Exploitation of Burbot Lota Lota in the Wind River
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

The Ecology and Management of the European Grayling Thymallus Thymallus (Linnaeus)
The ecology and management of the European grayling Thymallus thymallus (Linnaeus). Interim report Item Type monograph Authors Ingram, A.; Ibbotson, A.; Gallagher, M. Publisher Institute of Freshwater Ecology Download date 03/10/2021 22:03:11 Link to Item http://hdl.handle.net/1834/24874 The Ecology and Management of the European Grayling Thymallus thymallus (Linnaeus) Interim Report Ingram A Ibbotson A Gallagher M The Ecology and Management of the European Grayling Thymallus thymallus (Linnaeus) Interim Report Ingram A Ibbotson A Gallagher M INTELLECTUAL PROPERTY RIGHTS CONFIDENTIALITY STATEMENT 'In accordance with our normal practice, this report is for the use only of the party to whom it is addressed, and no responsibility is accepted to any third party for the whole or any part of its contents. Neither the whole nor any part of this report or any reference thereto may be included in any published document, circular or statement, nor published or referred to in any way without our written approval of the form and context in which it may appear.' 11 CHAPTER 1 1 Overall aim A collaborative research and development project between the Institute of Freshwater Ecology and the Environment Agency in the UK, to review the ecology, status and management of grayling in order to provide recommendations for future management of grayling fisheries in England and Wales. 2 Objectives • To review grayling ecology, status and management practice in concentrating on England and Wales but including published literature from Europe and North America, where appropriate. • To recommend best management practices on the basis of objective 1 and produce a guidance leaflet for internal and external circulation which promotes the key issues. -

Burbot Management Plan
BURBOT MANAGEMENT PLAN DEPARTMENT OF INLAND FISHERIES AND WILDLIFE DIVISION OF FISHERIES AND HATCHERIES PREPARED BY SCOTT A. ROY ASSISTANT REGIONAL FISHERIES BIOLOGIST REGION E MARCH 2001 BURBOT LIFE HISTORY The burbot, Lota lota (Linnaeus), is a unique member of the cod family. It is the only species in the family, which spends its entire life in fresh water. However, the burbot is similar to its marine relatives in that its distribution is circumpolar. It can be found in cool, fresh waters throughout northern Europe, Asia and North America. In North America its range extends as far south as the northern tier of States across the United States. In Maine, the burbot is commonly known as the cusk, although in other areas it is also called the ling, eelpout, loche and lawyer. Unlike the salmonids and Maine’s other coldwater species, the burbot is not noted for its grace and beauty. The body is elongate, almost eel-shaped, with long, soft-rayed dorsal and anal fins that meet a rounded tail. Although it is smooth and slimy to the touch, the skin is embedded with very small, cycloid scales. The head of the burbot is broad and somewhat flattened. It has a large mouth containing several rows of small teeth on the jaws. A single, whisker-like barbell protrudes from the tip of the chin. There are no obvious external differences between males and females. In general, adults are olive brown to dark brown on the back and sides. This background is overlaid with distinctive patterns of dark brown or black markings and spots. -

Indiana Species April 2007
Fishes of Indiana April 2007 The Wildlife Diversity Section (WDS) is responsible for the conservation and management of over 750 species of nongame and endangered wildlife. The list of Indiana's species was compiled by WDS biologists based on accepted taxonomic standards. The list will be periodically reviewed and updated. References used for scientific names are included at the bottom of this list. ORDER FAMILY GENUS SPECIES COMMON NAME STATUS* CLASS CEPHALASPIDOMORPHI Petromyzontiformes Petromyzontidae Ichthyomyzon bdellium Ohio lamprey lampreys Ichthyomyzon castaneus chestnut lamprey Ichthyomyzon fossor northern brook lamprey SE Ichthyomyzon unicuspis silver lamprey Lampetra aepyptera least brook lamprey Lampetra appendix American brook lamprey Petromyzon marinus sea lamprey X CLASS ACTINOPTERYGII Acipenseriformes Acipenseridae Acipenser fulvescens lake sturgeon SE sturgeons Scaphirhynchus platorynchus shovelnose sturgeon Polyodontidae Polyodon spathula paddlefish paddlefishes Lepisosteiformes Lepisosteidae Lepisosteus oculatus spotted gar gars Lepisosteus osseus longnose gar Lepisosteus platostomus shortnose gar Amiiformes Amiidae Amia calva bowfin bowfins Hiodonotiformes Hiodontidae Hiodon alosoides goldeye mooneyes Hiodon tergisus mooneye Anguilliformes Anguillidae Anguilla rostrata American eel freshwater eels Clupeiformes Clupeidae Alosa chrysochloris skipjack herring herrings Alosa pseudoharengus alewife X Dorosoma cepedianum gizzard shad Dorosoma petenense threadfin shad Cypriniformes Cyprinidae Campostoma anomalum central stoneroller -
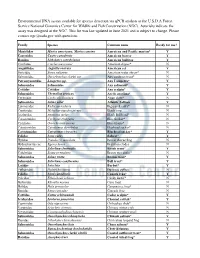
Edna Assay Development
Environmental DNA assays available for species detection via qPCR analysis at the U.S.D.A Forest Service National Genomics Center for Wildlife and Fish Conservation (NGC). Asterisks indicate the assay was designed at the NGC. This list was last updated in June 2021 and is subject to change. Please contact [email protected] with questions. Family Species Common name Ready for use? Mustelidae Martes americana, Martes caurina American and Pacific marten* Y Castoridae Castor canadensis American beaver Y Ranidae Lithobates catesbeianus American bullfrog Y Cinclidae Cinclus mexicanus American dipper* N Anguillidae Anguilla rostrata American eel Y Soricidae Sorex palustris American water shrew* N Salmonidae Oncorhynchus clarkii ssp Any cutthroat trout* N Petromyzontidae Lampetra spp. Any Lampetra* Y Salmonidae Salmonidae Any salmonid* Y Cottidae Cottidae Any sculpin* Y Salmonidae Thymallus arcticus Arctic grayling* Y Cyrenidae Corbicula fluminea Asian clam* N Salmonidae Salmo salar Atlantic Salmon Y Lymnaeidae Radix auricularia Big-eared radix* N Cyprinidae Mylopharyngodon piceus Black carp N Ictaluridae Ameiurus melas Black Bullhead* N Catostomidae Cycleptus elongatus Blue Sucker* N Cichlidae Oreochromis aureus Blue tilapia* N Catostomidae Catostomus discobolus Bluehead sucker* N Catostomidae Catostomus virescens Bluehead sucker* Y Felidae Lynx rufus Bobcat* Y Hylidae Pseudocris maculata Boreal chorus frog N Hydrocharitaceae Egeria densa Brazilian elodea N Salmonidae Salvelinus fontinalis Brook trout* Y Colubridae Boiga irregularis Brown tree snake* -

Sport-Fish-Identification.Pdf
Walleye Walleye have two distinct fins on their back, the first with large spines. Lake Sturgeon They have a yellow-olive back, brassy, silvery sides with yellow spots, a white underside, and white on the lower lobe of the tail. Dusky vertical Lake Sturgeon are a Threatened Species due to population size and bars are often found on the body as well. concerns with viability. Lake Sturgeon have a large brown or grey body covered with tough, leather- like tissue and five rows of bony plates. They have a shark-like, upturned tail and a pointed snout with four barbels. Sauger Lake Whitefish are olive-green to blue on the back, with silvery sides.They Sauger are a Threatened Species due to hybridization, habitat Lakehave a small Whitefish mouth below a rounded snout, and a deeply forked tail. degradation and overharvest. Sauger are golden olive on the back with silver-yellow sides and a white underside. They also have a large spiny dorsal fin, distinct rows of spots on the dorsal fins and three or four dusky vertical bars on the body. Mountain Whitefish have large scales, no spots and small mouths with no Burbot Mountainteeth. Their general Whitefish body colour is a bronze-white or greenish white. Burbot have a slim, brownish black body with smooth skin, a flattened head, and a fin that stretches along the back half of the body. Distinctive barbels hang from the lower jaw and nostrils. Goldeye Northern Pike Goldeye have prominent eyes with bright yellow pupils, a blunt head, and Northern Pike are a long, slender fish with duck-like jaws and a long, flat a deep, compressed body. -

Iowa's Curious Record for Lake Chub
5 American Currents Vol. 41, No. 4 IOWA’S CURIOUS RECORD FOR LAKE CHUB (COUESIUS PLUMBEUS) John Olson Iowa Department of Natural Resources, Des Moines The Lake Chub Couesius( plumbeus) is a relatively large, slender-bodied minnow (Family: Cyprinidae) commonly reaching lengths of 4–5 inches and reaching a reported maximum of nearly 9 inches (Figure 1). There is usually a slender barbel near the tip of the maxillary. The Lake Chub is dark olive dorsally and is dusky white ventrally with a dark lateral band extending from the base of the caudal fin to the snout. Although superficially resembling the Creek Chub (Semotilus atromaculatus), the Lake Chub lacks the Figure 1. Lake Chub (Couesius plumbeus). (Photo by Konrad Creek Chub’s dark spot at the base of the caudal fin and the Schmidt) dark spot at the anterior base of the dorsal fin. The North- ern Pearl Dace (Margariscus nachtriebi) also bears a re- record is based on a collection made in 1954 just west of semblance to the Lake Chub but has a shorter, more blunt Dubuque in northeastern Iowa. snout, has a somewhat less laterally compressed body, and According to published accounts, the Lake Chub is a lacks the red streak along the lower side of the body of habitat generalist, at least within its northern-trending and the large male Pearl Dace (Page and Burr 2011). The Lake Chub has the most northerly and widespread distribution of any North American cyprinid and it ranges from Alas- ka east to Nova Scotia and south to the Great Lakes of the northern United States with scattered relict populations known from the upper Missouri River basin drainage of Wyoming, Colorado, South Dakota, and Nebraska (Figure 2). -

Omineca Burbot Creel, Habitat and Population Assessment Project
Omineca Burbot Creel, Habitat and Population Assessment Project; Results of Surveys of Sport-Ice Fisheries and Assessments of Burbot (Lota lota) Populations in Saxton, Nukko, Eaglet, Cluculz and Norman lakes Grant Agreement #TROP7047 BC Ministry of Environment, Mountain Pine Beetle Response Prepared by the: Lheidli T'enneh First Nation 1041 Whenun Road Prince George, B.C. V2K 5X8 Submitted to: Brady Nelless, Mountain Pine Beetle Biologist BC Ministry of Environment 4051 18th Avenue Prince George, BC V2N 1B3 December 2007 Omineca Burbot Creel, Habitat and Population Assessment Project; 2007 BC Ministry of Environment, Mountain Pine Beetle Response Table of Contents List of Figures .....................................................................................................ii List of Tables.......................................................................................................ii List of Appendices.............................................................................................iii Acknowledgements ...........................................................................................iv Executive Summary............................................................................................1 Introduction.........................................................................................................2 Study Area & Lakes...........................................................................................2 Purpose.............................................................................................................3 -

Huchen (Hucho Hucho) ERSS
Huchen (Hucho hucho) Ecological Risk Screening Summary U.S. Fish & Wildlife Service, April 2011 Revised, January 2019, February 2019 Web Version, 4/30/2019 Photo: Liquid Art. Licensed under CC-SA 4.0 International. Available: https://commons.wikimedia.org/wiki/File:Danube_Salmon_-_Huchen_(Hucho_hucho).jpg. (January 2019). 1 Native Range and Status in the United States Native Range From Froese and Pauly (2019): “Europe: Danube drainage [Austria, Bosnia and Herzegovina, Bulgaria, Croatia, Germany, Hungary, Italy, Romania, Serbia, Slovakia, Slovenia, Switzerland, and Ukraine].” “Population has declined [in Slovenia] due to pollution and river regulation. Conservation measures include artificial propagation and stocking [Povz 1996]. Status of threat: Regionally extinct [Bianco and Ketmaier 2016].” 1 “Considered locally extinct (extirpated) in 1990 [in Switzerland] [Vilcinskas 1993].” “Extinct in the wild in 2000 [in Czech Republic] [Lusk and Hanel 2000]. This species is a native species in the basin of the Black Sea (the rivers Morava and Dyje). At present, its local and time- limited occurrence depends on the stocking material from artificial culture. Conditions that will facilitate the formation of a permanent population under natural conditions are not available [Lusk et al. 2004]. […] Status of threat: extinct in the wild [Lusk et al. 2011].” From Freyhof and Kottelat (2008): “The species is severely fragmented within the Danube drainage, where most populations exclusively depend on stocking and natural reproduction is very limited due to habitat alterations and flow regime changes.” From Grabowska et al. (2010): “The exceptional case is huchen (or Danubian salmon), Hucho hucho. The huchen’s native range in Poland was restricted to two small rivers (Czarna Orawa and Czadeczka) of the Danube River basin, […]” Status in the United States Froese and Pauly (2019) report an introduction to the United States between 1870 and 1874 that did not result in an established population. -

Endangered Species
FEATURE: ENDANGERED SPECIES Conservation Status of Imperiled North American Freshwater and Diadromous Fishes ABSTRACT: This is the third compilation of imperiled (i.e., endangered, threatened, vulnerable) plus extinct freshwater and diadromous fishes of North America prepared by the American Fisheries Society’s Endangered Species Committee. Since the last revision in 1989, imperilment of inland fishes has increased substantially. This list includes 700 extant taxa representing 133 genera and 36 families, a 92% increase over the 364 listed in 1989. The increase reflects the addition of distinct populations, previously non-imperiled fishes, and recently described or discovered taxa. Approximately 39% of described fish species of the continent are imperiled. There are 230 vulnerable, 190 threatened, and 280 endangered extant taxa, and 61 taxa presumed extinct or extirpated from nature. Of those that were imperiled in 1989, most (89%) are the same or worse in conservation status; only 6% have improved in status, and 5% were delisted for various reasons. Habitat degradation and nonindigenous species are the main threats to at-risk fishes, many of which are restricted to small ranges. Documenting the diversity and status of rare fishes is a critical step in identifying and implementing appropriate actions necessary for their protection and management. Howard L. Jelks, Frank McCormick, Stephen J. Walsh, Joseph S. Nelson, Noel M. Burkhead, Steven P. Platania, Salvador Contreras-Balderas, Brady A. Porter, Edmundo Díaz-Pardo, Claude B. Renaud, Dean A. Hendrickson, Juan Jacobo Schmitter-Soto, John Lyons, Eric B. Taylor, and Nicholas E. Mandrak, Melvin L. Warren, Jr. Jelks, Walsh, and Burkhead are research McCormick is a biologist with the biologists with the U.S. -

ECOLOGY of NORTH AMERICAN FRESHWATER FISHES
ECOLOGY of NORTH AMERICAN FRESHWATER FISHES Tables STEPHEN T. ROSS University of California Press Berkeley Los Angeles London © 2013 by The Regents of the University of California ISBN 978-0-520-24945-5 uucp-ross-book-color.indbcp-ross-book-color.indb 1 44/5/13/5/13 88:34:34 AAMM uucp-ross-book-color.indbcp-ross-book-color.indb 2 44/5/13/5/13 88:34:34 AAMM TABLE 1.1 Families Composing 95% of North American Freshwater Fish Species Ranked by the Number of Native Species Number Cumulative Family of species percent Cyprinidae 297 28 Percidae 186 45 Catostomidae 71 51 Poeciliidae 69 58 Ictaluridae 46 62 Goodeidae 45 66 Atherinopsidae 39 70 Salmonidae 38 74 Cyprinodontidae 35 77 Fundulidae 34 80 Centrarchidae 31 83 Cottidae 30 86 Petromyzontidae 21 88 Cichlidae 16 89 Clupeidae 10 90 Eleotridae 10 91 Acipenseridae 8 92 Osmeridae 6 92 Elassomatidae 6 93 Gobiidae 6 93 Amblyopsidae 6 94 Pimelodidae 6 94 Gasterosteidae 5 95 source: Compiled primarily from Mayden (1992), Nelson et al. (2004), and Miller and Norris (2005). uucp-ross-book-color.indbcp-ross-book-color.indb 3 44/5/13/5/13 88:34:34 AAMM TABLE 3.1 Biogeographic Relationships of Species from a Sample of Fishes from the Ouachita River, Arkansas, at the Confl uence with the Little Missouri River (Ross, pers. observ.) Origin/ Pre- Pleistocene Taxa distribution Source Highland Stoneroller, Campostoma spadiceum 2 Mayden 1987a; Blum et al. 2008; Cashner et al. 2010 Blacktail Shiner, Cyprinella venusta 3 Mayden 1987a Steelcolor Shiner, Cyprinella whipplei 1 Mayden 1987a Redfi n Shiner, Lythrurus umbratilis 4 Mayden 1987a Bigeye Shiner, Notropis boops 1 Wiley and Mayden 1985; Mayden 1987a Bullhead Minnow, Pimephales vigilax 4 Mayden 1987a Mountain Madtom, Noturus eleutherus 2a Mayden 1985, 1987a Creole Darter, Etheostoma collettei 2a Mayden 1985 Orangebelly Darter, Etheostoma radiosum 2a Page 1983; Mayden 1985, 1987a Speckled Darter, Etheostoma stigmaeum 3 Page 1983; Simon 1997 Redspot Darter, Etheostoma artesiae 3 Mayden 1985; Piller et al. -

1 WYOMING GAME and FISH DEPARTMENT March 1, 2013
GOVERNOR MATTHEW H. MEAD DIRECTOR WYOMING GAME AND FISH DEPARTMENT SCOTT TALBOTT COMMISSIONERS AARON CLARK – President 5400 Bishop Blvd. Cheyenne, WY 82006 MIKE HEALY – Vice President 1 MARK ANSELMI Phone: (307) 777-4600 Fax: (307) 777-4699 KEITH CULVER RICHARD KLOUDA wgfd.wyo.gov T. CARRIE LITTLE CHARLES PRICE March 1, 2013 MEMORANDUM TO: Honorable Matthew H. Mead FROM: Mark Fowden, Chief, Fish Division COPY TO: Director’s Office, Mike Choma, file SUBJECT: Justification for Emergency Rule, Chapter 46, Fishing Regulations The Sixty-Second Legislature of the State of Wyoming 2013 General Session made a change to W.S. §23-1-101. This change was included in HB 0131/HEA 0052. Specifically, the provision enabled the Wyoming Game and Fish Commission to designate game fish as nongame fish in specific waters. This statutory change was intended to become effective immediately. Due to the Department’s cycle for updating fishing regulations only once every two years, an emergency rule is necessary in order to have this change effective prior to January 1, 2014. The Wyoming Game and Fish Department proposes to amend Wyoming Game and Fish Commission Chapter 46, Fishing Regulations, to edit the definition of “Game Fish” to allow a game fish to be designated as a nongame fish in specific waters. The Department also proposes to designate a game fish, burbot (ling), as a nongame fish in the Green River, Little Snake River, Bear River and Great Divide Basin drainages. This regulation change will allow anglers harvesting burbot in these drainages to dispose of edible portions of this illegally introduced species. -

Imagine the Silver Beauty and the Fighting Spirit of Atlantic Salmon; The
Sakhalin Silver Text and Photos: Clemens Ratschan Imagine the silver beauty and the fighting spirit of Atlantic salmon; the complex, unpredictable life- history of sea trout and combine with the ferocious take and body mass of a predatory taimen. This will give you a glimpse of what fishing for Sakhalin taimen, the silver of the Russian Far East, is about. AM PLEASED TO introduce Siberian taimen, Hucho taimen. No this fish to the readers of wonder, scientists also erroneously Chasing Silver, because in related this far-eastern species to many respects it forms a the large-sized, non-anadromous missing link between the predators of the genus Hucho, which Ifishery for anadromous salmon and is a branch of the salmonoid tree for huchen, a big predatory non- that occurs exclusively in Eurasia. anadromous salmonoid in my home In Central Europe, Hucho hucho is country of Austria (‘Danube salmon’ restricted to the Danube System, in English. See article “Taimen” by where self-sustaining stocks are Wolfgang Hauer, issue 3/2010). presently only found in a handful of Sakhalin taimen is one of the rivers in Germany, Austria, Slovakia least-known salmonid species among and former Yugoslavia. Huchen is non-Russian fishermen; even many very closely related to the already- Russians tend to confuse it with the mentioned Siberian taimen. The latter | 62 | Chasing Silver Fly Fishing Magazine April’s Fav Five www.chasingsilvermagazine.com | 63 | Sakhalin Silver inhabits a distant, vast range from a habits. But one ecological feature expeditions to Japan. Later, the fish few places in European Russia to the is unique – all members of the true was assigned to the genus Parahucho, Lena and Amur rivers in the very far huchen live exclusively in fresh water, with regard to some obvious east of northern Asia.