Permeation Guide
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hydrolysis of Haloacetonitriles: Linear Free Energy Relationship, Kinetics and Products
Wat. Res. Vol. 33, No. 8, pp. 1938±1948, 1999 # 1999 Elsevier Science Ltd. All rights reserved Printed in Great Britain PII: S0043-1354(98)00361-3 0043-1354/99/$ - see front matter HYDROLYSIS OF HALOACETONITRILES: LINEAR FREE ENERGY RELATIONSHIP, KINETICS AND PRODUCTS VICTOR GLEZER*, BATSHEVA HARRIS, NELLY TAL, BERTA IOSEFZON and OVADIA LEV{*M Division of Environmental Sciences, Fredy and Nadine Herrmann School of Applied Science, The Hebrew University of Jerusalem, 91904, Jerusalem, Israel (First received September 1997; accepted in revised form August 1998) AbstractÐThe hydrolysis rates of mono-, di- and trihaloacetonitriles were studied in aqueous buer sol- utions at dierent pH. The stability of haloacetonitriles decreases and the hydrolysis rate increases with increasing pH and number of halogen atoms in the molecule: The monochloroacetonitriles are the most stable and are also less aected by pH-changes, while the trihaloacetonitriles are the least stable and most sensitive to pH changes. The stability of haloacetonitriles also increases by substitution of chlorine atoms with bromine atoms. The hydrolysis rates in dierent buer solutions follow ®rst order kinetics with a minimum hydrolysis rate at intermediate pH. Thus, haloacetonitriles have to be preserved in weakly acid solutions between sampling and analysis. The corresponding haloacetamides are formed during hydrolysis and in basic solutions they can hydrolyze further to give haloacetic acids. Linear free energy relationship can be used for prediction of degradation of haloacetonitriles -

Gasket Chemical Services Guide
Gasket Chemical Services Guide Revision: GSG-100 6490 Rev.(AA) • The information contained herein is general in nature and recommendations are valid only for Victaulic compounds. • Gasket compatibility is dependent upon a number of factors. Suitability for a particular application must be determined by a competent individual familiar with system-specific conditions. • Victaulic offers no warranties, expressed or implied, of a product in any application. Contact your Victaulic sales representative to ensure the best gasket is selected for a particular service. Failure to follow these instructions could cause system failure, resulting in serious personal injury and property damage. Rating Code Key 1 Most Applications 2 Limited Applications 3 Restricted Applications (Nitrile) (EPDM) Grade E (Silicone) GRADE L GRADE T GRADE A GRADE V GRADE O GRADE M (Neoprene) GRADE M2 --- Insufficient Data (White Nitrile) GRADE CHP-2 (Epichlorohydrin) (Fluoroelastomer) (Fluoroelastomer) (Halogenated Butyl) (Hydrogenated Nitrile) Chemical GRADE ST / H Abietic Acid --- --- --- --- --- --- --- --- --- --- Acetaldehyde 2 3 3 3 3 --- --- 2 --- 3 Acetamide 1 1 1 1 2 --- --- 2 --- 3 Acetanilide 1 3 3 3 1 --- --- 2 --- 3 Acetic Acid, 30% 1 2 2 2 1 --- 2 1 2 3 Acetic Acid, 5% 1 2 2 2 1 --- 2 1 1 3 Acetic Acid, Glacial 1 3 3 3 3 --- 3 2 3 3 Acetic Acid, Hot, High Pressure 3 3 3 3 3 --- 3 3 3 3 Acetic Anhydride 2 3 3 3 2 --- 3 3 --- 3 Acetoacetic Acid 1 3 3 3 1 --- --- 2 --- 3 Acetone 1 3 3 3 3 --- 3 3 3 3 Acetone Cyanohydrin 1 3 3 3 1 --- --- 2 --- 3 Acetonitrile 1 3 3 3 1 --- --- --- --- 3 Acetophenetidine 3 2 2 2 3 --- --- --- --- 1 Acetophenone 1 3 3 3 3 --- 3 3 --- 3 Acetotoluidide 3 2 2 2 3 --- --- --- --- 1 Acetyl Acetone 1 3 3 3 3 --- 3 3 --- 3 The data and recommendations presented are based upon the best information available resulting from a combination of Victaulic's field experience, laboratory testing and recommendations supplied by prime producers of basic copolymer materials. -

Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water
PROJECT NO. 4992 Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water Prepared by: Keith A. Maruya Charles S. Wong Southern California Coastal Water Research Project Authority 2020 The Water Research Foundation (WRF) is a nonprofit (501c3) organization which provides a unified source for One Water research and a strong presence in relationships with partner organizations, government and regulatory agencies, and Congress. The foundation conducts research in all areas of drinking water, wastewater, stormwater, and water reuse. The Water Research Foundation’s research portfolio is valued at over $700 million. The Foundation plays an important role in the translation and dissemination of applied research, technology demonstration, and education, through creation of research‐based educational tools and technology exchange opportunities. WRF serves as a leader and model for collaboration across the water industry and its materials are used to inform policymakers and the public on the science, economic value, and environmental benefits of using and recovering resources found in water, as well as the feasibility of implementing new technologies. For more information, contact: The Water Research Foundation Alexandria, VA Office Denver, CO Office 1199 North Fairfax Street, Suite 900 6666 West Quincy Avenue Alexandria, VA 22314‐1445 Denver, Colorado 80235‐3098 Tel: 571.384.2100 Tel: 303.347.6100 www.waterrf.org [email protected] ©Copyright 2020 by The Water Research Foundation. All rights reserved. Permission to copy must be obtained from The Water Research Foundation. WRF ISBN: 978‐1‐60573‐503‐0 WRF Project Number: 4992 This report was prepared by the organization(s) named below as an account of work sponsored by The Water Research Foundation. -

Food and Drugs
21 Part 170 to 199 Revised as of April 1, 2001 Food and Drugs Containing a codification of documents of general applicability and future effect As of April 1, 2001 With Ancillaries Published by Office of the Federal Register National Archives and Records Administration A Special Edition of the Federal Register VerDate 11<MAY>2000 11:38 Apr 16, 2001 Jkt 194064 PO 00000 Frm 00001 Fmt 8091 Sfmt 8091 Y:\SGML\194064F.XXX pfrm02 PsN: 194064F U.S. GOVERNMENT PRINTING OFFICE WASHINGTON : 2001 For sale by the Superintendent of Documents, U.S. Government Printing Office Internet: bookstore.gpo.gov Phone: (202) 512-1800 Tax: (202) 512-2250 Mail Stop: SSOP, Washington, DC 20402–0001 VerDate 11<MAY>2000 11:38 Apr 16, 2001 Jkt 194064 PO 00000 Frm 00002 Fmt 8092 Sfmt 8092 Y:\SGML\194064F.XXX pfrm02 PsN: 194064F Table of Contents Page Explanation ................................................................................................ v Title 21: Chapter I—Food and Drug Administration, Department of Health and Human Services (Continued) ................................................. 3 Finding Aids: Material Approved for Incorporation by Reference ............................ 573 Table of CFR Titles and Chapters ....................................................... 587 Alphabetical List of Agencies Appearing in the CFR ......................... 605 Redesignation Table ............................................................................ 615 List of CFR Sections Affected ............................................................. 617 -

(12) Patent Application Publication (10) Pub. No.: US 2010/0168177 A1 Qin Et Al
US 20100168177A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2010/0168177 A1 Qin et al. (43) Pub. Date: Jul. 1, 2010 (54) STABLE INSECTICIDE COMPOSITIONS (22) Filed: Dec. 22, 2009 Related U.S. Application Data (75) Inventors: Kuide Qin, Westfield, IN (US); (60) Provisional application No. 61/203,689, filed on Dec. Raymond E. Boucher, JR., 26, 2008. Lebanon, IN (US) Publication Classification (51) Int. Cl. Correspondence Address: AOIN 43/40 (2006.01) DOWAGROSCIENCES, LLC AOIP3/00 (2006.01) ONE INDIANA SQUARE, SUITE 2800 AOIP 7/04 (2006.01) INDIANAPOLIS, IN 46204-2079 (US) (52) U.S. Cl. ......................................... 514/336; 514/357 (57) ABSTRACT (73) Assignee: Dow AgroSciences, LLC Insect controlling compositions including an N-Substituted (6-haloalkylpyridin-3-yl)alkylsulfoximine compoundandan (21) Appl. No.: 12/633,987 organic acid or a salt thereof exhibit increased stability. US 2010/01 681 77 A1 Jul. 1, 2010 STABLE INSECTICDE COMPOSITIONS their use in controlling insects and certain other invertebrates, particularly aphids and other Sucking insects. This invention CROSS-REFERENCE TO RELATED also includes new synthetic procedures for preparing the APPLICATIONS compositions and methods of controlling insects using the 0001. The present application claims priority to U.S. Pro compositions. visional Patent Application No. 61/203,689 filed Dec. 26, 0007. This invention concerns compositions useful for the 2008, the content of which is incorporated herein by reference control of insects, especially useful for the control of aphids in its entirety. and other sucking insects. More specifically, the invention FIELD OF THE INVENTION concerns compositions including an organic acid or a salt thereof and a compound of the formula (I) 0002. -

Electronic Supplementary Information For
Electronic Supplementary Material (ESI) for Molecular Systems Design & Engineering. This journal is © The Royal Society of Chemistry 2018 Electronic supplementary information for Understanding structural adaptability: a reactant informatics approach to experiment design Rosalind J. Xu, Jacob H. Olshansky, Philip D. F. Adler, Yongjia Huang, Matthew D. Smith, Matthias Zeller,† Joshua Schrier and Alexander J. Norquist Department of Chemistry, Haverford College, Haverford PA 19041 † Department of Chemistry, Purdue University, West Lafayette IN Author to whom correspondence should be addressed. Haverford College 370 Lancaster Avenue Haverford PA, 19041 USA tel (610) 896 2949 fax (610) 896 4963 email [email protected] http://www.haverford.edu/chem/Norquist/ Figure S1. Three dimensional packing of [C4H14N2]2[VO(SeO3)(HSeO3)]6·5H2O (1). Green octahedra represent [VO6] while purple, red, blue, white and gray spheres represent selenium oxygen, nitrogen, carbon and hydrogen atoms, respectively. Selected hydrogen atoms have been removed for clarity. Figure S2. Three dimensional packing of [C8H26N4][VO(SeO3)(HSeO3)]6·6H2O (3). Green octahedra represent [VO6] while purple, red, blue, white and gray spheres represent selenium oxygen, nitrogen, carbon and hydrogen atoms, respectively. Selected hydrogen atoms have been removed for clarity. Figure S3. (a) One-dimensional chain SBU and (b) and (c) two slice connectivities in [C4H12N2][(VO)3(SeO3)(HSeO3)4]·H2O (4). Green polyhedra represent [VO6] and [VO5] while purple, red, and gray spheres represent selenium oxygen, and hydrogen atoms, respectively. Figure S4. 10-fold cross validation results for the decision trees in this study. Figure S5. Decision tree containing the 75 historical reactions and the 128 reactions from the fractional factorial analysis. -

EPOXY CURING AGENTS Product Overview Guide
EPOXY CURING AGENTS Product Overview Guide NORTH AMERICA EPOXY CURING AGENTS A Robust Toolbox to Meet Diverse Formulation and End Use Requirements With a heritage built on more than 50 continuous years in the industry, Dow is the reliable leader in innovative epoxy product and technology solutions. Dow offers a robust portfolio of epoxy products, including a diverse range of curing agent chemistries and individual grades, to help meet specific formulating and application requirements. To meet increased demand for environmentally responsible formulations, our line also includes low emission and waterborne curing agents. 2 Dow leadership in epoxy technology is demonstrated by innovative technology, high quality products and attention to customer service. In addition to providing one of the broadest epoxy offerings today, our epoxy innovation pipeline – part of Dow’s $1.6 billion annual investment in research & development – ensures that the epoxy products you will require tomorrow are being created by Dow today. Beyond technology development, we are also a source of technical support to help you apply our solutions effectively so customers achieve desired results. Our comprehensive service model – which combines the Dow epoxy sales organization and our R&D and Technical Service Center in Freeport, Texas – gives you access to local know-how and support and world-class technical resources. Our commitment to the epoxy industry includes adhering to the highest standards for safety in the manufacture, storage, transport, use and disposal of our products. This guide presents a comprehensive listing of curing agents available from Dow. For assistance in choosing the right product for your formulation, or to investigate emerging innovation, please contact us. -

Dendrimer-Functionalized Hybrid Materials Based on Silica As Novel Carriers of Bioactive Acids
molecules Article Dendrimer-Functionalized Hybrid Materials Based on Silica as Novel Carriers of Bioactive Acids Mateusz Pawlaczyk * and Grzegorz Schroeder Faculty of Chemistry, Adam Mickiewicz University in Pozna´n,Uniwersytetu Pozna´nskiego 8, 61-614 Pozna´n, Poland; [email protected] * Correspondence: [email protected]; Tel.: +48-61-829-17-97 Received: 9 May 2020; Accepted: 3 June 2020; Published: 8 June 2020 Abstract: One of the major goals in the materials science is the design and development of non-toxic, versatile, and efficient drug delivery systems. The study reported in this paper concerns the syntheses of poly(amidoamine) (PAMAM) dendrimers with tris(2-aminoethyl)amine as an amine core and different terminal amines, and their attachment to silica matrix. The obtained ethylenediamine (EDA), triethylenetetramine (TETA), tris(2-aminoethyl)amine (TREN) and 4,7,10-trioxa-1,13-tridecanediamine (TRI-OXA) dendrimers were introduced to the support surface via an epoxy linker, leading to a loading 1 efficiency in the range of 0.054–0.113 mmol g− , determined using elemental and thermogravimetric analyses. The materials exhibited high adsorption capacities towards the chosen model drugs: folic, salicylic and nicotinic acid. The investigated adsorption processes were found to follow the Freundlich isotherm model, with indication of the drugs’ structure influence on the binding efficiency. Drug-loaded hybrid materials were also described for in vitro drug release in three pH-different paraphysiological media. The highest percentage release was obtained in the tests performed at pH 2.0, ranging between 35.42 and 99.83%. Satisfactory results and the versatility of PAMAM dendrimers may lead to the application of such materials not only as drug carriers dedicated to a wide range of pharmaceutics, but also as analytical tools for pre-concentration and/or the determination of biocompound contamination in samples. -

Tetraethylenepentamine Cas N°: 112-57-2
OECD SIDS TETRAETHYLENEPENTAMINE FOREWORD INTRODUCTION TETRAETHYLENEPENTAMINE CAS N°: 112-57-2 UNEP PUBLICATIONS 1 OECD SIDS TETRAETHYLENEPENTAMINE SIDS Initial Assessment Report For 13th SIAM (Bern, Switzerland November 6-9, 2001) Chemical Name: Tetraethylenepentamine CAS No.: 112-57-2 Sponsor Country: USA SIDS Contact: Oscar Hernandez United States Environmental Protection Agency (7403M) ICC Building, room 6220A 1200 Pennsylvania Avenue, NW Washington, DC 20460 History: Tetraethylenepentamine was volunteered for the U.S. HPV program and subsequently the ICCA program by the Ethyleneamines Product Stewardship Discussion Group in the U.S. Use of data from the analog triethylenetetramine is proposed to reduce testing needs. The panel/consortia (Dow, UCC and Azko-Nobel) searched company files and publicly available databases to obtain data on TEPA. The “Environmental Risk Assessment of Complexing Agents” submitted by Germany was also included in this evaluation. Testing: No testing (x) Testing ( ) Comments: Deadline for Circulation: September 14, 2001 Date of Circulation: 2 UNEP Publications OECD SIDS TETRAETHYLENEPENTAMINE SIDS INITIAL ASSESSMENT PROFILE CAS No. 112-57-2 Chemical Name 3,6,9-triazaundecamethylenediamine; tetraethylenepentamine (TEPA) Structural Formula NH2CH2CH2NHCH2CH2NHCH2CH2NHCH2CH2NH2 RECOMMENDATIONS The chemical is currently of low priority for further work. SUMMARY CONCLUSIONS OF THE SIAR Use of Analog TETA to suppleme nt TEPA data Tetraethylenepentamine (TEPA) is similar toxicologically to triethylenetetramine (TETA) based on its structure and chelation properties. Therefore, data obtained using TETA have been used to address the endpoints for reproductive and developmental toxicity. Human Health Tetraethylenepentamine (TEPA) has a low acute toxicity when administered orally to rats (LD50 = 3250 mg/kg). In an acute inhalation toxicity study with saturated vapor and whole body exposure, the LC50 was calculated to be >9.9 ppm (highest dose tested). -
![METHOD FINDER (Key on Page MF!34]](https://docslib.b-cdn.net/cover/8210/method-finder-key-on-page-mf-34-2618210.webp)
METHOD FINDER (Key on Page MF!34]
METHOD FINDER (Key on page MF!34] Meth Flow Rate Vol. (L) Extraction Analytical Chemical No. Method Name (L/min) Technique Sampler min max Acenaphthene 5506 PO LYN UC LEAR ARO MATIC 2.0 200 - 1000 aceto nitrile HPLC!FL/UV PTFE & XAD!2 HYDROCARBO NS by HPLC Acenaphthene 5515 PO LYN UC LEAR ARO MATIC 2.0 200 - 1000 varies GC!FID PTFE & XAD!2 HYDROCARBONS Acenaphthylene 5506 PO LYN UC LEAR ARO MATIC 2.0 200 - 1000 aceto nitrile HPLC!FL/UV PTFE & XAD!2 HYDROCARBO NS by HPLC Acenaphthylene 5515 PO LYN UC LEAR ARO MATIC 2.0 200 ! 1000 varies GC!FID PTFE & XAD!2 HYDROCARBONS Acetaldehyde 2018 ALIPHATIC ALDEHYDES 0.1 - 1.5 1 - 15 ACN HPLC/UV SG-DNPH Acetaldehyde 2538 ACETALDEHYDE 0.01 - 0.05 1 ! 12 tol GC!FID XAD!2/HMP Acetaldehyde 2539 ALDEHYDES, SCREENING 0.01 - 0.05 5 - 5 tol GC!FID/MS XAD-2/HMP Acetaldehyde 3507 ACETALDEHYDE 0.1 - 0.5 6 - 60 reag HPLC!UV BuB Ac etic acid 1603 AC ETIC AC ID 0.01 - 0.1 20 - 300 formic a GC!FID CCT Acetic anhydride 3506 ACETIC ANHYDRIDE 0.2 - 1.0 25 - 100 reag VIS BuB Ac eto in 2558 AC ETO IN 0.01 - 0.2 1 - 10 Acet-Meth GC-FID A-CMS Acetone 1300 KETONES I 0.01 - 0.2 0.5 - 3 CS2 GC!FID CCT Acetone 2549 VOLATILE ORGANIC CPDS 0.01 - 0.05 1 - 6 thermal TD/GC!MS TD (Screening) Acetone 3800 ORGANIC AND INORGANIC GASES 0.1 - 2.0 Instr. -

Chemicals Subject to TSCA Section 12(B) Export Notification Requirements (January 16, 2020)
Chemicals Subject to TSCA Section 12(b) Export Notification Requirements (January 16, 2020) All of the chemical substances appearing on this list are subject to the Toxic Substances Control Act (TSCA) section 12(b) export notification requirements delineated at 40 CFR part 707, subpart D. The chemicals in the following tables are listed under three (3) sections: Substances to be reported by Notification Name; Substances to be reported by Mixture and Notification Name; and Category Tables. TSCA Regulatory Actions Triggering Section 12(b) Export Notification TSCA section 12(b) requires any person who exports or intends to export a chemical substance or mixture to notify the Environmental Protection Agency (EPA) of such exportation if any of the following actions have been taken under TSCA with respect to that chemical substance or mixture: (1) data are required under section 4 or 5(b), (2) an order has been issued under section 5, (3) a rule has been proposed or promulgated under section 5 or 6, or (4) an action is pending, or relief has been granted under section 5 or 7. Other Section 12(b) Export Notification Considerations The following additional provisions are included in the Agency's regulations implementing section 12(b) of TSCA (i.e. 40 CFR part 707, subpart D): (a) No notice of export will be required for articles, except PCB articles, unless the Agency so requires in the context of individual section 5, 6, or 7 actions. (b) Any person who exports or intends to export polychlorinated biphenyls (PCBs) or PCB articles, for any purpose other than disposal, shall notify EPA of such intent or exportation under section 12(b). -
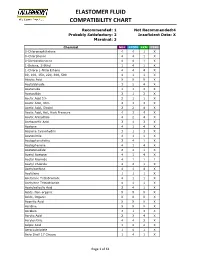
Elastomer Fluid Compatibility Chart
ELASTOMER FLUID COMPATIBILITY CHART Recommended: 1 Not Recommended:4 Probably Satisfactory: 2 Insuficient Data: X Marginal: 3 Chemical NBR EPDM FKM PTFE 0-Chloronaphthalene 4 4 1 X 0-Chlorphenol 4 4 1 X 0-Dichlorobenzene 4 4 1 X 1-Butene, 2-Ethyl 1 4 1 X 1-Chloro 1-Nitro Ethane 4 4 4 X 90, 100, 150, 220, 300, 500 4 1 1 X Abietic Acid X X X X Acetaldehyde 3 2 4 X Acetamide 1 1 3 X Acetanilide 3 1 3 X Acetic Acid 5% 2 1 1 X Acetic Acid, 30% 2 1 3 X Acetic Acid, Glacial 2 2 4 X Acetic Acid, Hot, High Pressure 4 3 4 X Acetic Anhydride 4 2 4 X Acetoacetic Acid 3 1 3 X Acetone 4 1 4 X Acetone Cyanohydrin 3 1 3 X Acetonitrile 3 1 1 X Acetophenetidine 2 4 1 X Acetophenone 4 1 4 X Acetotoluidide 2 4 1 X Acetyl Acetone 4 1 4 X Acetyl Bromide 4 1 1 1 Acetyl Chloride 4 4 1 X Acetylacetone 4 1 4 X Acetylene 1 1 1 X Acetylene Tetrabromide 4 1 1 X Acetylene Tetrachloride 4 1 1 X Acetylsalicylic Acid 2 4 1 X Acids, Non-organic X X X X Acids, Organic X X X X Aconitic Acid X X X X Acridine X X X X Acrolein 3 1 3 X Acrylic Acid 2 3 4 X Acrylonitrile 4 4 3 X Adipic Acid 1 2 2 X Aero Lubriplate 1 4 1 X Aero Shell 17 Grease 1 4 1 X Page 1 of 61 ELASTOMER FLUID COMPATIBILITY CHART Recommended: 1 Not Recommended:4 Probably Satisfactory: 2 Insuficient Data: X Marginal: 3 Chemical NBR EPDM FKM PTFE Aero Shell 1AC Grease 1 4 1 X Aero Shell 750 2 4 1 X Aero Shell 7A Grease 1 4 1 X Aerosafe 2300 4 1 4 X Aerosafe 2300W 4 1 4 X Aerozene 50 (50% Hydrazine 50% UDMH) 3 1 4 X Air, 1 1 1 X Air, 200 - 300° F 3 2 1 X Air, 300 - 400° F 4 4 1 X Air, 400 - 500° F 4 4 3 X Air,