10-4 Surface and Lateral Area of Prism and Cylinders.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hydraulic Cylinder Replacement for Cylinders with 3/8” Hydraulic Hose Instructions
HYDRAULIC CYLINDER REPLACEMENT FOR CYLINDERS WITH 3/8” HYDRAULIC HOSE INSTRUCTIONS INSTALLATION TOOLS: INCLUDED HARDWARE: • Knife or Scizzors A. (1) Hydraulic Cylinder • 5/8” Wrench B. (1) Zip-Tie • 1/2” Wrench • 1/2” Socket & Ratchet IMPORTANT: Pressure MUST be relieved from hydraulic system before proceeding. PRESSURE RELIEF STEP 1: Lower the Power-Pole Anchor® down so the Everflex™ spike is touching the ground. WARNING: Do not touch spike with bare hands. STEP 2: Manually push the anchor into closed position, relieving pressure. Manually lower anchor back to the ground. REMOVAL STEP 1: Remove the Cylinder Bottom from the Upper U-Channel using a 1/2” socket & wrench. FIG 1 or 2 NOTE: For Blade models, push bottom of cylinder up into U-Channel and slide it forward past the Ram Spacers to remove it. Upper U-Channel Upper U-Channel Cylinder Bottom 1/2” Tools 1/2” Tools Ram 1/2” Tools Spacers Cylinder Bottom If Ram-Spacers fall out Older models have (1) long & (1) during removal, push short Ram Spacer. Ram Spacers 1/2” Tools bushings in to hold MUST be installed on the same side Figure 1 Figure 2 them in place. they were removed. Blade Models Pro/Spn Models Need help? Contact our Customer Service Team at 1 + 813.689.9932 Option 2 HYDRAULIC CYLINDER REPLACEMENT FOR CYLINDERS WITH 3/8” HYDRAULIC HOSE INSTRUCTIONS STEP 2: Remove the Cylinder Top from the Lower U-Channel using a 1/2” socket & wrench. FIG 3 or 4 Lower U-Channel Lower U-Channel 1/2” Tools 1/2” Tools Cylinder Top Cylinder Top Figure 3 Figure 4 Blade Models Pro/SPN Models STEP 3: Disconnect the UP Hose from the Hydraulic Cylinder Fitting by holding the 1/2” Wrench 5/8” Wrench Cylinder Fitting Base with a 1/2” wrench and turning the Hydraulic Hose Fitting Cylinder Fitting counter-clockwise with a 5/8” wrench. -

An Introduction to Topology the Classification Theorem for Surfaces by E
An Introduction to Topology An Introduction to Topology The Classification theorem for Surfaces By E. C. Zeeman Introduction. The classification theorem is a beautiful example of geometric topology. Although it was discovered in the last century*, yet it manages to convey the spirit of present day research. The proof that we give here is elementary, and its is hoped more intuitive than that found in most textbooks, but in none the less rigorous. It is designed for readers who have never done any topology before. It is the sort of mathematics that could be taught in schools both to foster geometric intuition, and to counteract the present day alarming tendency to drop geometry. It is profound, and yet preserves a sense of fun. In Appendix 1 we explain how a deeper result can be proved if one has available the more sophisticated tools of analytic topology and algebraic topology. Examples. Before starting the theorem let us look at a few examples of surfaces. In any branch of mathematics it is always a good thing to start with examples, because they are the source of our intuition. All the following pictures are of surfaces in 3-dimensions. In example 1 by the word “sphere” we mean just the surface of the sphere, and not the inside. In fact in all the examples we mean just the surface and not the solid inside. 1. Sphere. 2. Torus (or inner tube). 3. Knotted torus. 4. Sphere with knotted torus bored through it. * Zeeman wrote this article in the mid-twentieth century. 1 An Introduction to Topology 5. -

Volumes of Prisms and Cylinders 625
11-4 11-4 Volumes of Prisms and 11-4 Cylinders 1. Plan Objectives What You’ll Learn Check Skills You’ll Need GO for Help Lessons 1-9 and 10-1 1 To find the volume of a prism 2 To find the volume of • To find the volume of a Find the area of each figure. For answers that are not whole numbers, round to prism a cylinder the nearest tenth. • To find the volume of a 2 Examples cylinder 1. a square with side length 7 cm 49 cm 1 Finding Volume of a 2. a circle with diameter 15 in. 176.7 in.2 . And Why Rectangular Prism 3. a circle with radius 10 mm 314.2 mm2 2 Finding Volume of a To estimate the volume of a 4. a rectangle with length 3 ft and width 1 ft 3 ft2 Triangular Prism backpack, as in Example 4 2 3 Finding Volume of a Cylinder 5. a rectangle with base 14 in. and height 11 in. 154 in. 4 Finding Volume of a 6. a triangle with base 11 cm and height 5 cm 27.5 cm2 Composite Figure 7. an equilateral triangle that is 8 in. on each side 27.7 in.2 New Vocabulary • volume • composite space figure Math Background Integral calculus considers the area under a curve, which leads to computation of volumes of 1 Finding Volume of a Prism solids of revolution. Cavalieri’s Principle is a forerunner of ideas formalized by Newton and Leibniz in calculus. Hands-On Activity: Finding Volume Explore the volume of a prism with unit cubes. -
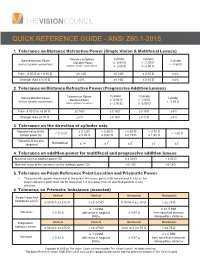
Quick Reference Guide - Ansi Z80.1-2015
QUICK REFERENCE GUIDE - ANSI Z80.1-2015 1. Tolerance on Distance Refractive Power (Single Vision & Multifocal Lenses) Cylinder Cylinder Sphere Meridian Power Tolerance on Sphere Cylinder Meridian Power ≥ 0.00 D > - 2.00 D (minus cylinder convention) > -4.50 D (minus cylinder convention) ≤ -2.00 D ≤ -4.50 D From - 6.50 D to + 6.50 D ± 0.13 D ± 0.13 D ± 0.15 D ± 4% Stronger than ± 6.50 D ± 2% ± 0.13 D ± 0.15 D ± 4% 2. Tolerance on Distance Refractive Power (Progressive Addition Lenses) Cylinder Cylinder Sphere Meridian Power Tolerance on Sphere Cylinder Meridian Power ≥ 0.00 D > - 2.00 D (minus cylinder convention) > -3.50 D (minus cylinder convention) ≤ -2.00 D ≤ -3.50 D From -8.00 D to +8.00 D ± 0.16 D ± 0.16 D ± 0.18 D ± 5% Stronger than ±8.00 D ± 2% ± 0.16 D ± 0.18 D ± 5% 3. Tolerance on the direction of cylinder axis Nominal value of the ≥ 0.12 D > 0.25 D > 0.50 D > 0.75 D < 0.12 D > 1.50 D cylinder power (D) ≤ 0.25 D ≤ 0.50 D ≤ 0.75 D ≤ 1.50 D Tolerance of the axis Not Defined ° ° ° ° ° (degrees) ± 14 ± 7 ± 5 ± 3 ± 2 4. Tolerance on addition power for multifocal and progressive addition lenses Nominal value of addition power (D) ≤ 4.00 D > 4.00 D Nominal value of the tolerance on the addition power (D) ± 0.12 D ± 0.18 D 5. Tolerance on Prism Reference Point Location and Prismatic Power • The prismatic power measured at the prism reference point shall not exceed 0.33Δ or the prism reference point shall not be more than 1.0 mm away from its specified position in any direction. -

A Three-Dimensional Laguerre Geometry and Its Visualization
A Three-Dimensional Laguerre Geometry and Its Visualization Hans Havlicek and Klaus List, Institut fur¨ Geometrie, TU Wien 3 We describe and visualize the chains of the 3-dimensional chain geometry over the ring R("), " = 0. MSC 2000: 51C05, 53A20. Keywords: chain geometry, Laguerre geometry, affine space, twisted cubic. 1 Introduction The aim of the present paper is to discuss in some detail the Laguerre geometry (cf. [1], [6]) which arises from the 3-dimensional real algebra L := R("), where "3 = 0. This algebra generalizes the algebra of real dual numbers D = R("), where "2 = 0. The Laguerre geometry over D is the geometry on the so-called Blaschke cylinder (Figure 1); the non-degenerate conics on this cylinder are called chains (or cycles, circles). If one generator of the cylinder is removed then the remaining points of the cylinder are in one-one correspon- dence (via a stereographic projection) with the points of the plane of dual numbers, which is an isotropic plane; the chains go over R" to circles and non-isotropic lines. So the point space of the chain geometry over the real dual numbers can be considered as an affine plane with an extra “improper line”. The Laguerre geometry based on L has as point set the projective line P(L) over L. It can be seen as the real affine 3-space on L together with an “improper affine plane”. There is a point model R for this geometry, like the Blaschke cylinder, but it is more compli- cated, and belongs to a 7-dimensional projective space ([6, p. -

Math 366 Lecture Notes Section 11.4 – Geometry in Three Dimensions
Section 11-4 Math 366 Lecture Notes Section 11.4 – Geometry in Three Dimensions Simple Closed Surfaces A simple closed surface has exactly one interior, no holes, and is hollow. A sphere is the set of all points at a given distance from a given point, the center . A sphere is a simple closed surface. A solid is a simple closed surface with all interior points. (see p. 726) A polyhedron is a simple closed surface made up of polygonal regions, or faces . The vertices of the polygonal regions are the vertices of the polyhedron, and the sides of each polygonal region are the edges of the polyhedron. (see p. 726-727) A prism is a polyhedron in which two congruent faces lie in parallel planes and the other faces are bounded by parallelograms. The parallel faces of a prism are the bases of the prism. A prism is usually names after its bases. The faces other than the bases are the lateral faces of a prism. A right prism is one in which the lateral faces are all bounded by rectangles. An oblique prism is one in which some of the lateral faces are not bounded by rectangles. To draw a prism: 1) Draw one of the bases. 2) Draw vertical segments of equal length from each vertex. 3) Connect the bottom endpoints to form the second base. Use dashed segments for edges that cannot be seen. A pyramid is a polyhedron determined by a polygon and a point not in the plane of the polygon. The pyramid consists of the triangular regions determined by the point and each pair of consecutive vertices of the polygon and the polygonal region determined by the polygon. -

Natural Cadmium Is Made up of a Number of Isotopes with Different Abundances: Cd106 (1.25%), Cd110 (12.49%), Cd111 (12.8%), Cd
CLASSROOM Natural cadmium is made up of a number of isotopes with different abundances: Cd106 (1.25%), Cd110 (12.49%), Cd111 (12.8%), Cd 112 (24.13%), Cd113 (12.22%), Cd114(28.73%), Cd116 (7.49%). Of these Cd113 is the main neutron absorber; it has an absorption cross section of 2065 barns for thermal neutrons (a barn is equal to 10–24 sq.cm), and the cross section is a measure of the extent of reaction. When Cd113 absorbs a neutron, it forms Cd114 with a prompt release of γ radiation. There is not much energy release in this reaction. Cd114 can again absorb a neutron to form Cd115, but the cross section for this reaction is very small. Cd115 is a β-emitter C V Sundaram, National (with a half-life of 53hrs) and gets transformed to Indium-115 Institute of Advanced Studies, Indian Institute of Science which is a stable isotope. In none of these cases is there any large Campus, Bangalore 560 012, release of energy, nor is there any release of fresh neutrons to India. propagate any chain reaction. Vishwambhar Pati The Möbius Strip Indian Statistical Institute Bangalore 560059, India The Möbius strip is easy enough to construct. Just take a strip of paper and glue its ends after giving it a twist, as shown in Figure 1a. As you might have gathered from popular accounts, this surface, which we shall call M, has no inside or outside. If you started painting one “side” red and the other “side” blue, you would come to a point where blue and red bump into each other. -

Year 6 – Wednesday 24Th June 2020 – Maths
1 Year 6 – Wednesday 24th June 2020 – Maths Can I identify 3D shapes that have pairs of parallel or perpendicular edges? Parallel – edges that have the same distance continuously between them – parallel edges never meet. Perpendicular – when two edges or faces meet and create a 90o angle. 1 4. 7. 10. 2 5. 8. 11. 3. 6. 9. 12. C1 – Using the shapes above: 1. Name and sort the shapes into: a. Pyramids b. Prisms 2. Draw a table to identify how many faces, edges and vertices each shape has. 3. Write your own geometric definition: a. Prism b. pyramid 4. Which shape is the odd one out? Explain why. C2 – Using the shapes above; 1. Which of the shapes have pairs of parallel edges in: a. All their faces? b. More than one half of the faces? c. One face only? 2. The following shapes have pairs of perpendicular edges. Identify the faces they are in: 2 a. A cube b. A square based pyramid c. A triangular prism d. A cuboid 3. Which shape with straight edges has no perpendicular edges? 4. Which shape has perpendicular edges in the shape but not in any face? C3 – Using the above shapes: 1. How many faces have pairs of parallel edges in: a. A hexagonal pyramid? b. A decagonal (10-sided) based prism? c. A heptagonal based prism? d. Which shape has no face with parallel edges but has parallel edges in the shape? 2. How many faces have perpendicular edges in: a. A pentagonal pyramid b. A hexagonal pyramid c. -

Unit 6 Visualising Solid Shapes(Final)
• 3D shapes/objects are those which do not lie completely in a plane. • 3D objects have different views from different positions. • A solid is a polyhedron if it is made up of only polygonal faces, the faces meet at edges which are line segments and the edges meet at a point called vertex. • Euler’s formula for any polyhedron is, F + V – E = 2 Where F stands for number of faces, V for number of vertices and E for number of edges. • Types of polyhedrons: (a) Convex polyhedron A convex polyhedron is one in which all faces make it convex. e.g. (1) (2) (3) (4) 12/04/18 (1) and (2) are convex polyhedrons whereas (3) and (4) are non convex polyhedron. (b) Regular polyhedra or platonic solids: A polyhedron is regular if its faces are congruent regular polygons and the same number of faces meet at each vertex. For example, a cube is a platonic solid because all six of its faces are congruent squares. There are five such solids– tetrahedron, cube, octahedron, dodecahedron and icosahedron. e.g. • A prism is a polyhedron whose bottom and top faces (known as bases) are congruent polygons and faces known as lateral faces are parallelograms (when the side faces are rectangles, the shape is known as right prism). • A pyramid is a polyhedron whose base is a polygon and lateral faces are triangles. • A map depicts the location of a particular object/place in relation to other objects/places. The front, top and side of a figure are shown. Use centimetre cubes to build the figure. -

Triangulation of Simple 3D Shapes with Well-Centered Tetrahedra
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Illinois Digital Environment for Access to Learning and Scholarship Repository TRIANGULATION OF SIMPLE 3D SHAPES WITH WELL-CENTERED TETRAHEDRA EVAN VANDERZEE, ANIL N. HIRANI, AND DAMRONG GUOY Abstract. A completely well-centered tetrahedral mesh is a triangulation of a three dimensional domain in which every tetrahedron and every triangle contains its circumcenter in its interior. Such meshes have applications in scientific computing and other fields. We show how to triangulate simple domains using completely well-centered tetrahedra. The domains we consider here are space, infinite slab, infinite rectangular prism, cube, and regular tetrahedron. We also demonstrate single tetrahedra with various combinations of the properties of dihedral acuteness, 2-well-centeredness, and 3-well-centeredness. 1. Introduction 3 In this paper we demonstrate well-centered triangulation of simple domains in R . A well- centered simplex is one for which the circumcenter lies in the interior of the simplex [8]. This definition is further refined to that of a k-well-centered simplex which is one whose k-dimensional faces have the well-centeredness property. An n-dimensional simplex which is k-well-centered for all 1 ≤ k ≤ n is called completely well-centered [15]. These properties extend to simplicial complexes, i.e. to meshes. Thus a mesh can be completely well-centered or k-well-centered if all its simplices have that property. For triangles, being well-centered is the same as being acute-angled. But a tetrahedron can be dihedral acute without being 3-well-centered as we show by example in Sect. -

Plus Cylinder Lensometry Tara L Bragg, CO August 2015
Mark E Wilkinson, OD Plus Cylinder Lensometry Tara L Bragg, CO August 2015 Lensometry A lensometer is an optical bench consisting of an illuminated moveable target, a powerful fixed lens, and a telescopic eyepiece focused at infinity. The key element is the field lens that is fixed in place so that its focal point is on the back surface of the lens being analyzed. The lensometer measures the back vertex power of the spectacle lens. The vertex power is the reciprocal of the distance between the back surface of the lens and its secondary focal point. This is also known as the back focal length. For this reason, a lensometer does not really measure the true focal length of a lens, which is measured from the principal planes, not from the lens surface. The lensometer works on the Badal principle with the addition of an astronomical telescope for precise detection of parallel rays at neutralization. The Badel principle is Knapp’s law applied to lensometers. Performing Manual Lensometry Focusing the Eyepiece The focus of the lensometer eyepiece must be verified each time the instrument is used, to avoid erroneous readings. 1. With no lens or a plano lens in place on the lensometer, look through the eyepiece of the instrument. Turn the power drum until the mires (the perpendicular cross lines), viewed through the eyepiece, are grossly out of focus. 2. Turn the eyepiece in the plus direction to fog (blur) the target seen through the eyepiece. 3. Slowly turn the eyepiece in the opposite direction until the target is clear, then stop turning. -

Bicapped Tetrahedral, Trigonal Prismatic, and Octahedral Alternatives in Main and Transition Group Six-Coordination
2484 Bicapped Tetrahedral, Trigonal Prismatic, and Octahedral Alternatives in Main and Transition Group Six-Coordination Roald Hoffmann,*laJames M. Howell,lb and Angelo R. RossilC Contribution from the Departments of Chemistry, Cornell University, Ithaca, New York 14853, Brooklyn College, City University of New York. Brooklyn, New York, 1121 0, and The University of Connecticut, Storrs, Connecticut 06268. Received June 30, 1975 Abstract: We examine theoretically the bicapped tetrahedron and trigonal prism as alternatives to the common octahedron for six-coordinate main and transition group compounds. The pronounced preference of six-coordinate sulfur for an octahe- dral geometry is traced by a molecular orbital analysis to a pair of nonbonding levels, whose higher energy in the nonoctahe- dral geometries is due to the molecular orbital equivalent of a ligand-ligand repulsion. For transition element complexes we draw a correlation diagram for the trigonal twist interrelating octahedral and trigonal prismatic extremes. A possible prefer- ence for the trigonal prism in systems with few d electrons is discussed, as well as a strategy for lowering the energy of the trigonal prism by symmetry-conditioned 7r bonding. The bicapped tetrahedron geometry for transition metal complexes can be stabilized by a substitution pattern ML4D2 where D is a good u donor and M is a d6 metal center. In main and transition group six-coordinate compounds logical, or group theoretical analysis of rearrangement the octahedral geometry predominates. There is, however, a modes or basic permutational sets. However, the solution of growing class of trigonal prismatic complexes and struc- the mathematical problem leaves the question of the physi- tures intermediate between the octahedron and trigonal cal mechanism of a rearrangement still unsolved.