Microbial Diversity in Natural Environments: Focusing on Fundamental Questions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Prokaryotes (Domains Bacteria & Archaea)
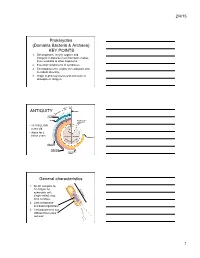
2/4/15 Prokaryotes (Domains Bacteria & Archaea) KEY POINTS 1. Decomposers: recycle organic and inorganic molecules in environment; makes them available to other organisms. 2. Essential components of symbioses. 3. Encompasses the origins of metabolism and metabolic diversity. 4. Origin of photosynthesis and formation of atmospheric Oxygen Ceno- Meso- zoic zoic ANTIQUITY Humans Paleozoic Colonization of land Animals Origin of solar system and Earth • >3.5 BILLION years old. • Alone for 2 1 4 billion years Proterozoic Archaean Prokaryotes Billions of 2 years ago3 Multicellular eukaryotes Single-celled eukaryotes Atmospheric oxygen General characteristics 1. Small: compare to 10-100µm for 0.5-5µm eukaryotic cell; single-celled; may form colonies. 2. Lack membrane- enclosed organelles. 3. Cell wall present, but different from plant cell wall. 1 2/4/15 General characteristics 4. Occur everywhere, most numerous organisms. – More individuals in a handful of soil then there are people that have ever lived. – By far more individuals in our gut than eukaryotic cells that are actually us. General characteristics 5. Metabolic diversity established nutritional modes of eukaryotes. General characteristics 6. Important decomposers and recyclers 2 2/4/15 General characteristics 6. Important decomposers and recyclers • Form the basis of global nutrient cycles. General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • About 1/2 of all human diseases are caused by Bacteria General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • Extremely important in agriculture as well. Pierce’s disease is caused by Xylella fastidiosa, a Gamma Proteobacteria. It causes over $56 million in damage annually in California. That’s with $34 million spent to control it! = $90 million in California alone. -

Group a Streptococcus Produce Pilus-Like Structures Containing Protective Antigens and Lancefield T Antigens
Group A Streptococcus produce pilus-like structures containing protective antigens and Lancefield T antigens Marirosa Mora*†, Giuliano Bensi*†, Sabrina Capo*, Fabiana Falugi*, Chiara Zingaretti*, Andrea G. O. Manetti*, Tiziana Maggi*, Anna Rita Taddei‡, Guido Grandi*, and John L. Telford*§ *Chiron Vaccines, Via Fiorentina 1, 53100 Siena, Italy; and ‡Centro Interdipartimentale di Microscopia Elettronica, University of Tuscia, 01100 Viterbo, Italy Communicated by Rino Rappuoli, Chiron Corporation, Siena, Italy, September 8, 2005 (received for review July 29, 2005) Although pili have long been recognized in Gram-negative patho- extensively characterized and despite five decades of study, there gens as important virulence factors involved in adhesion and is still very little known about the structure and variability of T invasion, very little is known about extended surface organelles in antigens, although a gene of unknown function has been shown Gram-positive pathogens. Here we report that Group A Strepto- to code for the antigen recognized by T6 sera (9). Here we show coccus (GAS), a Gram-positive human-specific pathogen that that four of the 20 T antigens correspond to trypsin-resistant pili causes pharyngitis, impetigo, invasive disease, necrotizing fasciitis, composed of putative adhesion proteins and that recombinant and autoimmune sequelae has long, surface-exposed, pilus-like pilus proteins confer protection against lethal GAS challenge in structures composed of members of a family of extracellular a mouse model of infection and invasive disease. matrix-binding proteins. We describe four variant pili and show that each is recognized by a specific serum of the Lancefield Materials and Methods T-typing system, which has been used for over five decades to Bacterial Strains, Media, and Growth Conditions. -

The First Cells Were Most Likely Very Simple Prokaryotic Forms. Ra- Spirochetes
T HE O RIGIN OF E UKARYOTIC C ELLS The first cells were most likely very simple prokaryotic forms. Ra- spirochetes. Ingestion of prokaryotes that resembled present-day diometric dating indicates that the earth is 4 to 5 billion years old cyanobacteria could have led to the endosymbiotic development of and that prokaryotes may have arisen more than 3.5 billion years chloroplasts in plants. ago. Eukaryotes are thought to have first appeared about 1.5 billion Another hypothesis for the evolution of eukaryotic cells proposes years ago. that the prokaryotic cell membrane invaginated (folded inward) to en- The eukaryotic cell might have evolved when a large anaerobic close copies of its genetic material (figure 1b). This invagination re- (living without oxygen) amoeboid prokaryote ingested small aerobic (liv- sulted in the formation of several double-membrane-bound entities ing with oxygen) bacteria and stabilized them instead of digesting them. (organelles) in a single cell. These entities could then have evolved This idea is known as the endosymbiont hypothesis (figure 1a) and into the eukaryotic mitochondrion, nucleus, and chloroplasts. was first proposed by Lynn Margulis, a biologist at Boston Univer- Although the exact mechanism for the evolution of the eu- sity. (Symbiosis is an intimate association between two organisms karyotic cell will never be known with certainty, the emergence of of different species.) According to this hypothesis, the aerobic bac- the eukaryotic cell led to a dramatic increase in the complexity and teria developed into mitochondria, which are the sites of aerobic diversity of life-forms on the earth. At first, these newly formed eu- respiration and most energy conversion in eukaryotic cells. -

Structural Changes in the Oral Microbiome of the Adolescent
www.nature.com/scientificreports OPEN Structural changes in the oral microbiome of the adolescent patients with moderate or severe dental fuorosis Qian Wang1,2, Xuelan Chen1,4, Huan Hu2, Xiaoyuan Wei3, Xiaofan Wang3, Zehui Peng4, Rui Ma4, Qian Zhao4, Jiangchao Zhao3*, Jianguo Liu1* & Feilong Deng1,2,3* Dental fuorosis is a very prevalent endemic disease. Although oral microbiome has been reported to correlate with diferent oral diseases, there appears to be an absence of research recognizing any relationship between the severity of dental fuorosis and the oral microbiome. To this end, we investigated the changes in oral microbial community structure and identifed bacterial species associated with moderate and severe dental fuorosis. Salivary samples of 42 individuals, assigned into Healthy (N = 9), Mild (N = 14) and Moderate/Severe (M&S, N = 19), were investigated using the V4 region of 16S rRNA gene. The oral microbial community structure based on Bray Curtis and Weighted Unifrac were signifcantly changed in the M&S group compared with both of Healthy and Mild. As the predominant phyla, Firmicutes and Bacteroidetes showed variation in the relative abundance among groups. The Firmicutes/Bacteroidetes (F/B) ratio was signifcantly higher in the M&S group. LEfSe analysis was used to identify diferentially represented taxa at the species level. Several genera such as Streptococcus mitis, Gemella parahaemolysans, Lactococcus lactis, and Fusobacterium nucleatum, were signifcantly more abundant in patients with moderate/severe dental fuorosis, while Prevotella melaninogenica and Schaalia odontolytica were enriched in the Healthy group. In conclusion, our study indicates oral microbiome shift in patients with moderate/severe dental fuorosis. -

Prokaryotes, Protists, Reconstructing the Photosynthesis, Evolution of Living Things
Prokaryotes, Protists, Reconstructing the Photosynthesis, evolution of living things Endosymbiosis • Systematists study evolutionary relationships Ch 28 & 29 •Look for shared derived (=different from ancestor) traits to group organisms • Evidence used: morphology, 26 February 2009 development, and molecular data (especially DNA sequences) ECOL 182R UofA K. E. Bonine 1 2 Why can’t we figure it out perfectly? • More distant history is obscured by more changes trypanosomes • Among oldest lineages of Bacteria and Archaea in particular, lots of “lateral gene transfer.” Makes it difficult to infer relationships from phylogeny of red blood cells single genes. 3 4 Early prokaryote fossil Diversity of Prokaryotes Bacteria & Archaea 5 6 1 What are microbes? Life can be divided into 3 domains • Only a minority make us sick •Robert Koch, Germ Theory of Disease • In ordinary English, might be anything small 3.8bya –bacteria 1.5bya –yeast –protists – viruses •Prokaryotes = bacteria + archaea • In science, classify by evolutionary • Prokaryote was ancestral and only form for relationships… 7 billions of years 8 Eukarya Scheme has been revised before: Haeckel (1894) Whittaker (1959) Woese (1977) Woese (1990) Three kingdoms Five kingdoms Six kingdoms Three domains Eubacteria Bacteria Monera (prokaryotes) Protista Archaebacteria Archaea Protista Protista Fungi Fungi Plantae Eukarya are Prokaryotes monophyletic, Plantae Plantae Animalia Animalia Animalia paraphyleticparaphyletic, polyphyletic? 9 modified from Wikipedia 10 Shared by all 3 domains Unique to -

Oral Microbiota Features in Subjects with Down Syndrome and Periodontal Diseases: a Systematic Review
International Journal of Molecular Sciences Review Oral Microbiota Features in Subjects with Down Syndrome and Periodontal Diseases: A Systematic Review Maria Contaldo 1,* , Alberta Lucchese 1, Antonio Romano 1 , Fedora Della Vella 2 , Dario Di Stasio 1 , Rosario Serpico 1 and Massimo Petruzzi 2 1 Multidisciplinary Department of Medical-Surgical and Dental Specialties, University of Campania Luigi Vanvitelli, Via Luigi de Crecchio, 6, 80138 Naples, Italy; [email protected] (A.L.); [email protected] (A.R.); [email protected] (D.D.S.); [email protected] (R.S.) 2 Interdisciplinary Department of Medicine, University of Bari “Aldo Moro”, 70121 Bari, Italy; [email protected] (F.D.V.); [email protected] (M.P.) * Correspondence: [email protected] or [email protected]; Tel.: +39-3204876058 Abstract: Down syndrome (DS) is a genetic disorder associated with early-onset periodontitis and other periodontal diseases (PDs). The present work aimed to systematically review the scientific literature reporting studies in vivo on oral microbiota features in subjects with DS and related periodontal health and to highlight any correlation and difference with subjects not affected by DS, with and without PDs. PubMed, Web of Science, Scopus and Cochrane were searched for relevant studies in May 2021. The participants were subjects affected by Down syndrome (DS) with and without periodontal diseases; the study compared subjects with periodontal diseases but not affected by DS, and DS without periodontal diseases; the outcomes were the differences in oral microbiota/periodontopathogen bacterial composition among subjects considered; the study Citation: Contaldo, M.; Lucchese, A.; design was a systematic review. -

Prokaryote Vs. Eukaryote Campaign Biology Name: ______Period: ______
Prokaryote vs. Eukaryote Campaign Biology Name: ___________________________________________ Period: ____________ Prokaryotes vs. Eukaryotes: A Campaign for Election as Coolest Cell Type! Purpose: While prokaryotes and eukaryotes share some similar structures and characteristics of life, the two lead some pretty different lives! Your job in this assignment is to create a campaign poster and a campaign platform commercial for a prokaryote or a eukaryote as they campaign to be elected coolest cell type! Directions: Each group will be assigned a campaign platform from those listed below. Use the resources listed (as well as any others you might find) to understand why your platform makes your cell type the coolest. Create a campaign poster (using one slide on a Google Presentation, shared between partners, and submitted by due date via a form) and a short campaign platform commercial (given as a speech in class) using the attached rubric. Prokaryotic campaign platforms: 1. Ability to form endospores http://goo.gl/Y1tUK http://goo.gl/DhVNe 2. Ability to form biofilms with other cells http://goo.gl/umy7Z http://goo.gl/m9d5W 3. Quorum sensing neighboring cells http://goo.gl/DSiJU http://goo.gl/IFFtA 4. Production of bacteriocins http://goo.gl/lW75y http://goo.gl/WzFv1 5. Endosymbiotic theory http://goo.gl/6VIba http://goo.gl/uSYh4 Eukaryotic campaign platforms: 6. Contain organelles that have their own genetic material in addition to that found in the nucleus http://goo.gl/6VIba http://goo.gl/Nqesq 7. Vesicles http://goo.gl/bwF3x http://goo.gl/j3qY3 8. Membrane bound organelles that facilitate transport (endomembrane system) http://goo.gl/j3qY3 http://goo.gl/i4brZ 9. -

Cell Structure and Function in the Bacteria and Archaea
4 Chapter Preview and Key Concepts 4.1 1.1 DiversityThe Beginnings among theof Microbiology Bacteria and Archaea 1.1. •The BacteriaThe are discovery classified of microorganismsinto several Cell Structure wasmajor dependent phyla. on observations made with 2. theThe microscope Archaea are currently classified into two 2. •major phyla.The emergence of experimental 4.2 Cellscience Shapes provided and Arrangements a means to test long held and Function beliefs and resolve controversies 3. Many bacterial cells have a rod, spherical, or 3. MicroInquiryspiral shape and1: Experimentation are organized into and a specific Scientificellular c arrangement. Inquiry in the Bacteria 4.31.2 AnMicroorganisms Overview to Bacterialand Disease and Transmission Archaeal 4.Cell • StructureEarly epidemiology studies suggested how diseases could be spread and 4. Bacterial and archaeal cells are organized at be controlled the cellular and molecular levels. 5. • Resistance to a disease can come and Archaea 4.4 External Cell Structures from exposure to and recovery from a mild 5.form Pili allowof (or cells a very to attach similar) to surfacesdisease or other cells. 1.3 The Classical Golden Age of Microbiology 6. Flagella provide motility. Our planet has always been in the “Age of Bacteria,” ever since the first 6. (1854-1914) 7. A glycocalyx protects against desiccation, fossils—bacteria of course—were entombed in rocks more than 3 billion 7. • The germ theory was based on the attaches cells to surfaces, and helps observations that different microorganisms years ago. On any possible, reasonable criterion, bacteria are—and always pathogens evade the immune system. have been—the dominant forms of life on Earth. -

Cell Theory VOCABULARY Unit 2: Cells Unit 2: > RE B
3.1 Cell Theory KEY CONCEPT Cells are the basic unit of life. VOCABULARY cell theory MAIN IDEAS cytoplasm Early studies led to the development of the cell theory. organelle Prokaryotic cells lack a nucleus and most internal structures of eukaryotic cells. prokaryotic cell eukaryotic cell Connect to Your World You and all other organisms are made of cells. As you saw on the previous page, > a cell’s structure is closely related to its function. Today we know that cells are the smallest unit of living matter that can carry out all processes required for life. But before the 1600s, people had many other ideas about the basis of life. Like many breakthroughs, the discovery of cells was aided by the development of new technology—in this case, the microscope. MAIN IDEA Early studies led to the development of the READING TOOLBOX cell theory. TAKING NOTES As you read, make an outline Almost all cells are too small to see without the aid of a microscope. Although using the headings as topics. glass lenses had been used to magnify images for hundreds of years, the early Summarize details that further lenses were not powerful enough to reveal individual cells. The invention of explain those ideas. the compound microscope in the late 1500s was an early step toward this I. Main Idea discovery. The Dutch eyeglass maker Zacharias Janssen, who was probably A. Supporting idea assisted by his father, Hans, usually gets credit for this invention. 1. Detail A compound microscope contains two or more lenses. Total magnification, the 2. -

D086p093.Pdf
Vol. 86: 93–106, 2009 DISEASES OF AQUATIC ORGANISMS Published September 23 doi: 10.3354/dao02132 Dis Aquat Org OPENPEN ACCESSCCESS Streptococcosis in farmed Litopenaeus vannamei: a new emerging bacterial disease of penaeid shrimp Ken W. Hasson*, Ernesto Matheu Wyld, Yaping Fan, Sonia W. Lingsweiller, Stephanie J. Weaver, Jinling Cheng, Patricia W. Varner Texas Veterinary Medical Diagnostic Lab, 1 Sippel Rd, College Station, Texas 77843, USA ABSTRACT: Presumptive systemic streptococcal infections were detected histologically in farmed Litopenaeus vannamei juveniles submitted from a Latin American country and the bacteria isolated. Characterization work demonstrated that the Gram-positive cocci form chains, grow aerobically and anaerobically, are oxidase- and catalase-negative, non-hemolytic, non-motile, Lancefield Group B positive and PCR positive when amplified with a universal streptococcal primer set. Differing Strep- tococcus identifications were obtained using API 20 Strep and Biolog systems, the former identifying the isolate as S. uberis and the latter as S. parauberis. Injection of specific pathogen-free (SPF) L. van- namei with the bacteria resulted in 100% mortality by 3 d post-injection with successful recovery of the agent from moribund test shrimp hemolymph samples. The recovered isolate was used in per os and waterborne exposure studies of SPF L. vannamei with mortalities ranging from 40 to 100% and 80 to 100%, respectively. Histologic analysis of 5 to 8 moribund shrimp from each exposure method demonstrated that all contained a severe bacteremia characterized by numerous free cocci within the hemolymph and aggregates of vacuolated hemocytes with notable intravacuolar cocci. This unique lesion type was most pronounced within the lymphoid organ and considered pathodiagnostic for this disease. -

Bacteriology
SECTION 1 High Yield Microbiology 1 Bacteriology MORGAN A. PENCE Definitions Obligate/strict anaerobe: an organism that grows only in the absence of oxygen (e.g., Bacteroides fragilis). Spirochete Aerobe: an organism that lives and grows in the presence : spiral-shaped bacterium; neither gram-positive of oxygen. nor gram-negative. Aerotolerant anaerobe: an organism that shows signifi- cantly better growth in the absence of oxygen but may Gram Stain show limited growth in the presence of oxygen (e.g., • Principal stain used in bacteriology. Clostridium tertium, many Actinomyces spp.). • Distinguishes gram-positive bacteria from gram-negative Anaerobe : an organism that can live in the absence of oxy- bacteria. gen. Bacillus/bacilli: rod-shaped bacteria (e.g., gram-negative Method bacilli); not to be confused with the genus Bacillus. • A portion of a specimen or bacterial growth is applied to Coccus/cocci: spherical/round bacteria. a slide and dried. Coryneform: “club-shaped” or resembling Chinese letters; • Specimen is fixed to slide by methanol (preferred) or heat description of a Gram stain morphology consistent with (can distort morphology). Corynebacterium and related genera. • Crystal violet is added to the slide. Diphtheroid: clinical microbiology-speak for coryneform • Iodine is added and forms a complex with crystal violet gram-positive rods (Corynebacterium and related genera). that binds to the thick peptidoglycan layer of gram-posi- Gram-negative: bacteria that do not retain the purple color tive cell walls. of the crystal violet in the Gram stain due to the presence • Acetone-alcohol solution is added, which washes away of a thin peptidoglycan cell wall; gram-negative bacteria the crystal violet–iodine complexes in gram-negative appear pink due to the safranin counter stain. -

Structure of Bacterial and Archaeal Cells
© Jones & Bartlett Learning, LLC. NOT FOR SALE OR DISTRIBUTION 4 CHAPTER PREVIEW 4.1 There Is Tremendous Diversity Structure of Among the Bacteria and Archaea 4.2 Prokaryotes Can Be Distinguished by Their Cell Shape and Arrangements Bacterial and 4.3 An Overview to Bacterial and Archaeal Cell Structure 4.4 External Cell Structures Interact Archaeal Cells with the Environment Investigating the Microbial World 4: Our planet has always been in the “Age of Bacteria,” ever since the The Role of Pili first fossils—bacteria of course—were entombed in rocks more than TexTbook Case 4: An Outbreak of 3 billion years ago. On any possible, reasonable criterion, bacteria Enterobacter cloacae Associated with a Biofilm are—and always have been—the dominant forms of life on Earth. —Paleontologist Stephen J. Gould (1941–2002) 4.5 Most Bacterial and Archaeal Cells Have a Cell Envelope “Double, double toil and trouble; Fire burn, and cauldron bubble” is 4.6 The Cell Cytoplasm Is Packed the refrain repeated several times by the chanting witches in Shakespeare’s with Internal Structures Macbeth (Act IV, Scene 1). This image of a hot, boiling cauldron actu- MiCroinquiry 4: The Prokaryote/ ally describes the environment in which many bacterial, and especially Eukaryote Model archaeal, species happily grow! For example, some species can be iso- lated from hot springs or the hot, acidic mud pits of volcanic vents ( Figure 4.1 ). When the eminent evolutionary biologist and geologist Stephen J. Gould wrote the opening quote of this chapter, he, as well as most micro- biologists at the time, had no idea that embedded in these “bacteria” was another whole domain of organisms.