Comparison of Long-Term Cidr-Based Protocols to Synchronize Estrus and Ovulation Prior to Fixed-Time Artificial Insemination in Postpartum Beef Cows
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Rapid and Effective Nonsurgical Artificial Insemination Protocol Using
Transgenic Res (2015) 24:775–781 DOI 10.1007/s11248-015-9887-3 TECHNICAL REPORT A rapid and effective nonsurgical artificial insemination protocol using the NSETTM device for sperm transfer in mice without anesthesia Barbara J. Stone . Kendra H. Steele . Angelika Fath-Goodin Received: 27 January 2015 / Accepted: 3 June 2015 / Published online: 12 June 2015 Ó The Author(s) 2015. This article is published with open access at Springerlink.com Abstract Artificial insemination (AI) is an assisted Introduction reproductive technique that is implemented success- fully in humans as a fertility treatment, performed Artificial insemination (AI) is used to assist in extensively for commercial breeding of livestock, and reproduction by directly delivering motile sperm to is also successful in laboratory rodents. AI in the the female reproductive tract. Artificial insemination mouse may be especially useful for breeding of in mice can be useful as an alternative to breeding for transgenic or mutant mice with fertility problems, strains which are not easily bred due to physical or expansion of mouse colonies, and as an alternative to behavioral issues. The techniques of AI and in vitro in vitro fertilization. Nonsurgical AI techniques for the fertilization (IVF) can both be used to generate viable mouse have been described previously but are not embryos. Development after AI simply continues in often implemented due to technical difficulties. Here the recipient female, while embryos generated from we compare various protocols for preparation of CD1 IVF must be transferred to a pseudopregnant recipient. recipients prior to AI for naı¨ve (in estrus), ovulation- AI or IVF can be used to generate embryos after induced, and superovulated females. -

The Artificial Way
SPOTLIGHT Breeding Rhinos for the future The Artificial way BY FELIX PATTON aptive breeding programmes have many decades about the husbandry and had some spectacular successes in reproductive needs of captive Black and 310 Csaving species and reintroducing Indian rhinos, when applied to White them into the wild. All rhino species Number of oestrous cycles rhinos, it has been unsuccessful. The are at risk from poaching, disease and non-reproducing White rhino main problem has been the absence of or natural disaster. Developing captive female in captivity can exhibit. erratic nature of oestrous cycle activity in populations is an essential safeguard to over half of the females in the European avoid extinction but breeding success has and North American Species Survival been limited. If a female rhino does not Program. get pregnant, her uterus starts to develop White rhino females in the wild irreversible problems, such as cysts To date, captive reproduction in usually experience short intervals and tumours. Can the failure to become the White rhino has been poor. Despite between successive births, even as pregnant be overcome? the wealth of knowledge acquired over little as 18 months. This indicates that pregnancy and lactation are the most common endocrine profile with possibly as few as 30 oestrous cycles per reproductive life span. Ultrasound technology for use in rhinoceros has been A reproducing female White rhino in used in well over 150 reproductive assessments in more captivity may produce up to nine calves. With a pregnancy of 16 months and than 70 rhinos with the result that the causes of poor subsequent lactation of approximately captive performance are now known. -

Artificial Insemination
Ch36-A03309.qxd 1/23/07 5:16 PM Page 539 Section 6 Infertility and Recurrent Pregnancy Loss Chapter Artificial Insemination 36 Ashok Agarwal and Shyam S. R. Allamaneni INTRODUCTION widely available, the terms homologous artificial insemination and heterologous artificial insemination were used to differentiate Artificial insemination is an assisted conception method that can these two alternative sources. However, the use of these bio- be used to alleviate infertility in selected couples. The rationale medical terms in this manner is at variance with their scientific behind the use of artificial insemination is to increase the gamete meaning, where they denote different species or organisms (as in, density near the site of fertilization.1 The effectiveness of artificial e.g., homologous and heterologous tissue grafts). insemination has been clearly established in specific subsets of In the latter half of the 20th century, the terms artificial infertile patients such as those with idiopathic infertility, infertility insemination, donor (AID) and artificial insemination, husband related to a cervical factor, or a mild male factor infertility (AIH) found common use. However, the widespread use of the (Table 36-1).2,3 An accepted advantage of artificial insemination acronym AIDS for acquired immunodeficiency syndrome resulted is that it is generally less expensive and invasive than other in the replacement of AID with therapeutic donor insemination assisted reproductive technology (ART) procedures.4 (TDI). An analogous alternative term for AIH has not evolved, This chapter provides a comprehensive description of probably in part because of the increasingly common situation indications for artificial insemination, issues to consider before where the woman’s partner is not her legal husband. -

In Vitro Fertilization and Artificial Insemination: Ethical Consideration
Loyola University Chicago Loyola eCommons Dissertations Theses and Dissertations 1997 In Vitro Fertilization and Artificial Insemination: Ethical Consideration Joseph Ibegbulem Ekweariri Loyola University Chicago Follow this and additional works at: https://ecommons.luc.edu/luc_diss Part of the Philosophy Commons Recommended Citation Ekweariri, Joseph Ibegbulem, "In Vitro Fertilization and Artificial Insemination: Ethical Consideration" (1997). Dissertations. 3716. https://ecommons.luc.edu/luc_diss/3716 This Dissertation is brought to you for free and open access by the Theses and Dissertations at Loyola eCommons. It has been accepted for inclusion in Dissertations by an authorized administrator of Loyola eCommons. For more information, please contact [email protected]. This work is licensed under a Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 License. Copyright © 1997 Joseph Ibegbulem Ekweariri LOYOLA UNIVERSITY OF CHICAGO IN VITRO FERTILIZATION AND ARTIFICIAL INSEMINATION: ETHICAL CONSIDERATION A DISSERTATION SUBMITTED TO THEFACULTYOFTHEGRADUATESCHOOL IN CANDIDACY FOR THE DEGREE OF DOCTOR OF PHILOSOPHY DEPARTMENT OF PHILOSOPHY BY JOSEPH IBEGBULEM EKWEARIRI CHICAGO, ILLINOIS MAY, 1997 Copyright by Joseph Ibegbulem Ekweariri, 1997 All rights reserved. ACKNOWLEDGMENTS I thank you God for my life; even when ill-health threatened my studies and this work you sustained me throughout. Many people contributed to the success of this work in different capacities. I wish to thank them. I am grateful to my dissertation committee: Prof. David T. Ozar, the Chairman of the committee and director of my dissertation, Prof. Richard Westley, and Prof. John Langan S.J. I wish to thank all my professors at Loyola University, especially Prof. Kenneth Thompson, the director of the ~raduate School of Philosophy and Max Caproni Asst. -

The Protection of the Human Embryo in Vitro
Strasbourg, 19 June 2003 CDBI-CO-GT3 (2003) 13 STEERING COMMITTEE ON BIOETHICS (CDBI) THE PROTECTION OF THE HUMAN EMBRYO IN VITRO Report by the Working Party on the Protection of the Human Embryo and Fetus (CDBI-CO-GT3) Table of contents I. General introduction on the context and objectives of the report ............................................... 3 II. General concepts............................................................................................................................... 4 A. Biology of development ....................................................................................................................... 4 B. Philosophical views on the “nature” and status of the embryo............................................................ 4 C. The protection of the embryo............................................................................................................... 8 D. Commercialisation of the embryo and its parts ................................................................................... 9 E. The destiny of the embryo ................................................................................................................... 9 F. “Freedom of procreation” and instrumentalisation of women............................................................10 III. In vitro fertilisation (IVF).................................................................................................................. 12 A. Presentation of the procedure ...........................................................................................................12 -

Assisted Reproduction in Developing Countries—Facing up to the Issues
1 Progress, No. 63 No. 63 2003 UNDP/UNFPA/WHO/World Assisted reproduction in developing Bank Special Programme of Research, Development and countriesfacing up to the issues Research Training in Human Reproduction (HRP) For millions of couples around the Attitudes to assisted reproduction are Department of Reproductive world, the inability to have children is very much influenced by the specific Health and Research, a personal tragedy. For a significant cultural and social context. Manipula- World Health Organization, proportion of them, the private agony tion of sperm, oocytes and embryos Geneva, Switzerland is compounded by a social stigma, raises numerous questions to which which can have serious and far-reach- there are no easy answers, and which Launched by the World Health ing consequences. It is not surprising often arouse strong emotions. The Organization in 1972, the UNDP/ therefore that the demand for as- article on page 5 outlines some of the UNFPA/WHO/World Bank sisted reproductive technologies ethical and legal issues surrounding Special Programme of (ART) is growing in all regions. But the use of ART, which need to be Research, Development and how can the provision of these tech- discussed openly by all involved par- Research Training in Human nologieswhich are expensive and ties wherever such services are made Reproduction is a global have a success rate of less than available. programme of technical 30%be justified in developing coun- cooperation. It promotes, tries with poorly developed health In the 25 years since the birth of the coordinates, supports, conducts, services that are still struggling with first baby resulting from in vitro ferti- and evaluates research on infectious and chronic diseases, such lization, there have been tremendous reproductive health, with as tuberculosis, malaria and HIV/ advances in scientific techniques and particular reference to the needs AIDS? medical applications of ART. -
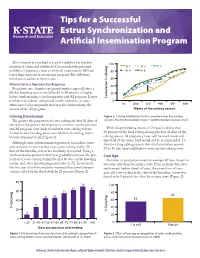
MF2574 Tips for a Successful Estrus Synchronization and Artificial
Tips for a Successful Estrus Synchronization and Artificial Insemination Program Do you know if your herd is a good candidate for synchro- 100 Yr 1 Yr 2 Yr 3 nization of estrus and ovulation? Can you identify potential problems if pregnancy rates to artificial insemination (AI) are 80 Yr 4 Yr 5 lower than expected in an existing program? The following information addresses these issues. 60 Natural Service Reproductive Response 40 22% Pregnancy rates (number pregnant/number exposed) after a 60-day breeding season should be 85 to 90 percent or higher 20 before implementing a synchronization and AI program. Lower % claving Cumulative 0 fertility may indicate suboptimal health, nutrition, or some 1st 2nd 3rd 6th5th4th other aspect of management that may be compromising the Weeks of the calving season success of the AI program. Calving Distribution Figure 2. Calving distribution for the same herd over five calving seasons. No synchronization in year 1; synchronization in years 2 to 5. The greater the proportion of cows calving the first 21 days of the season, the greater the response to an estrus synchronization and AI program. One study found that cows calving at least With longer breeding seasons (>70 days) and less than 71 days before breeding were more likely to be cycling and to 60 percent of the herd calving during the first 42 days of the become pregnant to AI (Figure 1). calving season, AI pregnancy rates, will be much lower and timed AI of the entire herd would not be recommended. To Although some synchronization protocols can induce estrus shorten a long calving season, two synchronization periods and ovulation in non-cycling cows, cows calving within 30 20 to 30 days apart will help to move up late-calving cows. -

The Effect of Expression of Estrus at Artificial Insemination on Target Genes in the Endometrium, Conceptus and Corpus Luteum of Beef Cows
THE EFFECT OF EXPRESSION OF ESTRUS AT ARTIFICIAL INSEMINATION ON TARGET GENES IN THE ENDOMETRIUM, CONCEPTUS AND CORPUS LUTEUM OF BEEF COWS by Saeideh Davoodi A THESIS SUBMITTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE in The Faculty of Graduate and Postdoctoral Studies (Applied Animal Biology) THE UNIVERSITY OF BRITISH COLUMBIA (Vancouver) April 2015 © Saeideh Davoodi, 2015 Abstract The aim of this study was to test the effect of expression of estrus at artificial insemination (AI) on the endometrium, conceptus and corpus luteum (CL) gene expression. Twenty-three multiparous non-lactating Nelore cows were enrolled on an estradiol (E2) and progesterone (P4) based timed-AI protocol (AI = d 0), then slaughtered for endometrium, CL and conceptus collection on d 19. Body condition score (BCS), blood samples and ultrasound examination was performed on d 0, 7 and 18 of the experiment followed by RNA extraction and quantitative reverse transcription polymerase chain reaction (qRT-PCR) analysis of 58 target genes. Data was checked for normality and analysed by ANOVA for repeated measures using proc GLM, MIXED and UNIVARIATE. Estrous expression had no correlation with parameters such as BCS, pre-ovulatory follicle and CL diameter, P4 concentration in plasma on d 7 and 18 after AI and IFN-tau concentration in the uterine flushing (P > 0.05); however, a significant increase was observed in conceptus size (P = 0.02; 38.3 ± 2.8 vs 28.2 ± 2.9). The majority of transcripts affected by estrous expression in the endometrium belong to the immune system and adhesion molecule family (MX1, MX2, MYL12A, MMP19, CXCL10, IGLL1 and SLPI) (P ≤ 0.05). -

MG Infertility Services Commercial
Infertility Services — Commercial Last Review Date: June 11, 2021 Number: MG.MM.ME.53f Medical Guideline Disclaimer Property of EmblemHealth. All rights reserved. The treating physician or primary care provider must submit to EmblemHealth the clinical evidence that the patient meets the criteria for the treatment or surgical procedure. Without this documentation and information, EmblemHealth will not be able to properly review the request for prior authorization. The clinical review criteria expressed below reflects how EmblemHealth determines whether certain services or supplies are medically necessary. EmblemHealth established the clinical review criteria based upon a review of currently available clinical information (including clinical outcome studies in the peer reviewed published medical literature, regulatory status of the technology, evidence-based guidelines of public health and health research agencies, evidence-based guidelines and positions of leading national health professional organizations, views of physicians practicing in relevant clinical areas, and other relevant factors). EmblemHealth expressly reserves the right to revise these conclusions as clinical information changes and welcomes further relevant information. Each benefit program defines which services are covered. The conclusion that a particular service or supply is medically necessary does not constitute a representation or warranty that this service or supply is covered and/or paid for by EmblemHealth, as some programs exclude coverage for services or supplies that EmblemHealth considers medically necessary. If there is a discrepancy between this guideline and a member's benefits program, the benefits program will govern. In addition, coverage may be mandated by applicable legal requirements of a state, the Federal Government or the Centers for Medicare & Medicaid Services (CMS) for Medicare and Medicaid members. -

What Is Intrauterine Insemination? Updated: December 2018
What is Intrauterine Insemination? Updated: December 2018 Intrauterine insemination (IUI) is a low tech form of assisted reproductive technology, which is typically performed in a doctor’s office. When individuals or couples begin • Blocked fallopian tubes or tubal fertility treatment, their health damage care professional may recommend • Ovarian failure or lack of ovulation Intrauterine insemination for several reasons, some of which include: • Significantly reduced ovarian reserve • Advanced endometriosis • Issues with cervical mucus • Unexplained Infertility Sometimes referred to as artificial insemination, IUIs are typically the • Ovulation problems, such as those first procedure utilized if someone is that occur with polycystic ovarian interested in assisted reproductive syndrome (PCOS) support. IUIs can be done as a natural • Mild endometriosis cycle without the use of any fertility medication, or with drugs such as clomid Intrauterine insemination is not or injected gonadotropins1. IUI can be considered to be an effective treatment timed via ovulation induction protocols, for: as when the patient’s ovulation is being Advancing Equality for LGBTQ Families www.familyequality.org 1 monitored by a physician or, through a speculum will be inserted into the the use of ovulation predictor kits that vagina and then a thin, flexible catheter, the patient utilizes at home2. or tube, will be placed into the cervix. Washed sperm are then injected directly If you have been prescribed fertility into the uterus with a syringe. The medication in conjunction with IUI, your procedure is only mildly uncomfortable physician will monitor your ovarian for most individuals and takes less than follicle growth (length and number of five minutes from start to finish. -

Pregnancy Rate Following Artificial Insemination Or Natural Service In
Turk. J. Vet. Anim. Sci. 2012; 36(4): 451-455 © TÜBİTAK Short Communication doi:10.3906/vet-1008-15 Pregnancy rate following artifi cial insemination or natural service in postpartum estrus synchronized beef cattle Abdul MALIK1,2, Abd Wahid HARON2*, Rosnina YUSOFF2, Azhar KASIM3, Sabri Mohd YUSOFF4 1Department of Animal Science, Faculty of Agriculture, Islamic Kalimantan University, Banjarmasin - INDONESIA 2Department of Veterinary Clinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan - MALAYSIA 3Department of Animal Science, Faculty of Agriculture. Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan - MALAYSIA 4Department of Veterinary Pathology and Microbiology, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan - MALAYSIA Received: 10.08.2010 ● Accepted: 29.03.2011 Abstract: Th e objective of this study was to investigate the pregnancy rate following artifi cial insemination (AI) or natural service (NS) in postpartum estrous synchronized beef cattle. A total of 85 Brangus cows, postpartum, were divided into 2 groups. Group 1 consisted of 35 cows and was further subdivided into 7 groups, each consisted of 5 cows that were naturally mated. Group 2 consisted of 40 cows that were artifi cially inseminated. Estrus synchronization was carried out using a controlled internal drug releasing device (CIDR) containing 1.38 mg of progesterone, for 7 days. Group 1 cows were mixed with bulls at a 1:5 ratio of bull:cows for 7 days and group 2 cows were inseminated 55-58 h aft er the CIDR removal. Pregnancy was determined using transrectal ultrasonography 32 days aft er NS and AI. -

Management of Klinefelter Syndrome During Transition
I Gies and others Klinefelter syndrome during 171:2 R67–R77 Review transition TRANSITION IN ENDOCRINOLOGY Management of Klinefelter syndrome during transition Inge Gies1,3,†, David Unuane2,3, Brigitte Velkeniers2,3 and Jean De Schepper1,2,3 Correspondence 1Division of Pediatric Endocrinology, 2Department of Endocrinology and 3Klinefelter Clinic, UZ Brussel, should be addressed Vrije Universiteit Brussel, Brussels, Belgium to I Gies †(I Gies is now at Department of Pediatrics, UZ Brussel, Laarbeeklaan 101, 1090 Brussels, Belgium) Email [email protected] Abstract Klinefelter syndrome (KS) is the most common sex chromosomal disorder in males. Key findings in older adolescents and young men are small testes with variable hypo-androgenism, but almost universal azoospermia, most frequently in combination with a history of learning difficulties and behavior problems. Males with KS may come to medical attention through different medical presentations, given its association with several congenital malformations, and psychiatric, endocrine, and metabolic disorders. Preventive care is to be provided from diagnosis, preferentially through a multidisciplinary approach, including that from an endocrinologist, clinical psychologist or psychiatrist, neurologist, urologist, geneticist, sexologist, and a fertility team. Accurate information about the condition and assessment of associated medical conditions should be offered at diagnosis and should be followed by psychological counseling. Medical treatment during transition into adulthood is focused on fertility preservation and testosterone replacement therapy in the case of hypo-androgenism, and alleviation of current or future consequences of testicular fibrosis. However, more research is needed to determine the need for pro-active testosterone treatment in adolescence, as well as the conditions for an optimal testosterone replacement and sperm retrieval in adolescents and young men with KS.