Effect of GPVI-Fc in Combination with Standard Antiplatelet Drugs and of GPVI-Fc Fused to CD39 on Platelet Thrombus Formation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
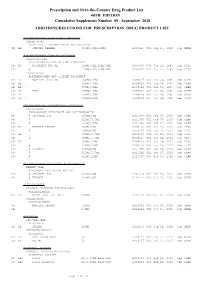
Additions and Deletions to the Drug Product List
Prescription and Over-the-Counter Drug Product List 40TH EDITION Cumulative Supplement Number 09 : September 2020 ADDITIONS/DELETIONS FOR PRESCRIPTION DRUG PRODUCT LIST ACETAMINOPHEN; BUTALBITAL; CAFFEINE TABLET;ORAL BUTALBITAL, ACETAMINOPHEN AND CAFFEINE >A> AA STRIDES PHARMA 325MG;50MG;40MG A 203647 001 Sep 21, 2020 Sep NEWA ACETAMINOPHEN; CODEINE PHOSPHATE SOLUTION;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >D> AA WOCKHARDT BIO AG 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC >A> @ 120MG/5ML;12MG/5ML A 087006 001 Jul 22, 1981 Sep DISC TABLET;ORAL ACETAMINOPHEN AND CODEINE PHOSPHATE >A> AA NOSTRUM LABS INC 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >A> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >A> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN >D> AA TEVA 300MG;15MG A 088627 001 Mar 06, 1985 Sep CAHN >D> AA 300MG;30MG A 088628 001 Mar 06, 1985 Sep CAHN >D> AA ! 300MG;60MG A 088629 001 Mar 06, 1985 Sep CAHN ACETAMINOPHEN; HYDROCODONE BITARTRATE TABLET;ORAL HYDROCODONE BITARTRATE AND ACETAMINOPHEN >A> @ CEROVENE INC 325MG;5MG A 211690 001 Feb 07, 2020 Sep CAHN >A> @ 325MG;7.5MG A 211690 002 Feb 07, 2020 Sep CAHN >A> @ 325MG;10MG A 211690 003 Feb 07, 2020 Sep CAHN >D> AA VINTAGE PHARMS 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >A> @ 300MG;5MG A 090415 001 Jan 24, 2011 Sep DISC >D> AA 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >A> @ 300MG;7.5MG A 090415 002 Jan 24, 2011 Sep DISC >D> AA 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >A> @ 300MG;10MG A 090415 003 Jan 24, 2011 Sep DISC >D> @ XIROMED 325MG;5MG A 211690 -

Distinct but Critical Roles for Integrin Aiibb3 in Platelet Lamellipodia Formation on Fibrinogen, Collagen-Related Peptide and T
Distinct but critical roles for integrin aIIbb3 in platelet lamellipodia formation on fibrinogen, collagen-related peptide and thrombin Kelly Thornber1, Owen J. T. McCarty2,3, Steve P. Watson2 and Catherine J. Pears1 1 Department of Biochemistry, University of Oxford, UK 2 Centre for Cardiovascular Sciences, Institute of Biomedical Research, University of Birmingham, UK 3 Department of Biomedical Engineering, Oregon Health & Science University, Portland, OR, USA Keywords Integrins are the major receptor type known to facilitate cell adhesion and aIIbb3; adhesion; integrins; lamellipodia; lamellipodia formation on extracellular matrix proteins. However, collagen- platelets related peptide and thrombin have recently been shown to mediate platelet lamellipodia formation when presented as immobilized surfaces. The aims Correspondence C. Pears, Department of Biochemistry, of this study were to establish if there exists a role for the platelet integrin South Parks Road, University of Oxford, aIIbb3 in this response; and if so, whether signalling from the integrin is Oxford, OX1 3QU, UK required for lamellipodia formation on these surfaces. Real-time analysis Fax: +44 1865 275259 was used to compare platelet morphological changes on surfaces of fibrino- Tel: +44 1865 275737 gen, collagen-related peptide or thrombin in the presence of various E-mail: [email protected] pharmacological inhibitors and platelets from ‘knockout’ mice. We demon- Website: http://www.bioch.ox.ac.uk strate that collagen-related peptide and thrombin stimulate distinct patterns 2+ (Received 11 July 2006, revised 22 August of platelet lamellipodia formation and elevation of intracellular Ca to 2006, accepted 12 September 2006) that induced by the integrin aIIbb3 ligand, fibrinogen. -

Hemoglobin Interaction with Gp1ba Induces Platelet Activation And
ARTICLE Platelet Biology & its Disorders Hemoglobin interaction with GP1bα induces platelet activation and apoptosis: a novel mechanism associated with intravascular hemolysis Rashi Singhal,1,2,* Gowtham K. Annarapu,1,2,* Ankita Pandey,1 Sheetal Chawla,1 Amrita Ojha,1 Avinash Gupta,1 Miguel A. Cruz,3 Tulika Seth4 and Prasenjit Guchhait1 1Disease Biology Laboratory, Regional Centre for Biotechnology, National Capital Region, Biotech Science Cluster, Faridabad, India; 2Biotechnology Department, Manipal University, Manipal, Karnataka, India; 3Thrombosis Research Division, Baylor College of Medicine, Houston, TX, USA, and 4Hematology, All India Institute of Medical Sciences, New Delhi, India *RS and GKA contributed equally to this work. ABSTRACT Intravascular hemolysis increases the risk of hypercoagulation and thrombosis in hemolytic disorders. Our study shows a novel mechanism by which extracellular hemoglobin directly affects platelet activation. The binding of Hb to glycoprotein1bα activates platelets. Lower concentrations of Hb (0.37-3 mM) significantly increase the phos- phorylation of signaling adapter proteins, such as Lyn, PI3K, AKT, and ERK, and promote platelet aggregation in vitro. Higher concentrations of Hb (3-6 mM) activate the pro-apoptotic proteins Bak, Bax, cytochrome c, caspase-9 and caspase-3, and increase platelet clot formation. Increased plasma Hb activates platelets and promotes their apoptosis, and plays a crucial role in the pathogenesis of aggregation and development of the procoagulant state in hemolytic disorders. Furthermore, we show that in patients with paroxysmal nocturnal hemoglobinuria, a chronic hemolytic disease characterized by recurrent events of intravascular thrombosis and thromboembolism, it is the elevated plasma Hb or platelet surface bound Hb that positively correlates with platelet activation. -

A Comparative Study of Molecular Structure, Pka, Lipophilicity, Solubility, Absorption and Polar Surface Area of Some Antiplatelet Drugs
International Journal of Molecular Sciences Article A Comparative Study of Molecular Structure, pKa, Lipophilicity, Solubility, Absorption and Polar Surface Area of Some Antiplatelet Drugs Milan Remko 1,*, Anna Remková 2 and Ria Broer 3 1 Department of Pharmaceutical Chemistry, Faculty of Pharmacy, Comenius University in Bratislava, Odbojarov 10, SK-832 32 Bratislava, Slovakia 2 Department of Internal Medicine, Faculty of Medicine, Slovak Medical University, Limbová 12, SK–833 03 Bratislava, Slovakia; [email protected] 3 Department of Theoretical Chemistry, Zernike Institute for Advanced Materials, University of Groningen, Nijenborgh 4, 9747 AG Groningen, The Netherlands; [email protected] * Correspondence: [email protected]; Tel.: +421-2-5011-7291 Academic Editor: Michael Henein Received: 18 February 2016; Accepted: 11 March 2016; Published: 19 March 2016 Abstract: Theoretical chemistry methods have been used to study the molecular properties of antiplatelet agents (ticlopidine, clopidogrel, prasugrel, elinogrel, ticagrelor and cangrelor) and several thiol-containing active metabolites. The geometries and energies of most stable conformers of these drugs have been computed at the Becke3LYP/6-311++G(d,p) level of density functional theory. Computed dissociation constants show that the active metabolites of prodrugs (ticlopidine, clopidogrel and prasugrel) and drugs elinogrel and cangrelor are completely ionized at pH 7.4. Both ticagrelor and its active metabolite are present at pH = 7.4 in neutral undissociated form. The thienopyridine prodrugs ticlopidine, clopidogrel and prasugrel are lipophilic and insoluble in water. Their lipophilicity is very high (about 2.5–3.5 logP values). The polar surface area, with regard to the structurally-heterogeneous character of these antiplatelet drugs, is from very large interval of values of 3–255 Å2. -

Health and Social Outcomes Associated with High-Risk Alcohol Use
Manitoba Centre for Health Policy Health and Social Outcomes Associated with High-Risk Alcohol Use Summer 2018 Nathan C Nickel, MPH, PhD Jeff Valdivia, MNRM, CAPM Deepa Singal, PhD James Bolton, MD Christine Leong, PharmD Susan Burchill, BMus Leonard MacWilliam, MSc, MNRM Geoffrey Konrad, MD Randy Walld, BSc, BComm (Hons) Okechukwu Ekuma, MSc Greg Finlayson, PhD Leanne Rajotte, BComm (Hons) Heather Prior, MSc Josh Nepon, MD Michael Paille, BHSc This report is produced and published by the Manitoba Centre for Health Policy (MCHP). It is also available in PDF format on our website at: http://mchp-appserv.cpe.umanitoba.ca/deliverablesList.html Information concerning this report or any other report produced by MCHP can be obtained by contacting: Manitoba Centre for Health Policy Rady Faculty of Health Sciences Max Rady College of Medicine, University of Manitoba 4th Floor, Room 408 727 McDermot Avenue Winnipeg, Manitoba, Canada R3E 3P5 Email: [email protected] Phone: (204) 789-3819 Fax: (204) 789-3910 How to cite this report: Nathan C Nickel, James Bolton, Leonard MacWilliam, Okechukwu Ekuma, Heather Prior, Jeff Valdivia, Christine Leong, Geoffrey Konrad, Greg Finlayson, Josh Nepon, Deepa Singal, Susan Burchill, Randy Walld, Leanne Rajotte, Michael Paille. Health and Social Outcomes Associated with High-Risk Alcohol Use. Winnipeg, MB. Manitoba Centre for Health Policy, Summer 2018. Legal Deposit: Manitoba Legislative Library National Library of Canada ISBN 978-1-896489-90-2 ©Manitoba Health This report may be reproduced, in whole or in part, provided the source is cited. 1st printing (Summer 2018) This report was prepared at the request of Manitoba Health, Seniors and Active Living (MHSAL), a department within the Government of Manitoba, as part of the contract between the University of Manitoba and MHSAL. -

The Delivery Strategy of Paclitaxel Nanostructured Lipid Carrier Coated with Platelet Membrane
cancers Article The Delivery Strategy of Paclitaxel Nanostructured Lipid Carrier Coated with Platelet Membrane 1, 1, 1 2 3 Ki-Hyun Bang y, Young-Guk Na y , Hyun Wook Huh , Sung-Joo Hwang , Min-Soo Kim , Minki Kim 1, Hong-Ki Lee 1,* and Cheong-Weon Cho 1,* 1 College of Pharmacy, Chungnam National University, Daejeon 34134, Korea; [email protected] (K.-H.B.); [email protected] (Y.-G.N.); [email protected] (H.W.H.); [email protected] (M.K.) 2 College of Pharmacy and Yonsei Institute of Pharmaceutical Sciences, Yonsei University, 162-1 Songdo-dong, Yeonsu-gu, Incheon 406-840, Korea; [email protected] 3 College of Pharmacy, Pusan National University, 63 Busandaehak-ro, Geumjeong-gu, Busan 609-735, Korea; [email protected] * Correspondence: [email protected] (H.-K.L.); [email protected] (C.-W.C.); Tel.: +82-42-821-5934 (H.-K.L. & C.-W.C.); Fax: +82-42-823-6566 (H.-K.L. & C.-W.C.) These authors contributed equally. y Received: 2 May 2019; Accepted: 10 June 2019; Published: 11 June 2019 Abstract: Strategies for the development of anticancer drug delivery systems have undergone a dramatic transformation in the last few decades. Lipid-based drug delivery systems, such as a nanostructured lipid carrier (NLC), are one of the systems emerging to improve the outcomes of tumor treatments. However, NLC can act as an intruder and cause an immune response. To overcome this limitation, biomimicry technology was introduced to decorate the surface of the nanoparticles with various cell membrane proteins. Here, we designed paclitaxel (PT)-loaded nanostructured lipid carrier (PT-NLC) with platelet (PLT) membrane protein because PLT is involved with angiogenesis and interaction of circulating tumor cells. -

A Microfluidic Approach for Evaluating Novel Antithrombotic Targets
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2017 A Microfluidic Approach For Evaluating Novel Antithrombotic Targets Shu Zhu University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Chemical Engineering Commons Recommended Citation Zhu, Shu, "A Microfluidic Approach For Evaluating Novel Antithrombotic Targets" (2017). Publicly Accessible Penn Dissertations. 2670. https://repository.upenn.edu/edissertations/2670 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/2670 For more information, please contact [email protected]. A Microfluidic Approach For Evaluating Novel Antithrombotic Targets Abstract Microfluidic systems allow precise control of the anticoagulation/pharmacology protocols, defined reactive surfaces, hemodynamic flow and optical imaging outines,r and thus are ideal for studies of platelet function and coagulation response. This thesis describes the use of a microfluidic approach to investigate the role of the contact pathway factors XII and XI, platelet-derived polyphosphate, and thiol isomerases in thrombus growth and to evaluate their potential as safer antithrombotic drug targets. The use of low level of corn trypsin inhibitor allowed the study of the contact pathway on collagen/kaolin surfaces with minimally disturbed whole blood sample and we demonstrated the sensitivity of this assay to antithrombotic drugs. On collagen/tissue factor surfaces, we found -

Urokinase Plasminogen Activator: a Potential Thrombolytic Agent for Ischaemic Stroke
Urokinase Plasminogen Activator: A Potential Thrombolytic Agent for Ischaemic Stroke Rais Reskiawan A. Kadir1, Ulvi Bayraktutan1 1Stroke, Division of Clinical Neuroscience, School of Medicine, The University of Nottingham Address for correspondence: Dr. Ulvi Bayraktutan Associate Professor Stroke, Division of Clinical Neuroscience, School of Medicine, The University of Nottingham, Clinical Sciences Building, Hucknall Road, Nottingham NG5 1PB, United Kingdom (UK) Tel: +44-(115) 8231764 Fax: +44-(115) 8231767 E-mail: [email protected] 1 Abstract Stroke continues to be one of the leading causes of mortality and morbidity worldwide. Restoration of cerebral blood flow by recombinant plasminogen activator (rtPA) with or without mechanical thrombectomy is considered the most effective therapy for rescuing brain tissue from ischaemic damage, but this requires advanced facilities and highly skilled professionals, entailing high costs, thus in resource-limited contexts urokinase plasminogen activator (uPA) is commonly used as an alternative. This literature review summarises the existing studies relating to the potential clinical application of uPA in ischaemic stroke patients. In translational studies of ischaemic stroke, uPA has been shown to promote nerve regeneration and reduce infarct volume and neurological deficits. Clinical trials employing uPA as a thrombolytic agent have replicated these favourable outcomes and reported consistent increases in recanalisation, functional improvement, and cerebral haemorrhage rates, similar to those observed with rtPA. Single-chain zymogen pro-urokinase (pro-uPA) and rtPA appear to be complementary and synergistic in their action, suggesting that their co-administration may improve the efficacy of thrombolysis without affecting the overall risk of haemorrhage. Large clinical trials examining the efficacy of uPA or the combination of pro-uPA and rtPA are desperately required to unravel whether either therapeutic approach may be a safe first-line treatment option for patients with ischaemic stroke. -

Biomechanical Thrombosis: the Dark Side of Force and Dawn of Mechano- Medicine
Open access Review Stroke Vasc Neurol: first published as 10.1136/svn-2019-000302 on 15 December 2019. Downloaded from Biomechanical thrombosis: the dark side of force and dawn of mechano- medicine Yunfeng Chen ,1 Lining Arnold Ju 2 To cite: Chen Y, Ju LA. ABSTRACT P2Y12 receptor antagonists (clopidogrel, pras- Biomechanical thrombosis: the Arterial thrombosis is in part contributed by excessive ugrel, ticagrelor), inhibitors of thromboxane dark side of force and dawn platelet aggregation, which can lead to blood clotting and A2 (TxA2) generation (aspirin, triflusal) or of mechano- medicine. Stroke subsequent heart attack and stroke. Platelets are sensitive & Vascular Neurology 2019;0. protease- activated receptor 1 (PAR1) antag- to the haemodynamic environment. Rapid haemodynamcis 1 doi:10.1136/svn-2019-000302 onists (vorapaxar). Increasing the dose of and disturbed blood flow, which occur in vessels with these agents, especially aspirin and clopi- growing thrombi and atherosclerotic plaques or is caused YC and LAJ contributed equally. dogrel, has been employed to dampen the by medical device implantation and intervention, promotes Received 12 November 2019 platelet thrombotic functions. However, this platelet aggregation and thrombus formation. In such 4 Accepted 14 November 2019 situations, conventional antiplatelet drugs often have also increases the risk of excessive bleeding. suboptimal efficacy and a serious side effect of excessive It has long been recognized that arterial bleeding. Investigating the mechanisms of platelet thrombosis -

Ticagrelor – out with the Old, in with the New?
Ticagrelor – out with the old, in with the new? Ticagrelor (Brilinta) is a new oral antiplatelet medicine, which has been available, fully subsidised, Special Authority criteria for ticagrelor with Special Authority, since 1 July, 2013. Ticagrelor, The Special Authority criteria for ticagrelor are the same co-administered with low dose aspirin, is an for both an initial application for use in a patient with an acute coronary syndrome (valid for 12 months) and for alternative to clopidogrel for the prevention of a second application for a subsequent acute coronary atherothrombotic events in patients with acute syndrome. Applications are able to be made by any coronary syndromes. In most cases, ticagrelor will relevant practitioner. Both the initial approval and renewal be initiated in hospital and continued for 12 months are valid for 12 months. after discharge. Prerequisites: 1. Patient has recently* been diagnosed with an Ticagrelor (pronounced tie-kag-re-lore), co-administered with ST elevation or non-ST elevation acute coronary aspirin as dual antiplatelet treatment, is indicated for the syndrome AND prevention of atherothrombotic events in adult patients with 2. Fibrinolytic therapy has not been given in the last acute coronary syndromes, such as ST elevation myocardial 24 hours and is not planned infarction (STEMI) and non-ST elevation myocardial infarction (NSTEMI). It is anticipated that for the majority of patients, * “Recently” in the context of this Special Authority is acute coronary ticagrelor will be initiated in hospital and then continued in syndrome diagnosed within a few days or weeks rather than months. the community after discharge. Patients will qualify under the Special Authority criteria (for the subsidised prescription of ticagrelor) whether they are treated with medical management N.B. -

Exploratory Aspirin Resistance Trial in Healthy Japanese Volunteers (J-ART) Using Platelet Aggregation As a Measure of Thrombogenicity
The Pharmacogenomics Journal (2007) 7, 395–403 & 2007 Nature Publishing Group All rights reserved 1470-269X/07 $30.00 www.nature.com/tpj ORIGINAL ARTICLE Exploratory aspirin resistance trial in healthy Japanese volunteers (J-ART) using platelet aggregation as a measure of thrombogenicity T Fujiwara1, M Ikeda2, K Esumi3, Aspirin prevents the production of thromboxane A2 (TXA2) by irreversibly 4 5 inhibiting platelet cyclooxygenase, exhibiting antiplatelet actions. This agent TD Fujita , M Kono , has been reported to prevent relapse in patients with ischemic heart disease 6 4 H Tokushige , T Hatoyama , or cerebral infarction via this action mechanism. However, there are T Maeda7, T Asai4, T Ogawa6, individual differences in this action, and aspirin is not effective in some T Katsumata4, S Sasaki8, patients, which is referred to as ‘aspirin resistance’. In this study, we analyzed E Suzuki7, M Suzuki5, F Hino9, laboratory aspirin resistance by platelet aggregation in 110 healthy adult 10 10 Japanese males using 24 single-nucleotide polymorphisms (SNPs) of nine TK Fujita , H Zaima , genes involved in platelet aggregation/hemorrhage. Among SNPs involved M Shimada9, T Sugawara8, in platelet aggregation, aspirin was less effective for 924T homozygote of a 10 3 Y Tsuzuki , Y Hashimoto , TXA2 receptor, 924T4C, and 1018C homozygote of a platelet membrane H Hishigaki1, S Horimoto5, glycoprotein GPIba, 1018C4T, suggesting that 924T and 1018C alleles are 2 4 involved in aspirin resistance. N Miyajima , T Yamamoto , The Pharmacogenomics Journal (2007) -

P2X and P2Y Receptors
Tocris Scientific Review Series Tocri-lu-2945 P2X and P2Y Receptors Kenneth A. Jacobson Subtypes and Structures of P2 Receptor Molecular Recognition Section, Laboratory of Bioorganic Families Chemistry, National Institute of Diabetes and Digestive and The P2 receptors for extracellular nucleotides are widely Kidney Diseases, National Institutes of Health, Bethesda, distributed in the body and participate in regulation of nearly Maryland 20892, USA. E-mail: [email protected] every physiological process.1,2 Of particular interest are nucleotide Kenneth Jacobson serves as Chief of the Laboratory of Bioorganic receptors in the immune, inflammatory, cardiovascular, muscular, Chemistry and the Molecular Recognition Section at the National and central and peripheral nervous systems. The ubiquitous Institute of Diabetes and Digestive and Kidney Diseases, National signaling properties of extracellular nucleotides acting at two Institutes of Health in Bethesda, Maryland, USA. Dr. Jacobson is distinct families of P2 receptors – fast P2X ion channels and P2Y a medicinal chemist with interests in the structure and receptors (G-protein-coupled receptors) – are now well pharmacology of G-protein-coupled receptors, in particular recognized. These extracellular nucleotides are produced in receptors for adenosine and for purine and pyrimidine response to tissue stress and cell damage and in the processes nucleotides. of neurotransmitter release and channel formation. Their concentrations can vary dramatically depending on circumstances. Thus, the state of activation of these receptors can be highly dependent on the stress conditions or disease states affecting a given organ. The P2 receptors respond to various extracellular mono- and dinucleotides (Table 1). The P2X receptors are more structurally restrictive than P2Y receptors in agonist selectivity.