Lactation and Reproduction *
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Index Vol. 12-15
353 INDEX VOL. 12-15 Die Stichworte des Sachregisters sind in der jeweiligen Sprache der einzelnen Beitrage aufgefiihrt. Les termes repris dans la Table des matieres sont donnes selon la langue dans laquelle l'ouvrage est ecrit. The references of the Subject Index are given in the language of the respective contribution. 14 AAG (Alpha-acid glycoprotein) 120 14 Adenosine 108 12 Abortion 151 12 Adenosine-phosphate 311 13 Abscisin 12, 46, 66 13 Adenosine-5'-phosphosulfate 148 14 Absorbierbarkeit 317 13 Adenosine triphosphate 358 14 Absorption 309, 350 15 S-Adenosylmethionine 261 13 Absorption of drugs 139 13 Adipaenin (Spasmolytin) 318 14 - 15 12 Adrenal atrophy 96 14 Absorptionsgeschwindigkeit 300, 306 14 - 163, 164 14 Absorptionsquote 324 13 Adrenal gland 362 14 ACAI (Anticorticocatabolic activity in 12 Adrenalin(e) 319 dex) 145 14 - 209, 210 12 Acalo 197 15 - 161 13 Aceclidine (3-Acetoxyquinuclidine) 307, 13 {i-Adrenergic blockers 119 308, 310, 311, 330, 332 13 Adrenergic-blocking activity 56 13 Acedapsone 193,195,197 14 O(-Adrenergic blocking drugs 36, 37, 43 13 Aceperone (Acetabutone) 121 14 {i-Adrenergic blocking drugs 38 12 Acepromazin (Plegizil) 200 14 Adrenergic drugs 90 15 Acetanilid 156 12 Adrenocorticosteroids 14, 30 15 Acetazolamide 219 12 Adrenocorticotropic hormone (ACTH) 13 Acetoacetyl-coenzyme A 258 16,30,155 12 Acetohexamide 16 14 - 149,153,163,165,167,171 15 1-Acetoxy-8-aminooctahydroindolizin 15 Adrenocorticotropin (ACTH) 216 (Slaframin) 168 14 Adrenosterone 153 13 4-Acetoxy-1-azabicyclo(3, 2, 2)-nonane 12 Adreson 252 -

Pp375-430-Annex 1.Qxd
ANNEX 1 CHEMICAL AND PHYSICAL DATA ON COMPOUNDS USED IN COMBINED ESTROGEN–PROGESTOGEN CONTRACEPTIVES AND HORMONAL MENOPAUSAL THERAPY Annex 1 describes the chemical and physical data, technical products, trends in produc- tion by region and uses of estrogens and progestogens in combined estrogen–progestogen contraceptives and hormonal menopausal therapy. Estrogens and progestogens are listed separately in alphabetical order. Trade names for these compounds alone and in combination are given in Annexes 2–4. Sales are listed according to the regions designated by WHO. These are: Africa: Algeria, Angola, Benin, Botswana, Burkina Faso, Burundi, Cameroon, Cape Verde, Central African Republic, Chad, Comoros, Congo, Côte d'Ivoire, Democratic Republic of the Congo, Equatorial Guinea, Eritrea, Ethiopia, Gabon, Gambia, Ghana, Guinea, Guinea-Bissau, Kenya, Lesotho, Liberia, Madagascar, Malawi, Mali, Mauritania, Mauritius, Mozambique, Namibia, Niger, Nigeria, Rwanda, Sao Tome and Principe, Senegal, Seychelles, Sierra Leone, South Africa, Swaziland, Togo, Uganda, United Republic of Tanzania, Zambia and Zimbabwe America (North): Canada, Central America (Antigua and Barbuda, Bahamas, Barbados, Belize, Costa Rica, Cuba, Dominica, El Salvador, Grenada, Guatemala, Haiti, Honduras, Jamaica, Mexico, Nicaragua, Panama, Puerto Rico, Saint Kitts and Nevis, Saint Lucia, Saint Vincent and the Grenadines, Suriname, Trinidad and Tobago), United States of America America (South): Argentina, Bolivia, Brazil, Chile, Colombia, Dominican Republic, Ecuador, Guyana, Paraguay, -

61/2003, Uredbeni
Stran 7380 / Št. 61 / 26. 06. 2003 Uradni list Republike Slovenije ODDELEK VI PROIZVODI KEMIČNE INDUSTRIJE ALI PODOBNIH INDUSTRIJ OPOMBE: 1. (a) Proizvodi (razen radioaktivnih rud), ki ustrezajo poimenovanjem v tar. št. 2844 ali 2845, se uvrščajo v omenjeni tarifni številki, ne pa v druge tariFne številke Nomenklature. (b) V skladu s prejšnjim odstavkom se proizvodi, ki ustrezajo poimenovanjem v tar.št. 2843 ali 2846., uvrščajo samo v omenjeni tarifni številki, ne pa v druge tariFne številke tega oddelka. 2. V skladu z opombo št.1 se proizvodi, ki bi se zaradi pakiranja na drobno ali v odmerjene količine uvrstili v tar. št. 3004, 3005, 3006, 3212, 3303, 3304, 3305, 3306, 3307, 3506, 3707 ali 3808, se uvrščajo samo v omenjeni tar. št., (2844 ali 2845) ne pa v druge tariFne številke Nomenklature. 3. Proizvodi, pripravljeni v garniturah, ki sestoje iz dveh ali več ločenih sestavin, od katerih se neka- tere ali vse uvrščajo v ta oddelek in so namenjene za to, da se pomešajo skupaj, da bi se dobil proizvod VI. ali VII. oddelka, se uvrščajo v ustrezno tarifno številko za ta proizvod pod pogojem, da so sestavine: (a) take, da je iz tega, kako so pripravljene, razvidno, da so namenjene za uporabo skupaj brez poprejšnjega prepakiranja, (b) da se skupaj carinijo in (c) da so take, da je razvidno iz njihove narave ali po sorazmernih količinah, v katerih so zasto- pane, da se med seboj dopolnjujejo. SPLOŠNA DOLOČILA Opomba št. 1 Skladno z določili točke (a) opombe št. 1 se vsi radioaktivni kemični elementi, radioaktivni izotopi in spojine teh elementov in izotopov (anorganskih ali organskih, kemično določenih ali nedoločenih) uvrščajo v tar. -

MMC International BV
M.M.C. International Steroid Substances Steroid Test A Colour Steroid Test B Colour Steroid Test B Colour with UV Light Stanozolol/ Oxandrolone Test Clenbuterol/ Oxymetholone Test Ephedrine Test Alfadolone Orange Yellow Nil - - - Androsterone Orange Yellow White - - - Beclometasone Brown–yellow Orange Nil - - - Betamethasone Orange–brown Pink–Orange Nil - - - Boldenone Base (Equipoise, Ganabol) (pure powder) Warm red after 2 min. Dark Orange after 2 min. Bright Light Orange - - - Boldenone Undecanoate (oil) Dark brownish-red Dark Red Bright Light Orange - - - Boldenone Undecylenate (oil) Orange - Light Brown Dark Orange → Brown Bright Light Orange-Yellow - - - Carbenoxolone (CBX) Orange Yellow Yellow - - - Cholesterol Violet Orange White - - - Clenbuterol (Spiropent, Ventipulmin) - - - - Purple - Dark brown with yellow-green on the Dark brown with yellow-green on the Clomiphene (Androxal, Clomid, Omifin) Nil Dark brown to black No reaction Dark brown to black sides of the ampoule sides of the ampoule Cortisone Orange Yellow Green - - - Desoxycortone Blue–black Yellow Yellow - - - Dexamethasone Yellow Orange–pink Nil - - - Dienestrol Yellow Orange–red Nil - - - Diethylstilbestrol (DES) Orange (→yellow–green) Nil - - - Dimethisterone Brown–green Orange–red Yellow - - - Drostanolone Propionate (Masteron) (oil) Bright green Yellow-Orange Orange - - - Dydrogesterone (Duphaston) - Orange Green-Yellow - - - Enoxolone Orange Yellow Green-Yellow - - - Ephedrine (also for Pseudo- and Nor-Ephedrine) - - - - - Orange Estradiol (Oestradiol) Orange -

Combined Estrogen–Progestogen Menopausal Therapy
COMBINED ESTROGEN–PROGESTOGEN MENOPAUSAL THERAPY Combined estrogen–progestogen menopausal therapy was considered by previous IARC Working Groups in 1998 and 2005 (IARC, 1999, 2007). Since that time, new data have become available, these have been incorporated into the Monograph, and taken into consideration in the present evaluation. 1. Exposure Data 1.1.2 Progestogens (a) Chlormadinone acetate Combined estrogen–progestogen meno- Chem. Abstr. Serv. Reg. No.: 302-22-7 pausal therapy involves the co-administration Chem. Abstr. Name: 17-(Acetyloxy)-6-chlo- of an estrogen and a progestogen to peri- or ropregna-4,6-diene-3,20-dione menopausal women. The use of estrogens with IUPAC Systematic Name: 6-Chloro-17-hy- progestogens has been recommended to prevent droxypregna-4,6-diene-3,20-dione, acetate the estrogen-associated risk of endometrial Synonyms: 17α-Acetoxy-6-chloro-4,6- cancer. Evidence from the Women’s Health pregnadiene-3,20-dione; 6-chloro-Δ6-17- Initiative (WHI) of adverse effects from the use acetoxyprogesterone; 6-chloro-Δ6-[17α] of a continuous combined estrogen–progestogen acetoxyprogesterone has affected prescribing. Patterns of exposure Structural and molecular formulae, and relative are also changing rapidly as the use of hormonal molecular mass therapy declines, the indications are restricted, O CH and the duration of the therapy is reduced (IARC, 3 C 2007). CH3 CH3 O C 1.1 Identification of the agents CH3 H O 1.1.1 Estrogens HH For Estrogens, see the Monograph on O Estrogen-only Menopausal Therapy in this Cl volume. C23H29ClO4 Relative molecular mass: 404.9 249 IARC MONOGRAPHS – 100A (b) Cyproterone acetate Structural and molecular formulae, and relative Chem. -

Use of Postmenopausal Hormone Therapy and Risk of Alzheimer’S BMJ: First Published As 10.1136/Bmj.L665 on 6 March 2019
RESEARCH Use of postmenopausal hormone therapy and risk of Alzheimer’s BMJ: first published as 10.1136/bmj.l665 on 6 March 2019. Downloaded from disease in Finland: nationwide case-control study Hanna Savolainen-Peltonen,1,2 Päivi Rahkola-Soisalo,1 Fabian Hoti,3 Pia Vattulainen,3 Mika Gissler,4,5,6 Olavi Ylikorkala,1 Tomi S Mikkola1,2 1University of Helsinki and ABSTRACT ratio 1.09, 95% confidence interval 1.05 to 1.14) and Helsinki University Hospital, OBJECTIVES those of oestrogen-progestogen (1.17, 1.13 to 1.21). Obstetrics and Gynecology, To compare the use of hormone therapy between The risk increases in users of oestrogen-progestogen Haartmaninkatu 2, PO Box 140, FIN-00029 HUS, 00029 Finnish postmenopausal women with and without a therapy were not related to different progestogens Helsinki, Finland diagnosis for Alzheimer’s disease. (noreth isterone acetate, medroxyprogesterone 2 Folkhälsan Research Center, DESIGN acetate, or other progestogens); but in women Biomedicum, Helsinki, Finland younger than 60 at hormone therapy initiation, 3 Nationwide case-control study. EPID Research Oy, Espoo, these risk increases were associated with hormone Finland SETTING therapy exposure over 10 years. Furthermore, the 4National Institute for Health Finnish national population and drug register, age at initiation of systemic hormone therapy was and Welfare, Helsinki, Finland between 1999 and 2013. 5Karolinska Institute, not a decisive determinant for the increase in risk Department of Neurobiology, PARTICIPANTS of Alzheimer’s disease. The exclusive use of vaginal Care Sciences and Society, All postmenopausal women (n=84 739) in Finland estradiol did not affect the risk of the disease (0.99, Division of Family Medicine, who, between 1999 and 2013, received a diagnosis of 0.96 to 1.01). -

Guidance on Bioequivalence Studies for Reproductive Health Medicines
Medicines Guidance Document 23 October 2019 Guidance on Bioequivalence Studies for Reproductive Health Medicines CONTENTS 1. Introduction........................................................................................................................................................... 2 2. Which products require a bioequivalence study? ................................................................................................ 3 3. Design and conduct of bioequivalence studies .................................................................................................... 4 3.1 Basic principles in the demonstration of bioequivalence ............................................................................... 4 3.2 Good clinical practice ..................................................................................................................................... 4 3.3 Contract research organizations .................................................................................................................... 5 3.4 Study design .................................................................................................................................................. 5 3.5 Comparator product ....................................................................................................................................... 6 3.6 Generic product .............................................................................................................................................. 6 3.7 Study subjects -

(ESI) for Integrative Biology. This Journal Is © the Royal Society of Chemistry 2017
Electronic Supplementary Material (ESI) for Integrative Biology. This journal is © The Royal Society of Chemistry 2017 Table 1 Enriched GO terms with p-value ≤ 0.05 corresponding to the over-expressed genes upon perturbation with the lung-toxic compounds. Terms with corrected p-value less than 0.001 are shown in bold. GO:0043067 regulation of programmed GO:0010941 regulation of cell death cell death GO:0042981 regulation of apoptosis GO:0010033 response to organic sub- stance GO:0043068 positive regulation of pro- GO:0010942 positive regulation of cell grammed cell death death GO:0006357 regulation of transcription GO:0043065 positive regulation of apop- from RNA polymerase II promoter tosis GO:0010035 response to inorganic sub- GO:0043066 negative regulation of stance apoptosis GO:0043069 negative regulation of pro- GO:0060548 negative regulation of cell death grammed cell death GO:0016044 membrane organization GO:0042592 homeostatic process GO:0010629 negative regulation of gene ex- GO:0001568 blood vessel development pression GO:0051172 negative regulation of nitrogen GO:0006468 protein amino acid phosphoryla- compound metabolic process tion GO:0070482 response to oxygen levels GO:0045892 negative regulation of transcrip- tion, DNA-dependent GO:0001944 vasculature development GO:0046907 intracellular transport GO:0008202 steroid metabolic process GO:0045934 negative regulation of nucle- obase, nucleoside, nucleotide and nucleic acid metabolic process GO:0006917 induction of apoptosis GO:0016481 negative regulation of transcrip- tion GO:0016125 sterol metabolic process GO:0012502 induction of programmed cell death GO:0001666 response to hypoxia GO:0051253 negative regulation of RNA metabolic process GO:0008203 cholesterol metabolic process GO:0010551 regulation of specific transcrip- tion from RNA polymerase II promoter 1 Table 2 Enriched GO terms with p-value ≤ 0.05 corresponding to the under-expressed genes upon perturbation with the lung-toxic compounds. -
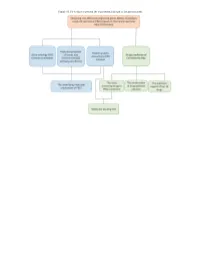
Figure S1. Flow Chart to Present the Experimental Design of the Present Study
Figure S1. Flow chart to present the experimental design of the present study. Figure S2. Volcano plot showing the differentially expressed genes between papillary renal cell carcinoma and non-cancer adjacent samples. Figure S3. The 2D molecular structures of the top 10 drugs. Table SI. The 60 potential drugs for papillary renal cell carcinoma treatment based on differently expressed genes. Drug name Dose Cell line Score Instance ID Pinacidil (C13H19N5) 16 µM HL60 -1 2406 Ciclosporin (C62H111N11O12) 3 µM HL60 -0.994 1331 Naftifine (C21H21N) 12 µM HL60 -0.966 2974 Vorinostat (C14H20N2O3) 10 µM HL60 -0.961 1161 Metacycline (C22H22N2O8) 8 µM HL60 -0.943 2901 Sulfacetamide (C8H10N2O3S) 16 µM HL60 -0.939 1859 Chlorprothixene (C18H18ClNS) 11 µM HL60 -0.938 1272 Amiodarone (C25H29I2NO3) 6 µM HL60 -0.933 2434 Noretynodrel (C20H26O2) 13 µM HL60 -0.914 1860 Valproic acid (C8H16O2) 200 µM HL60 -0.91 1155 Oxolinic acid (C13H11NO5) 15 µM HL60 -0.903 1419 Monocrotaline (C16H23NO6) 12 µM PC3 -0.881 7127 LY-294002 (C19H17NO3) 10 µM HL60 -0.869 1168 Aceclofenac (C16H13Cl2NO4) 11 µM PC3 -0.863 7269 Hyoscyamine (C17H23NO3) 14 µM HL60 -0.859 1424 Heliotrine (C16H27NO5) 13 µM HL60 -0.85 2180 Trichostatin A (C17H22N2O3) 1 µM HL60 -0.85 1175 Sulfachlorpyridazine (C10H8ClN4O2S) 14 µM HL60 -0.85 2191 Diflorasone (C22H28F2O5) 8 µM HL60 -0.838 2142 Yohimbine (C21H26N2O3) 10 µM MCF7 -0.833 6777 Sulfadiazine (C10H10N4O2S) 16 µM HL60 -0.833 1852 Pargyline (C11H13N) 20 µM PC3 -0.832 2102 Haloperidol (C21H23ClFNO2) 10 µM HL60 -0.829 6203 Methazolamide (C5H8N4O3S2) 17 -

Dosage Units of Steroids with a Seal Coating for the Steroids
~™ iiiiiii nun iiiiiiii inn mil mil inn iiiii inn urn mill mi mi m (19) J European Patent Office Office europeen des brevets (11) EP 0 927 556 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication:ation: (51) |nt. ci.6: A61 K 31/565, A61 K 9/28 07.07.1999 Bulletin 1999/27 (21) Application number: 97202908.6 (22) Date of filing: 23.09.1997 (84) Designated Contracting States: (72) Inventor: HARTHOLT GERTJAN PETER AT BE CH DE DK ES Fl FR GB GR IE IT LI LU MC 5406 BD UDEN (NL) NL PT SE (74) Representative: Kraak, Hajo (71) Applicant: Akzo Nobel N.V. N.V. Organon, 6824 BM Arnhem (NL) Postbus 20 5340 BH OSS (NL) (54) Dosage units of steroids with a seal coating for the steroids (57) Dosage units, such as tablet cores and gran- ules, containing a composition comprising a steroid having two hydrogen atoms at position 3 of the steroid skeleton, are coated. While a conventional sugar coat- ing process involves the application of several layers including, as a surface layer, a sugar coat, it was found that a seal coat such as normally used in a sugar coat- ing process can be used as the surface layer for the pur- pose of preventing the transfer of the steroid out of the composition. From this novel use of a seal coat, novel dosage units result. The process is advantageous, as it requires fewer steps than before. < CO LO LO r»- Csl o Q_ LU Printed by Xerox (UK) Business Services 2.16.7/3.6 1 EP 0 927 556 A1 Description ing the dosage unit to coating steps comprising the application of a seal coat. -

Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DIX to the HTSUS—Continued
20558 Federal Register / Vol. 60, No. 80 / Wednesday, April 26, 1995 / Notices DEPARMENT OF THE TREASURY Services, U.S. Customs Service, 1301 TABLE 1.ÐPHARMACEUTICAL APPEN- Constitution Avenue NW, Washington, DIX TO THE HTSUSÐContinued Customs Service D.C. 20229 at (202) 927±1060. CAS No. Pharmaceutical [T.D. 95±33] Dated: April 14, 1995. 52±78±8 ..................... NORETHANDROLONE. A. W. Tennant, 52±86±8 ..................... HALOPERIDOL. Pharmaceutical Tables 1 and 3 of the Director, Office of Laboratories and Scientific 52±88±0 ..................... ATROPINE METHONITRATE. HTSUS 52±90±4 ..................... CYSTEINE. Services. 53±03±2 ..................... PREDNISONE. 53±06±5 ..................... CORTISONE. AGENCY: Customs Service, Department TABLE 1.ÐPHARMACEUTICAL 53±10±1 ..................... HYDROXYDIONE SODIUM SUCCI- of the Treasury. NATE. APPENDIX TO THE HTSUS 53±16±7 ..................... ESTRONE. ACTION: Listing of the products found in 53±18±9 ..................... BIETASERPINE. Table 1 and Table 3 of the CAS No. Pharmaceutical 53±19±0 ..................... MITOTANE. 53±31±6 ..................... MEDIBAZINE. Pharmaceutical Appendix to the N/A ............................. ACTAGARDIN. 53±33±8 ..................... PARAMETHASONE. Harmonized Tariff Schedule of the N/A ............................. ARDACIN. 53±34±9 ..................... FLUPREDNISOLONE. N/A ............................. BICIROMAB. 53±39±4 ..................... OXANDROLONE. United States of America in Chemical N/A ............................. CELUCLORAL. 53±43±0 -

Evidence Review H: Management Options for Refractory Acne
National Institute for Health and Care Excellence Final Acne vulgaris: management [H] Management options for refractory acne NG198 Evidence reviews underpinning recommendations 1.5.15 and 1.5.16 as well as 1.6.1 to 1.6.4 in the NICE guideline June 2021 Final These evidence reviews were developed by the National Guideline Alliance which is a part of the Royal College of Obstetricians and Gynaecologists FINAL Error! No text of specified style in document. Disclaimer The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available. When exercising their judgement, professionals are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or service users. The recommendations in this guideline are not mandatory and the guideline does not override the responsibility of healthcare professionals to make decisions appropriate to the circumstances of the individual patient, in consultation with the patient and/or their carer or guardian. Local commissioners and/or providers have a responsibility to enable the guideline to be applied when individual health professionals and their patients or service users wish to use it. They should do so in the context of local and national priorities for funding and developing services, and in light of their duties to have due regard to the need to eliminate unlawful discrimination, to advance equality of opportunity and to reduce health inequalities. Nothing in this guideline should be interpreted in a way that would be inconsistent with compliance with those duties. NICE guidelines cover health and care in England.