Axonal Targeting of the 5-HT1B Serotonin Receptor Relies on Structure-Specific Constitutive Activation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Mapping the Physiological and Molecular Markers of Stress and SSRI Antidepressant Treatment in S100a10 Corticostriatal Neurons
Molecular Psychiatry (2020) 25:1112–1129 https://doi.org/10.1038/s41380-019-0473-6 ARTICLE Mapping the physiological and molecular markers of stress and SSRI antidepressant treatment in S100a10 corticostriatal neurons 1 2 1 3,4 1 Derya Sargin ● Revathy U. Chottekalapanda ● Kristina E. Perit ● Victoria Yao ● Duong Chu ● 1 2 1 3,4,5 6 2 Daniel W. Sparks ● Salina Kalik ● Saige K. Power ● Olga G. Troyanskaya ● Eric F. Schmidt ● Paul Greengard ● Evelyn K. Lambe 1,7,8 Received: 30 October 2018 / Revised: 8 April 2019 / Accepted: 17 May 2019 / Published online: 20 August 2019 © The Author(s) 2019. This article is published with open access Abstract In mood disorders, psychomotor and sensory abnormalities are prevalent, disabling, and intertwined with emotional and cognitive symptoms. Corticostriatal neurons in motor and somatosensory cortex are implicated in these symptoms, yet mechanisms of their vulnerability are unknown. Here, we demonstrate that S100a10 corticostriatal neurons exhibit distinct serotonin responses and have increased excitability, compared with S100a10-negative neurons. We reveal that prolonged social isolation disrupts the specific serotonin response which gets restored by chronic antidepressant treatment. We identify fi 1234567890();,: 1234567890();,: cell-type-speci c transcriptional signatures in S100a10 neurons that contribute to serotonin responses and strongly associate with psychomotor and somatosensory function. Our studies provide a strong framework to understand the pathogenesis and create new avenues for the treatment of mood disorders. Introduction Our understanding of mood and anxiety disorders is limited These authors contributed equally: Derya Sargin, Revathy U. Chottekalapanda due to the complexity of the circuitry involved and the diverse symptoms manifested in these diseases. -

Strategies to Increase ß-Cell Mass Expansion
This electronic thesis or dissertation has been downloaded from the King’s Research Portal at https://kclpure.kcl.ac.uk/portal/ Strategies to increase -cell mass expansion Drynda, Robert Lech Awarding institution: King's College London The copyright of this thesis rests with the author and no quotation from it or information derived from it may be published without proper acknowledgement. END USER LICENCE AGREEMENT Unless another licence is stated on the immediately following page this work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International licence. https://creativecommons.org/licenses/by-nc-nd/4.0/ You are free to copy, distribute and transmit the work Under the following conditions: Attribution: You must attribute the work in the manner specified by the author (but not in any way that suggests that they endorse you or your use of the work). Non Commercial: You may not use this work for commercial purposes. No Derivative Works - You may not alter, transform, or build upon this work. Any of these conditions can be waived if you receive permission from the author. Your fair dealings and other rights are in no way affected by the above. Take down policy If you believe that this document breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 02. Oct. 2021 Strategies to increase β-cell mass expansion A thesis submitted by Robert Drynda For the degree of Doctor of Philosophy from King’s College London Diabetes Research Group Division of Diabetes & Nutritional Sciences Faculty of Life Sciences & Medicine King’s College London 2017 Table of contents Table of contents ................................................................................................. -

The S100A10 Subunit of the Annexin A2 Heterotetramer Facilitates L2-Mediated Human Papillomavirus Infection
The S100A10 Subunit of the Annexin A2 Heterotetramer Facilitates L2-Mediated Human Papillomavirus Infection Andrew W. Woodham1, Diane M. Da Silva2,3, Joseph G. Skeate1, Adam B. Raff1, Mark R. Ambroso4, Heike E. Brand3, J. Mario Isas4, Ralf Langen4, W. Martin Kast1,2,3* 1 Departments of Molecular Microbiology & Immunology, University of Southern California, Los Angeles, California, United States of America, 2 Department of Obstetrics & Gynecology, University of Southern California, Los Angeles, California, United States of America, 3 Norris Comprehensive Cancer Center, University of Southern California, Los Angeles, California, United States of America, 4 Department of Biochemistry & Molecular Biology, University of Southern California, Los Angeles, California, United States of America Abstract Mucosotropic, high-risk human papillomaviruses (HPV) are sexually transmitted viruses that are causally associated with the development of cervical cancer. The most common high-risk genotype, HPV16, is an obligatory intracellular virus that must gain entry into host epithelial cells and deliver its double stranded DNA to the nucleus. HPV capsid proteins play a vital role in these steps. Despite the critical nature of these capsid protein-host cell interactions, the precise cellular components necessary for HPV16 infection of epithelial cells remains unknown. Several neutralizing epitopes have been identified for the HPV16 L2 minor capsid protein that can inhibit infection after initial attachment of the virus to the cell surface, which suggests an L2-specific secondary receptor or cofactor is required for infection, but so far no specific L2-receptor has been identified. Here, we demonstrate that the annexin A2 heterotetramer (A2t) contributes to HPV16 infection and co- immunoprecipitates with HPV16 particles on the surface of epithelial cells in an L2-dependent manner. -

Transcriptomic Profiling of Pancreatic Alpha, Beta and Delta Cell Populations Identifies Delta Cells As a Principal Target for Ghrelin in Mouse Islets
Diabetologia (2016) 59:2156–2165 DOI 10.1007/s00125-016-4033-1 ARTICLE Transcriptomic profiling of pancreatic alpha, beta and delta cell populations identifies delta cells as a principal target for ghrelin in mouse islets Alice E. Adriaenssens1 & Berit Svendsen2,3 & Brian Y. H. Lam1 & Giles S. H. Yeo1 & Jens J. Holst2,3 & Frank Reimann1 & Fiona M. Gribble 1 Received: 15 March 2016 /Accepted: 1 June 2016 /Published online: 7 July 2016 # The Author(s) 2016. This article is published with open access at Springerlink.com Abstract using islets with delta cell restricted expression of the calcium Aims/hypothesis Intra-islet and gut–islet crosstalk are critical reporter GCaMP3, and in perfused mouse pancreases. in orchestrating basal and postprandial metabolism. The aim Results A database was constructed of all genes expressed in of this study was to identify regulatory proteins and receptors alpha, beta and delta cells. The gene encoding the ghrelin underlying somatostatin secretion though the use of receptor, Ghsr, was highlighted as being highly expressed transcriptomic comparison of purified murine alpha, beta and enriched in delta cells. Activation of the ghrelin receptor and delta cells. raised cytosolic calcium levels in primary pancreatic delta Methods Sst-Cre mice crossed with fluorescent reporters were cells and enhanced somatostatin secretion in perfused used to identify delta cells, while Glu-Venus (with Venus re- pancreases, correlating with a decrease in insulin and gluca- ported under the control of the Glu [also known as Gcg]pro- gon release. The inhibition of insulin secretion by ghrelin was moter) mice were used to identify alpha and beta cells. -

Rage (Receptor for Advanced Glycation End Products) in Melanoma
RAGE (RECEPTOR FOR ADVANCED GLYCATION END PRODUCTS) IN MELANOMA PROGRESSION A Dissertation Submitted to the Graduate Faculty of the North Dakota State University of Agriculture and Applied Science By Varsha Meghnani In Partial Fulfillment for the Degree of DOCTOR OF PHILOSOPHY Major Department: Pharmaceutical Sciences May 2014 Fargo, North Dakota North Dakota State University Graduate School Title RAGE (RECEPTOR FOR ADVANCED GLYCATION END PRODUCTS) IN MELANOMA PROGRESSION By VARSHA MEGHNANI The Supervisory Committee certifies that this disquisition complies with North Dakota State University’s regulations and meets the accepted standards for the degree of DOCTOR OF PHILOSOPHY SUPERVISORY COMMITTEE: ESTELLE LECLERC Chair BIN GUO STEPHEN O’ROURKE JANE SCHUH Approved: 5/22/2014 JAGDISH SINGH Date Department Chair ABSTRACT The Receptor for Advanced Glycation End Products (RAGE) and its ligands are expressed in multiple cancer types and are implicated in cancer progression. Our lab has previously reported higher transcript levels of RAGE and S100B in advanced staged melanoma patients. The contribution of RAGE in the pathophysiology of melanoma has not been well studied. Based on previous reports, we hypothesized that RAGE, when over-expressed in melanoma cells, promotes melanoma progression. To study the pathogenic role of RAGE in melanoma, a primary melanoma cell line, WM115, was selected and stably transfected with full length RAGE (FL_RAGE) to generate a model cell line over-expressing RAGE (WM115_RAGE). WM266, a sister cell line of WM115, originated from a metastatic tumor of the same patient was used as a metastatic control cell line in the study. After assessing the expression levels of RAGE in the transfected cells, the influence of RAGE on their cellular properties was examined. -

S100A10 Antibody
Product Datasheet S100A10 Antibody Catalog No: #49352 Package Size: #49352-1 50ul #49352-2 100ul Orders: [email protected] Support: [email protected] Description Product Name S100A10 Antibody Host Species Rabbit Clonality Monoclonal Clone No. JF0987 Purification ProA affinity purified Applications WB, IHC Species Reactivity Hu, Ms Immunogen Description recombinant protein Other Names 42C antibody AA409961 antibody AL024248 antibody Annexin II ligand antibody Annexin II ligand, calpactin I, light polypeptide antibody Annexin II tetramer (AIIt) p11 subunit antibody Annexin II, light chain antibody ANX2L antibody ANX2LG antibody Ca[1] antibody CAL12 antibody CAL1L antibody Calpactin I light chain antibody Calpactin I, p11 subunit antibody Calpactin-1 light chain antibody Cellular ligand of annexin II antibody CLP11 antibody GP11 antibody MGC111133 antibody Nerve growth factor-induced protein 42C antibody OTTHUMP00000015269 antibody OTTHUMP00000015270 antibody p10 antibody p10 protein antibody p11 antibody Protein S100 A10 antibody Protein S100-A10 antibody S100 calcium binding protein A10 (annexin II ligand, calpactin I, light polypeptide (p11)) antibody S100 calcium binding protein A10 (calpactin) antibody S100 calcium binding protein A10 antibody S100 calcium-binding protein A10 antibody S100a10 antibody S10AA_HUMAN antibody Accession No. Swiss-Prot#:P60903 Calculated MW 11 kDa Formulation 1*TBS (pH7.4), 1%BSA, 40%Glycerol. Preservative: 0.05% Sodium Azide. Storage Store at -20°C Application Details WB: 1:1,000-1:2,000 IHC: 1:50-1:200 Images Western blot analysis of S100A10 on human lung lysates using anti-S100A10 antibody at 1/1,000 dilution. Address: 8400 Baltimore Ave. Suite 302 College Park MD 20740 USA http://www.sabbiotech.com 1 Immunohistochemical analysis of paraffin-embedded human colon cancer tissue using anti-S100A10 antibody. -

Guthrie Cdna Resource Center
cDNA Resource Center cDNA Resource Center Catalog cDNA Resource Center Missouri University of Science and Technology 400 W 11th Rolla, MO 65409 TEL: (573) 341-7610 FAX: (573) 341-7609 EMAIL: [email protected] www.cdna.org September, 2008 1 cDNA Resource Center Visit our web site for product updates 2 cDNA Resource Center The cDNA Resource Center The cDNA Resource Center is a service provided by the faculty of the Department of Biological Sciences of Missouri University of Science and Technology. The purpose of the cDNA Resource Center is to further scientific investigation by providing cDNA clones of human proteins involved in signal transduction processes. This is achieved by providing high quality clones for important signaling proteins in a timely manner. By high quality, we mean that the clones are • Sequence verified • Propagated in a versatile vector useful in bacterial and mammalian systems • Free of extraneous 3' and 5' untranslated regions • Expression verified (in most cases) by coupled in vitro transcription/translation assays • Available in wild-type, epitope-tagged and common mutant forms (e.g., constitutively- active or dominant negative) By timely, we mean that the clones are • Usually shipped within a day from when you place your order. Clones can be ordered from our web pages, by FAX or by phone. Within the United States, clones are shipped by overnight courier (FedEx); international orders are shipped International Priority (FedEx). The clones are supplied for research purposes only. Details on use of the material are included on the Material Transfer Agreement (page 3). Clones are distributed by agreement in Invitrogen's pcDNA3.1+ vector. -
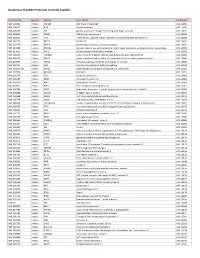
Quantigene Flowrna Probe Sets Currently Available
QuantiGene FlowRNA Probe Sets Currently Available Accession No. Species Symbol Gene Name Catalog No. NM_003452 Human ZNF189 zinc finger protein 189 VA1-10009 NM_000057 Human BLM Bloom syndrome VA1-10010 NM_005269 Human GLI glioma-associated oncogene homolog (zinc finger protein) VA1-10011 NM_002614 Human PDZK1 PDZ domain containing 1 VA1-10015 NM_003225 Human TFF1 Trefoil factor 1 (breast cancer, estrogen-inducible sequence expressed in) VA1-10016 NM_002276 Human KRT19 keratin 19 VA1-10022 NM_002659 Human PLAUR plasminogen activator, urokinase receptor VA1-10025 NM_017669 Human ERCC6L excision repair cross-complementing rodent repair deficiency, complementation group 6-like VA1-10029 NM_017699 Human SIDT1 SID1 transmembrane family, member 1 VA1-10032 NM_000077 Human CDKN2A cyclin-dependent kinase inhibitor 2A (melanoma, p16, inhibits CDK4) VA1-10040 NM_003150 Human STAT3 signal transducer and activator of transcripton 3 (acute-phase response factor) VA1-10046 NM_004707 Human ATG12 ATG12 autophagy related 12 homolog (S. cerevisiae) VA1-10047 NM_000737 Human CGB chorionic gonadotropin, beta polypeptide VA1-10048 NM_001017420 Human ESCO2 establishment of cohesion 1 homolog 2 (S. cerevisiae) VA1-10050 NM_197978 Human HEMGN hemogen VA1-10051 NM_001738 Human CA1 Carbonic anhydrase I VA1-10052 NM_000184 Human HBG2 Hemoglobin, gamma G VA1-10053 NM_005330 Human HBE1 Hemoglobin, epsilon 1 VA1-10054 NR_003367 Human PVT1 Pvt1 oncogene homolog (mouse) VA1-10061 NM_000454 Human SOD1 Superoxide dismutase 1, soluble (amyotrophic lateral sclerosis 1 (adult)) -

Role of P11 in Cellular and Behavioral Effects of 5-HT4 Receptor Stimulation
The Journal of Neuroscience, February 11, 2009 • 29(6):1937–1946 • 1937 Behavioral/Systems/Cognitive Role of p11 in Cellular and Behavioral Effects of 5-HT4 Receptor Stimulation Jennifer L. Warner-Schmidt,1 Marc Flajolet,1 Abigail Maller,1 Emily Y. Chen,1 Hongshi Qi,2 Per Svenningsson,1,2 and Paul Greengard1 1Laboratory of Molecular and Cellular Neuroscience, The Rockefeller University, New York, New York 10065, and 2Center for Molecular Medicine, Department of Physiology and Pharmacology, Karolinska Institute, SE-171 77 Stockholm, Sweden p11 (S100A10), a member of a large family of S100 proteins, interacts with serotonin receptor 1B (5-HTR1B), modulates 5-HT1B receptor signal transduction, and is required for antidepressant responses to activation of this receptor. In the current study, we investigated the specificity of the interaction between 5-HTR1B and p11 by screening brain-expressed S100 proteins against serotonin and noradrenergic receptors. The data indicate that p11 is unique among its family members for its interactions with defined serotonin receptors. We identify a novel p11-interacting receptor (5-HTR4) and characterize the interaction between p11 and 5-HTR4, demonstrating that (1) p11 and 5-HTR4 mRNA and protein are coexpressed in brain regions that are relevant for major depression, (2) p11 increases 5-HTR4 surface expression and facilitates 5-HTR4 signaling, and (3) p11 is required for the behavioral antidepressant responses to 5-HTR4 stimulation in vivo. The essential role played by p11 in modulating signaling through 5-HT4 as well as 5-HT1B receptors supports the concept that this protein may be a key determinant of vulnerability to depression. -

Biased Signaling of G Protein Coupled Receptors (Gpcrs): Molecular Determinants of GPCR/Transducer Selectivity and Therapeutic Potential
Pharmacology & Therapeutics 200 (2019) 148–178 Contents lists available at ScienceDirect Pharmacology & Therapeutics journal homepage: www.elsevier.com/locate/pharmthera Biased signaling of G protein coupled receptors (GPCRs): Molecular determinants of GPCR/transducer selectivity and therapeutic potential Mohammad Seyedabadi a,b, Mohammad Hossein Ghahremani c, Paul R. Albert d,⁎ a Department of Pharmacology, School of Medicine, Bushehr University of Medical Sciences, Iran b Education Development Center, Bushehr University of Medical Sciences, Iran c Department of Toxicology–Pharmacology, School of Pharmacy, Tehran University of Medical Sciences, Iran d Ottawa Hospital Research Institute, Neuroscience, University of Ottawa, Canada article info abstract Available online 8 May 2019 G protein coupled receptors (GPCRs) convey signals across membranes via interaction with G proteins. Origi- nally, an individual GPCR was thought to signal through one G protein family, comprising cognate G proteins Keywords: that mediate canonical receptor signaling. However, several deviations from canonical signaling pathways for GPCR GPCRs have been described. It is now clear that GPCRs can engage with multiple G proteins and the line between Gprotein cognate and non-cognate signaling is increasingly blurred. Furthermore, GPCRs couple to non-G protein trans- β-arrestin ducers, including β-arrestins or other scaffold proteins, to initiate additional signaling cascades. Selectivity Biased Signaling Receptor/transducer selectivity is dictated by agonist-induced receptor conformations as well as by collateral fac- Therapeutic Potential tors. In particular, ligands stabilize distinct receptor conformations to preferentially activate certain pathways, designated ‘biased signaling’. In this regard, receptor sequence alignment and mutagenesis have helped to iden- tify key receptor domains for receptor/transducer specificity. -

Unique S100 Target Protein Interactions
Gen. Physiol. Biophys. (2009), 28, Focus Issue, F39–F46 F39 Review Unique S100 target protein interactions Atoosa Rezvanpour and Gary S. Shaw Department of Biochemistry, The University of Western Ontario, London, Ontario, Canada N6A 5C1 Abstract. Three-dimensional structures of S100B, S100A1, S100A6 and S100A11 have shown that calcium binding to these proteins results in a conformational change allowing them to interact with many biological targets. The structures of some S100 proteins in the presence of peptide targets from Ndr kinase, p53, CapZ, annexins A1 and A2 and the Siah-1 Interacting Protein indicate there are at least three modes of recognition that utilize two distinct surfaces in the S100 proteins. These surfaces have been hypothesized to simultaneously accommodate multiple binding partners. This review focuses on potential multiprotein complexes involving calcium-insensitive S100A10, annexin A2 and several other proteins including AHNAK, dysferlin, NS3, TASK-1 and TRPV5/6. Keywords: Annexin — Multi-protein complex — Calcium-signaling — Membrane repair — Three- dimensional structure Introduction binding to the second EF-hand, comprised of helices III and IV. Three-dimensional structures of several S100 proteins The S100 proteins are a group of proteins comprising at least in the calcium-free (apo) and calcium-bound states show 25 members in humans including S100B, S100A1, S100A6, that the major structural change involves the movement of S100A10 and S100A11 (Donato 2001; Heizmann et al. 2002). helix III to expose previously buried residues which create The proteins are dimeric having two “EF-hand” calcium- a hydrophobic surface. In one S100 protein, S100A10, substi- binding motifs in each subunit. In vivo experiments have tutions in both its calcium-binding sites have left this protein shown that both homo- and heterodimeric S100 complexes with the inability to coordinate calcium. -

Deletion of Annexin 2 Light Chain P11 in Nociceptors Causes Deficits in Somatosensory Coding and Pain Behavior
The Journal of Neuroscience, October 11, 2006 • 26(41):10499–10507 • 10499 Cellular/Molecular Deletion of Annexin 2 Light Chain p11 in Nociceptors Causes Deficits in Somatosensory Coding and Pain Behavior Thomas Foulkes,1 Mohammed A. Nassar,1 Tim Lane,1 Elizabeth A. Matthews,2 Mark D. Baker,1 Volker Gerke,3 Kenji Okuse,4 Anthony H. Dickenson,2 and John N. Wood1 1Molecular Nociception Group, Department of Biology, and 2Department of Pharmacology, University College London, London WC1E 6BT, United Kingdom, 3Institute of Medical Biochemistry, Center for Molecular Biology of Inflammation, University of Muenster, 48149 Muenster, Germany, and 4Division of Cell and Molecular Biology, Imperial College, London SW7 2AZ, United Kingdom The S100 family protein p11 (S100A10, annexin 2 light chain) is involved in the trafficking of the voltage-gated sodium channel NaV1.8, TWIK-related acid-sensitive K ϩ channel (TASK-1), the ligand-gated ion channels acid-sensing ion channel 1a (ASIC1a) and transient receptor potential vanilloid 5/6 (TRPV5/V6), as well as 5-hydroxytryptamine receptor 1B (5-HT1B ), a G-protein-coupled receptor. To evaluate the role of p11 in peripheral pain pathways, we generated a loxP-flanked (floxed) p11 mouse and used the Cre-loxP recombinase system to delete p11 exclusively from nociceptive primary sensory neurons in mice. p11-null neurons showed deficits in the expression of NaV1.8, but not of annexin 2. Damage-sensing primary neurons from these animals show a reduced tetrodotoxin-resistant sodium current density, consistent with a loss of membrane-associated NaV1.8. Noxious coding in wide-dynamic-range neurons in the dorsal horn was markedly compromised.