute of Research, Walter Reed Army Medical Center, Washington, DC Hikmat A. Musallam Department of Medicinal Chemistry, Division of Experimental Therapeutics, Walter Reed Army Institute of Research, Walter Reed Army Medical Center, Washington, DC Follow this and additional works at: http://digitalcommons.unl.edu/usarmyresearch Nodiff, Edward A.; Chatterjee, Sankar; and Musallam, Hikmat A., "1 Antimalarial Activity of the 8 - Aminoquinolines" (1991). US Army Research. 337. http://digitalcommons.unl.edu/usarmyresearch/337 This Article is brought to you for free and open access by the U.S. Department of Defense at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in US Army Research by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. Progress in Medicinal Chemistry - Vol. 28, edited by G.P. Ellis and G.B. West 0 1991, Elsevier Science Publishers, B.V. 1 Antimalarial Activity of the 8- Aminoquinoline s EDWARD A. NODIFF, B.A. SANKAR CHATTERJEE, Ph.D. and HIKMAT A. MUSALLAM, Ph.D.2 Franklin Research Center, Arvin Calspan Corporation, Valley Forge Corporate Center, 2600 Monroe Boulevard, Norristown, PA 19403 and 'Department of Medicinal Chemistry, Division of Experimental Therapeutics, Walter Reed Army Institute of Research, Walter Reed Army Medical Center, Washington, DC 20307-5100, U.S.A. INTRODUCTION 2 THE PARASITE Exoerythrocytic phase Erythrocytic phase Sporogony THE DISEASE 4 CLASSIFICATION OF ANTIMALARIAL DRUGS 4 HISTORY 5 METHODS FOR ANTIMALARIAL DRUG EVALUATION 8 Rane blood schizontocidal screen (P.