Chapter 4 Nomenclature
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

Safety Data Sheet Chloride
UNIVAR USA INC. SDS NO:DW79537 ISSUE DATE:2015-06-08 VERSION:014 2015-08-04 Annotation: SAFETY DATA SHEET M48005 - ANSI - EN _____________________________________________________________________________________________________ CHLORIDE SDS No.: M48005 SDS Revision Date: 08-Jun-2015 _____________________________________________________________________________________________________ _____________________________________________________________________________________________________ 1. CHEMICAL PRODUCT AND COMPANY IDENTIFICATION _____________________________________________________________________________________________________ Company Identification: Occidental Chemical Corporation 5005 LBJ Freeway P.O. Box 809050 Dallas, TX 75380-9050 1-800-752-5151 24 Hour Emergency Telephone 1-800-733-3665 or 1-972-404-3228 (USA); CHEMTREC (within USA and Number: Canada): 1-800-424-9300; CHEMTREC (outside USA and Canada): +1 703-527-3887; CHEMTREC Contract No: CCN16186 To Request an SDS: [email protected] or 1-972-404-3245 Customer Service: 1-800-752-5151 or 1-972-404-3700 Product Identifier: Synonyms: Calcium Dichloride, Calcium Chloride, Peladow, Calcium Chloride Pellets Product Use: Ice Melting Uses Advised Against: None identified. Additional Information: CONSUMER PRODUCTS: When packaged in quantities of 50 lbs. or less, and used in a manner and frequency typical of consumer use, OxyChem considers this product a consumer use product which is regulated by the Consumer Product Safety Commission (CPSC). Because CPSC labeling requirements differ from the -

WTR-Core Product Code SMI2337-1 SDS No
Conforms to Regulation (EC) No. 1907/2006 (REACH), Annex II, as amended by Commission Regulation (EU) 2015/830 SAFETY DATA SHEET SECTION 1: Identification of the substance/mixture and of the company/undertaking 1.1 Product identifier Product name WTR-Core Product code SMI2337-1 SDS no. SMI2337-1 Product type Paste 1.2 Relevant identified uses of the substance or mixture and uses advised against Use of the substance/ Analytical reagent that is mixed with activator to form a test kit. mixture For specific application advice see appropriate Technical Data Sheet or consult our company representative. 1.3 Details of the supplier of the safety data sheet Supplier BP Marine Limited Chertsey Road Sunbury-on-Thames Middlesex TW16 7LN United Kingdom E-mail address [email protected] 1.4 Emergency telephone number EMERGENCY Carechem: +44 (0) 1235 239 670 (24/7) TELEPHONE NUMBER SECTION 2: Hazards identification 2.1 Classification of the substance or mixture Product definition Mixture Classification according to Regulation (EC) No. 1272/2008 [CLP/GHS] Not classified. Ingredients of unknown Percentage of the mixture consisting of ingredient(s) of unknown acute oral toxicity: 1% toxicity Percentage of the mixture consisting of ingredient(s) of unknown acute dermal toxicity: 1% Percentage of the mixture consisting of ingredient(s) of unknown acute inhalation toxicity: 1% Ingredients of unknown Percentage of the mixture consisting of ingredient(s) of unknown hazards to the aquatic ecotoxicity environment: 1% See sections 11 and 12 for more detailed information on health effects and symptoms and environmental hazards. 2.2 Label elements Signal word No signal word. -

Calcium Hydride, Grade S
TECHNICAL DATA SHEET Date of Issue: 2016/09/02 Calcium Hydride, Grade S CAS-No. 7789-78-8 EC-No. 232-189-2 Molecular Formula CaH₂ Product Number 455150 APPLICATION Calcium hydride is used primarily as a source of hydrogen, as a drying agent for liquids and gases, and as a reducing agent for metal oxides. SPECIFICATION Ca total min. 92 % H min. 980 ml/g CaH2 Mg max. 0.8 % N max. 0.2 % Al max. 0.01 % Cl max. 0.5 % Fe max. 0.01 % METHOD OF ANALYSIS Calcium complexometric, impurities by spectral analysis and special analytical procedures. Gas volumetric determination of hydrogen. Produces with water approx. 1,010 ml hydrogen per gram. PHYSICAL PROPERTIES Appearance powder Color gray white The information presented herein is believed to be accurate and reliable, but is presented without guarantee or responsibility on the part of Albemarle Corporation and its subsidiaries and affiliates. It is the responsibility of the user to comply with all applicable laws and regulations and to provide for a safe workplace. The user should consider any health or safety hazards or information contained herein only as a guide, and should take those precautions which are necessary or prudent to instruct employees and to develop work practice procedures in order to promote a safe work environment. Further, nothing contained herein shall be taken as an inducement or recommendation to manufacture or use any of the herein materials or processes in violation of existing or future patent. Technical data sheets may change frequently. You can download the latest version from our website www.albemarle-lithium.com. -

Chemical Chemical Hazard and Compatibility Information
Chemical Chemical Hazard and Compatibility Information Acetic Acid HAZARDS & STORAGE: Corrosive and combustible liquid. Serious health hazard. Reacts with oxidizing and alkali materials. Keep above freezing point (62 degrees F) to avoid rupture of carboys and glass containers.. INCOMPATIBILITIES: 2-amino-ethanol, Acetaldehyde, Acetic anhydride, Acids, Alcohol, Amines, 2-Amino-ethanol, Ammonia, Ammonium nitrate, 5-Azidotetrazole, Bases, Bromine pentafluoride, Caustics (strong), Chlorosulfonic acid, Chromic Acid, Chromium trioxide, Chlorine trifluoride, Ethylene imine, Ethylene glycol, Ethylene diamine, Hydrogen cyanide, Hydrogen peroxide, Hydrogen sulfide, Hydroxyl compounds, Ketones, Nitric Acid, Oleum, Oxidizers (strong), P(OCN)3, Perchloric acid, Permanganates, Peroxides, Phenols, Phosphorus isocyanate, Phosphorus trichloride, Potassium hydroxide, Potassium permanganate, Potassium-tert-butoxide, Sodium hydroxide, Sodium peroxide, Sulfuric acid, n-Xylene. Acetone HAZARDS & STORAGE: Store in a cool, dry, well ventilated place. INCOMPATIBILITIES: Acids, Bromine trifluoride, Bromine, Bromoform, Carbon, Chloroform, Chromium oxide, Chromium trioxide, Chromyl chloride, Dioxygen difluoride, Fluorine oxide, Hydrogen peroxide, 2-Methyl-1,2-butadiene, NaOBr, Nitric acid, Nitrosyl chloride, Nitrosyl perchlorate, Nitryl perchlorate, NOCl, Oxidizing materials, Permonosulfuric acid, Peroxomonosulfuric acid, Potassium-tert-butoxide, Sulfur dichloride, Sulfuric acid, thio-Diglycol, Thiotrithiazyl perchlorate, Trichloromelamine, 2,4,6-Trichloro-1,3,5-triazine -
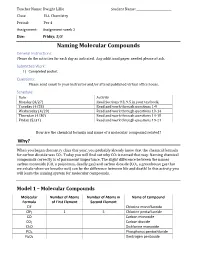
Naming Molecular Compounds General Instructions: Please Do the Activities for Each Day As Indicated
Teacher Name: Dwight Lillie Student Name: ________________________ Class: ELL Chemistry Period: Per 4 Assignment: Assignment week 2 Due: Friday, 5/8 Naming Molecular Compounds General Instructions: Please do the activities for each day as indicated. Any additional paper needed please attach. Submitted Work: 1) Completed packet. Questions: Please send email to your instructor and/or attend published virtual office hours. Schedule: Date Activity Monday (4/27) Read Sections 9.3, 9.5 in your textbook. Tuesday (4/28) Read and work through questions 1-9 Wednesday (4/29) Read and work through questions 10-14 Thursday (4/30) Read and work through questions 14-18 Friday (5/31) Read and work through questions 19-21 How are the chemical formula and name of a molecular compound related? Why? When you began chemistry class this year, you probably already knew that the chemical formula for carbon dioxide was CO2. Today you will find out why CO2 is named that way. Naming chemical compounds correctly is of paramount importance. The slight difference between the names carbon monoxide (CO, a poisonous, deadly gas) and carbon dioxide (CO2, a greenhouse gas that we exhale when we breathe out) can be the difference between life and death! In this activity you will learn the naming system for molecular compounds. Model 1 – Molecular Compounds Molecular Number of Atoms Number of Atoms in Name of Compound Formula of First Element Second Element ClF Chlorine monofluoride ClF5 1 5 Chlorine pentafluoride CO Carbon monoxide CO2 Carbon dioxide Cl2O Dichlorine monoxide PCl5 Phosphorus pentachloride N2O5 Dinitrogen pentoxide 1. Fill in the table to indicate the number of atoms of each type in the molecular formula. -

Hazardous Material Inventory Statement
City of Brooklyn Park FIRE DEPARTMENT 5200 - 85th Avenue North Brooklyn Park MN 55443 Phone: (763)493-8020 Fax: (763) 493-8391 Hazardous Materials Inventory Statement Users Guide A separate inventory statement shall be provided for each building. An amended inventory statement shall be provided within 30 days of the storage of any hazardous materials or plastics that changes or adds a hazard class or which is sufficient in quantity to cause an increase in the quantity which exceeds 5 percent for any hazard class. The hazardous materials inventory statement shall list by hazard class categories. Each grouping shall provide the following information for each hazardous material listed for that group including a total quantity for each group of hazard class. 1. Hazard class. (See attached Hazardous Materials Categories Listing) 2. Common or trade name. 3. Chemical Abstract Service Number (CAS number) found in 29 Code of Federal Regulations (C.F.R.). 4. Whether the material is pure or a mixture, and whether the material is a solid, liquid or gas 5. Maximum aggregate quantity stored at any one time. 6. Maximum aggregate quantity In-Use (Open to atmosphere) at any one time. 7. Maximum aggregate quantity In-Use (Closed to atmosphere) at any one time. 8. Storage conditions related to the storage type, high-pile, encapsulated, non-encapsulated. Attached is a listing of categories that all materials need to be organized to. Definitions of these categories are also attached for your use. At the end of this packet are blank forms for completing this project. For questions regarding Hazardous Materials Inventory Statement contact the Fire Department at 763-493-8020. -

Towards Better Understanding of C60 Organosols†
PCCP View Article Online PAPER View Journal | View Issue Towards better understanding of C60 organosols† a a a Cite this: Phys. Chem. Chem. Phys., Nikolay O. Mchedlov-Petrossyan,* Nika N. Kamneva, Younis T. M. Al-Shuuchi, b c d e 2016, 18,2517 Andriy I. Marynin, Olexii S. Zozulia, Alexander P. Kryshtal, Vladimir K. Klochkov and Sergey V. Shekhovtsova It is of common knowledge that fullerenes form colloids in polar solvents. However, the coagulation via electrolytes and the origin of the negative charge of species are still unexplored. Using a ‘radical À scavenger’ and electrospray ionization spectroscopy (ESI), we proved the formation of ion-radical C60 2À 2À and its (probable) transformation into C60 or (C60)2 . The coagulation of C60 organosols by NaClO4 and other perchlorates and nitrates in acetonitrile and its mixture with benzene obeys the Schulze– Hardy rule. At higher Ca(ClO4)2 and La(ClO4)3 concentrations, instead of coagulation, stable re-charged Received 7th November 2015, colloidal particles appeared, up to a zeta-potential of +(20–42) mV, as compared with À(33–35) mV of Accepted 7th December 2015 the initial organosols. The influence of both HClO4 and CF3SO3H was similar. This phenomenon is attrib- DOI: 10.1039/c5cp06806a uted to poor solvation of inorganic cations in cationo- and protophobic acetonitrile, which was proven using [2.2.2] cryptand. Further increasing the concentration of Ca(ClO4)2 led again to coagulation, thus www.rsc.org/pccp demonstrating a novel type of ‘coagulation zones’. 1. Introduction believe that -

Calcium Hydride Hazard Summary Identification Reason for Citation How to Determine If You Are Being Exposed Workpla
Common Name: CALCIUM HYDRIDE CAS Number: 7789-78-8 RTK Substance number: 0320 DOT Number: UN 1404 Date: March 1987 Revision: March 2000 ----------------------------------------------------------------------- ----------------------------------------------------------------------- HAZARD SUMMARY WORKPLACE EXPOSURE LIMITS * Calcium Hydride can affect you when breathed in. No occupational exposure limits have been established for * Contact can severely irritate and burn the skin and eyes Calcium Hydride. This does not mean that this substance is with possible eye damage. not harmful. Safe work practices should always be followed. * Breathing Calcium Hydride can irritate the nose and throat. WAYS OF REDUCING EXPOSURE * Breathing Calcium Hydride can irritate the lungs causing * Where possible, enclose operations and use local exhaust coughing and/or shortness of breath. Higher exposures ventilation at the site of chemical release. If local exhaust can cause a build-up of fluid in the lungs (pulmonary ventilation or enclosure is not used, respirators should be edema), a medical emergency, with severe shortness of worn. breath. * Wear protective work clothing. * Calcium Hydride is a FLAMMABLE and REACTIVE * Wash thoroughly immediately after exposure to Calcium chemical and a FIRE and EXPLOSION HAZARD. Hydride. * Post hazard and warning information in the work area. In IDENTIFICATION addition, as part of an ongoing education and training Calcium Hydride is a grayish-white crystalline (sand-like) effort, communicate all information on the health and solid. It is used as a drying and reducing agent and as a safety hazards of Calcium Hydride to potentially exposed cleaner for blocked up oil wells. workers. REASON FOR CITATION * Calcium Hydride is on the Hazardous Substance List because it is cited by DOT. -

Appendix a of Final Environmental Impact Statement for a Geologic Repository for the Disposal of Spent Nuclear Fuel and High-Lev
Appendix A Inventory and Characteristics of Spent Nuclear Fuel, High-Level Radioactive Waste, and Other Materials Inventory and Characteristics of Spent Nuclear Fuel, High-Level Radioactive Waste, and Other Materials TABLE OF CONTENTS Section Page A. Inventory and Characteristics of Spent Nuclear Fuel, High-Level Radioactive Waste, and Other Materials ................................................................................................................................. A-1 A.1 Introduction .............................................................................................................................. A-1 A.1.1 Inventory Data Summary .................................................................................................... A-2 A.1.1.1 Sources ......................................................................................................................... A-2 A.1.1.2 Present Storage and Generation Status ........................................................................ A-4 A.1.1.3 Final Waste Form ......................................................................................................... A-6 A.1.1.4 Waste Characteristics ................................................................................................... A-6 A.1.1.4.1 Mass and Volume ................................................................................................. A-6 A.1.1.4.2 Radionuclide Inventories ...................................................................................... A-8 A.1.1.4.3 -

Layered Iron Vanadate As a High-Capacity Cathode Material for Nonaqueous Calcium-Ion Batteries
batteries Article Layered Iron Vanadate as a High-Capacity Cathode Material for Nonaqueous Calcium-Ion Batteries Munseok S. Chae 1 , Dedy Setiawan 1, Hyojeong J. Kim 1 and Seung-Tae Hong 1,2,* 1 Department of Energy Science and Engineering, DGIST (Daegu Gyeongbuk Institute of Science and Technology), Daegu 42988, Korea; [email protected] (M.S.C.); [email protected] (D.S.); [email protected] (H.J.K.) 2 Energy Science and Engineering Research Center, DGIST (Daegu Gyeongbuk Institute of Science and Technology), Daegu 42988, Korea * Correspondence: [email protected]; Tel.: +82-53-785-6415 Abstract: Calcium-ion batteries represent a promising alternative to the current lithium-ion batteries. Nevertheless, calcium-ion intercalating materials in nonaqueous electrolytes are scarce, probably due to the difficulties in finding suitable host materials. Considering that research into calcium-ion batteries is in its infancy, discovering and characterizing new host materials would be critical to further development. Here, we demonstrate FeV3O9·1.2H2O as a high-performance calcium-ion battery cathode material that delivers a reversible discharge capacity of 303 mAh g−1 with a good cycling stability and an average discharge voltage of ~2.6 V (vs. Ca/Ca2+). The material was synthesized via a facile co-precipitation method. Its reversible capacity is the highest among calcium- ion battery materials, and it is the first example of a material with a capacity much larger than that of conventional lithium-ion battery cathode materials. Bulk intercalation of calcium into the host lattice contributed predominantly to the total capacity at a lower rate, but became comparable to that due to Citation: Chae, M.S.; Setiawan, D.; surface adsorption at a higher rate. -

Chemical Names and CAS Numbers Final
Chemical Abstract Chemical Formula Chemical Name Service (CAS) Number C3H8O 1‐propanol C4H7BrO2 2‐bromobutyric acid 80‐58‐0 GeH3COOH 2‐germaacetic acid C4H10 2‐methylpropane 75‐28‐5 C3H8O 2‐propanol 67‐63‐0 C6H10O3 4‐acetylbutyric acid 448671 C4H7BrO2 4‐bromobutyric acid 2623‐87‐2 CH3CHO acetaldehyde CH3CONH2 acetamide C8H9NO2 acetaminophen 103‐90‐2 − C2H3O2 acetate ion − CH3COO acetate ion C2H4O2 acetic acid 64‐19‐7 CH3COOH acetic acid (CH3)2CO acetone CH3COCl acetyl chloride C2H2 acetylene 74‐86‐2 HCCH acetylene C9H8O4 acetylsalicylic acid 50‐78‐2 H2C(CH)CN acrylonitrile C3H7NO2 Ala C3H7NO2 alanine 56‐41‐7 NaAlSi3O3 albite AlSb aluminium antimonide 25152‐52‐7 AlAs aluminium arsenide 22831‐42‐1 AlBO2 aluminium borate 61279‐70‐7 AlBO aluminium boron oxide 12041‐48‐4 AlBr3 aluminium bromide 7727‐15‐3 AlBr3•6H2O aluminium bromide hexahydrate 2149397 AlCl4Cs aluminium caesium tetrachloride 17992‐03‐9 AlCl3 aluminium chloride (anhydrous) 7446‐70‐0 AlCl3•6H2O aluminium chloride hexahydrate 7784‐13‐6 AlClO aluminium chloride oxide 13596‐11‐7 AlB2 aluminium diboride 12041‐50‐8 AlF2 aluminium difluoride 13569‐23‐8 AlF2O aluminium difluoride oxide 38344‐66‐0 AlB12 aluminium dodecaboride 12041‐54‐2 Al2F6 aluminium fluoride 17949‐86‐9 AlF3 aluminium fluoride 7784‐18‐1 Al(CHO2)3 aluminium formate 7360‐53‐4 1 of 75 Chemical Abstract Chemical Formula Chemical Name Service (CAS) Number Al(OH)3 aluminium hydroxide 21645‐51‐2 Al2I6 aluminium iodide 18898‐35‐6 AlI3 aluminium iodide 7784‐23‐8 AlBr aluminium monobromide 22359‐97‐3 AlCl aluminium monochloride