Drug-Eluting Stents and Balloons—Materials, Structure Designs, and Coating Techniques: a Review
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Magnesium Stearate
SAFETY DATA SHEET Magnesium Stearate SECTION 1 - PRODUCT AND COMPANY IDENTIFICATION 1.1 Product identifier Product Name: Magnesium Stearate Product Code(s): Magnesium Stearate Synonym(s): Octadecanoic acid, magnesium salt; Dibasic magnesium stearate; Stearic acid, magnesium salt REACH Registration Number: No data available 1.2 Relevant identified uses of the substance or mixture and uses advised against General use: For use in industrial and laboratory applications Uses advised against: None known 1.3 Details of the supplier and of the safety data sheet Manufacturer/Distributor Allan Chemical Corporation 235 Margaret King Avenue Ringwood, NJ 07456 USA +1-973-962-4014 1.4 Emergency telephone number Chem Tel +1-813-248-0585 +1-800-255-3924 SECTION 2 - HAZARDS IDENTIFICATION 2.1 Classification of substance or mixture Product definition: Substance Classification in accordance with 29 CFR 1910 (OSHA HCS) and Regulation EC No. 1272/2008 Not a dangerous substance according to OSHA or to European Union Legislation 2.2 Label Elements Not classified as dangerous according to GHS 2.3 Hazards not otherwise classified (HNOC) or not covered by GHS May form combustible dust concentrations in air SECTION 3 - COMPOSITION/INFORMATION ON INGREDIENTS 3.1 Substances % by Weight Ingredient CAS Number EC Number Index Number GHS Classification >99 Magnesium Stearate 557-04-0 209-150-3 ------------ ------------ There are no additional ingredients present which, within the current knowledge of the supplier and in the concentrations applicable, are classified as hazardous to health or the environment and hence, require reporting in this section. 3.2 Mixtures Not applicable SECTION 4 - FIRST AID MEASURES 4.1 Description of first aid measures Inhalation: If exposure to product mist causes respiratory irritation or distress, move the exposed person to fresh air immediately. -

Calcium Stearate Processing
National Organic Standards Board Technical Advisory Panel (TAP) Review Compiled by University of California Sustainable Agriculture Research and Education Program (UC SAREP) for the USDA National Organic Program Calcium Stearate Processing Executive Summary1 A petition is under consideration with respect to NOP regulations subpart G §205.605, governing the use of substances in processed products: Petitioned: Inclusion of calcium stearate on National List of nonagricultural substances allowed in or on processed products labeled as “organic” or “made with organic (specified ingredients or food group(s)).” Calcium stearate is a compound of calcium with a mixture of solid organic acids obtained from edible sources. It is generally used as a solid-phase lubricant that reduces friction between particles of the substance to which it is added. The Petitioner’s intended use is “as a flow agent (anti-dusting agent)” to be used in dry flour based ingredients sold to bakeries (NOSB Petition). The NOP has no prior listing or ruling on the substance. All three reviewers agreed that the substance should be considered synthetic. The reviewers were divided over the use of calcium stearate in food labeled as “organic.” Two of the reviewers felt it should not be allowed in these foods, while one reviewer felt it should be accepted. One reviewer who voted to restrict its use indicated that more information was needed on the nature of the substance and its potential applications, and the other reviewer felt that inclusion of a “synthetic” substance in organics runs contrary to consumer’s expectations regarding organic products. All three reviewers agreed that the substance should be allowed in products labeled as “made with organic…” ingredients. -

Fig. L COMPOSITIONS and METHODS to INHIBIT STEM CELL and PROGENITOR CELL BINDING to LYMPHOID TISSUE and for REGENERATING GERMINAL CENTERS in LYMPHATIC TISSUES
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (10) International Publication Number (43) International Publication Date Χ 23 February 2012 (23.02.2012) WO 2U12/U24519ft ft A2 (51) International Patent Classification: AO, AT, AU, AZ, BA, BB, BG, BH, BR, BW, BY, BZ, A61K 31/00 (2006.01) CA, CH, CL, CN, CO, CR, CU, CZ, DE, DK, DM, DO, DZ, EC, EE, EG, ES, FI, GB, GD, GE, GH, GM, GT, (21) International Application Number: HN, HR, HU, ID, IL, IN, IS, JP, KE, KG, KM, KN, KP, PCT/US201 1/048297 KR, KZ, LA, LC, LK, LR, LS, LT, LU, LY, MA, MD, (22) International Filing Date: ME, MG, MK, MN, MW, MX, MY, MZ, NA, NG, NI, 18 August 201 1 (18.08.201 1) NO, NZ, OM, PE, PG, PH, PL, PT, QA, RO, RS, RU, SC, SD, SE, SG, SK, SL, SM, ST, SV, SY, TH, TJ, TM, (25) Filing Language: English TN, TR, TT, TZ, UA, UG, US, UZ, VC, VN, ZA, ZM, (26) Publication Language: English ZW. (30) Priority Data: (84) Designated States (unless otherwise indicated, for every 61/374,943 18 August 2010 (18.08.2010) US kind of regional protection available): ARIPO (BW, GH, 61/441,485 10 February 201 1 (10.02.201 1) US GM, KE, LR, LS, MW, MZ, NA, SD, SL, SZ, TZ, UG, 61/449,372 4 March 201 1 (04.03.201 1) US ZM, ZW), Eurasian (AM, AZ, BY, KG, KZ, MD, RU, TJ, TM), European (AL, AT, BE, BG, CH, CY, CZ, DE, DK, (72) Inventor; and EE, ES, FI, FR, GB, GR, HR, HU, IE, IS, ΓΓ, LT, LU, (71) Applicant : DEISHER, Theresa [US/US]; 1420 Fifth LV, MC, MK, MT, NL, NO, PL, PT, RO, RS, SE, SI, SK, Avenue, Seattle, WA 98101 (US). -

Magnesium Stearate
United States Department of Agriculture Agricultural Marketing Service | National Organic Program Document Cover Sheet https://www.ams.usda.gov/rules-regulations/organic/national-list/petitioned Document Type: ☐ National List Petition or Petition Update A petition is a request to amend the USDA National Organic Program’s National List of Allowed and Prohibited Substances (National List). Any person may submit a petition to have a substance evaluated by the National Organic Standards Board (7 CFR 205.607(a)). Guidelines for submitting a petition are available in the NOP Handbook as NOP 3011, National List Petition Guidelines. Petitions are posted for the public on the NOP website for Petitioned Substances. ☒ Technical Report A technical report is developed in response to a petition to amend the National List. Reports are also developed to assist in the review of substances that are already on the National List. Technical reports are completed by third-party contractors and are available to the public on the NOP website for Petitioned Substances. Contractor names and dates completed are available in the report. Magnesium Stearate Handling/Processing 1 2 Identification of Petitioned Substance 13 3 Chemical Names: 14 Trade Names: 4 Magnesium stearate 15 N/A 5 Octadecanoic acid magnesium salt 6 Magnesium octadecanoate CAS Numbers: 7 557-04-0 8 Other Name: 9 Stearic acid magnesium salt 10 Magnesium distearate Other Codes: 11 EC-No. 209-150-3 12 INS No. 470(iii) 16 17 Summary of Petitioned Use 18 Magnesium stearate is used as a lubricant or anticaking agent in food processing and handling. Magnesium 19 stearate is currently listed on the National List of Allowed and Prohibited Substances as a synthetic 20 nonagricultural (nonorganic) substance allowed as ingredients in or on processed products labeled as “organic” 21 or “made with organic (specified ingredients or food group(s))” (7 Code of Federal Regulation (CFR) 205.605(b)). -

Pharmaceutical Compositions of Rifaximin Pharmazeutische Rifaximin-Zusammensetzungen Compositions Pharmaceutiques De Rifaximine
(19) TZZ Z__ T (11) EP 2 011 486 B2 (12) NEW EUROPEAN PATENT SPECIFICATION After opposition procedure (45) Date of publication and mention (51) Int Cl.: of the opposition decision: A61K 9/20 (2006.01) A61K 31/44 (2006.01) 12.08.2015 Bulletin 2015/33 (45) Mention of the grant of the patent: 23.05.2012 Bulletin 2012/21 (21) Application number: 08252198.0 (22) Date of filing: 26.06.2008 (54) Pharmaceutical compositions of rifaximin Pharmazeutische Rifaximin-Zusammensetzungen Compositions pharmaceutiques de rifaximine (84) Designated Contracting States: (56) References cited: AT BE BG CH CY CZ DE DK EE ES FI FR GB GR EP-A1- 0 616 808 EP-B1- 1 763 339 HR HU IE IS IT LI LT LU LV MC MT NL NO PL PT WO-A-2006/094737 WO-A2-2006/039022 RO SE SI SK TR US-A- 6 140 355 US-A1- 2005 101 598 (30) Priority: 06.07.2007 IN KO09682007 • DUPONT ET AL: "Treatment of Travelers’ 23.06.2008 EP 08252158 Diarrhea: Randomized Trial Comparing Rifaximin, Rifaximin Plus Loperamide, and (43) Date of publication of application: Loperamide Alone" CLINICAL 07.01.2009 Bulletin 2009/02 GASTROENTEROLOGY AND HEPATOLOGY, AMERICAN GASTROENTEROLOGICAL (60) Divisional application: ASSOCIATION, US, vol. 5, no. 4, 17 April 2007 11176043.5 / 2 420 226 (2007-04-17), pages 451-456, XP022029177 ISSN: 14186563.4 / 2 837 378 1542-3565 • ARYA ET AL: "Rifaximin-the promising anti- (73) Proprietor: Lupin Ltd. microbial for enteric infections" JOURNAL OF Mumbai, Maharashtra 400 098 (IN) INFECTION, ACADEMIC PRESS, LONDON, GB, vol. -

DRUG and MEDICAL DEVICE HIGHLIGHTS Helping You Maintain and Improve Your Health 2019
DRUG AND MEDICAL DEVICE HIGHLIGHTS Helping you maintain and improve your health 2019 DRUG AND MEDICAL DEVICE HIGHLIGHTS 2019 Helping you maintain and improve your health Learn about the new drugs and medical devices that Health Canada approved for sale in Canada, the information we published about potential safety issues, and our other accomplishments in 2019. Health Canada is the federal department responsible for helping the people of Canada maintain and improve their health. Health Canada is committed to improving the lives of all of Canada’s people and to making this country’s population among the healthiest in the world as measured by longevity, lifestyle and effective use of the public health care system. Également disponible en français sous le titre : Préserver et améliorer votre santé : Faits saillants sur les médicaments et les instruments médicaux 2019 To obtain additional information, please contact: Health Canada Address Locator 0900C2 Ottawa, ON K1A 0K9 Tel.: 613-957-2991 Toll free: 1-866-225-0709 Fax: 613-941-5366 TTY: 1-800-465-7735 E-mail: [email protected] © Her Majesty the Queen in Right of Canada, as represented by the Minister of Health, 2020 Publication date: May 2020 This publication may be reproduced for personal or internal use only without permission provided the source is fully acknowledged. Cat.: H161-11E-PDF ISSN: 2562-9816 Pub.: 190484 CONTENTS WELCOME TO OUR 2019 HIGHLIGHTS REPORT ..........................................................................................1 MESSAGE FROM THE CHIEF MEDICAL -

Two Cases of Immediate Stent Fracture After Zotarolimus-Eluting
Case Report http://dx.doi.org/10.4070/kcj.2015.45.1.67 Print ISSN 1738-5520 • On-line ISSN 1738-5555 Korean Circulation Journal Two Cases of Immediate Stent Fracture after Zotarolimus-Eluting Stent Implantation Pil Hyung Lee, MD, Seung-Whan Lee, MD, Jong-Young Lee, MD, Young-Hak Kim, MD, Cheol Whan Lee, MD, Duk-Woo Park, MD, Seong-Wook Park, MD, and Seung-Jung Park, MD Department of Cardiology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea Drug-eluting stent (DES) implantation is currently the standard treatment for various types of coronary artery disease. However, previous reports indicate that stent fractures, which usually occur after a period of time from the initial DES implantation, have increased during the DES era; stent fractures can contribute to unfavorable events such as in-stent restenosis and stent thrombosis. In our present report, we describe two cases of zotarolimus-eluting stent fracture: one that was detected six hours after implementation, and the other case that was detected immediately after deployment. Both anatomical and technical risk factors contributed to these unusual cases of imme- diate stent fracture. (Korean Circ J 2015;45(1):67-70) KEY WORDS: Drug-eluting stents; Percutaneous coronary intervention; Complications. Introduction eluting stent (ZES). Stent fractures have recently become an important concern in Cases the medical community due to their potential association with se- rious conditions such as in-stent restenosis (ISR) and stent throm- Case 1 bosis after drug-eluting stent (DES) implantation.1) Most currently A 62-year-old man, who had undergone cardiac transplantation reported stent fractures are found in lesions implanted with siroli- for advanced heart failure, was referred to our catheterization labo- mus-eluting stents (SES), likely due to the inherent platform material ratory for a regular surveillance coronary angiogram and concomi- and design of such stents.2) Furthermore, while the exact timing of tant intravascular ultrasound (IVUS) imaging. -

Lubricants in Pharmaceutical Solid Dosage Forms
Lubricants 2014, 2, 21-43; doi:10.3390/lubricants2010021 OPEN ACCESS lubricants ISSN 2075-4442 www.mdpi.com/journal/lubricants Review Lubricants in Pharmaceutical Solid Dosage Forms Jinjiang Li * and Yongmei Wu Drug Product Science & Technology, Bristol-Myers Squibb Corporation, 1 Squibb Dr., New Brunswick, NJ 08903, USA; E-Mail: [email protected] * Author to whom correspondence should be addressed; E-Mail: [email protected]; Tel.: +1-732-227-6584; Fax: +1-732-227-3784. Received: 18 December 2013; in revised form: 21 January 2014 / Accepted: 24 January 2014 / Published: 25 February 2014 Abstract: Lubrication plays a key role in successful manufacturing of pharmaceutical solid dosage forms; lubricants are essential ingredients in robust formulations to achieve this. Although many failures in pharmaceutical manufacturing operations are caused by issues related to lubrication, in general, lubricants do not gain adequate attention in the development of pharmaceutical formulations. In this paper, the fundamental background on lubrication is introduced, in which the relationships between lubrication and friction/adhesion forces are discussed. Then, the application of lubrication in the development of pharmaceutical products and manufacturing processes is discussed with an emphasis on magnesium stearate. In particular, the effect of its hydration state (anhydrate, monohydrate, dihydrate, and trihydrate) and its powder characteristics on lubrication efficiency, as well as product and process performance is summarized. In addition, the impact of lubrication on the dynamics of compaction/compression processes and on the mechanical properties of compacts/tablets is presented. Furthermore, the online monitoring of magnesium stearate in a blending process is briefly mentioned. Finally, the chemical compatibility of active pharmaceutical ingredient (API) with magnesium stearate and its reactive impurities is reviewed with examples from the literature illustrating the various reaction mechanisms involved. -

Related Kinases (VRK) Bound to Small-Molecule Inhibitors Identifies Different P-Loop Conformations
Structural characterization of human Vaccinia- Related Kinases (VRK) bound to small-molecule inhibitors identifies different P-loop conformations Rafael M. Couñago1,2*, Charles K. Allerston3, Pavel Savitsky3, Hatylas Azevedo4, Paulo H Godoi1,5, Carrow I. Wells6, Alessandra Mascarello4, Fernando H. de Souza Gama4, Katlin B. Massirer1,2, William J. Zuercher6, Cristiano R.W. Guimarães4 and Opher Gileadi1,3 1. Structural Genomics Consortium, Universidade Estadual de Campinas — UNICAMP, Campinas, SP, Brazil. 2. Centro de Biologia Molecular e Engenharia Genética, Universidade Estadual de Campinas, Campinas, SP, Brazil. 3. Structural Genomics Consortium and Target Discovery Institute, Nuffield Department of Clinical Medicine, University of Oxford, UK. 4. Aché Laboratórios Farmacêuticos SA, Guarulhos, SP, Brazil. 5. Department of Biochemistry and Tissue Biology, Institute of Biology, State University of Campinas, Campinas, Brazil. 6. Structural Genomics Consortium, UNC Eshelman School of Pharmacy, University of North Carolina at Chapel Hill, NC, USA. *Correspondence to Rafael M. Couñago: [email protected] (RMC) Supplementary information 1 SUPPLEMENTARY METHODS PKIS results analyses - hierarchical cluster analysis (HCL) A hierarchical clustering (HCL) analysis was performed to group kinases based on their inhibition patterns across the compounds. The average distance clustering method was employed, using sample tree selection and sample leaf order optimization. The distance metric used was the Pearson correlation and the HCL analysis was performed in the TmeV software 1. SUPPLEMENTARY REFERENCES 1 Saeed, A. I. et al. TM4: a free, open-source system for microarray data management and analysis. BioTechniques 34, 374-378, (2003). SUPPLEMENTARY FIGURES LEGENDS Supplementary Figure S1: Hierarchical clustering analysis of PKIS data. Hierarchical clustering analysis of PKIS data. -
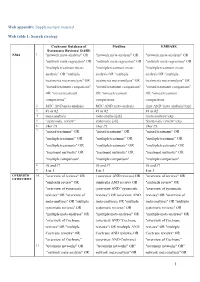
1 Web Appendix: Supplementary Material Web Table 1
Web appendix: Supplementary material Web table 1: Search strategy Cochrane Database of Medline EMBASE Systematic Reviews/ DARE NMA 1 "network meta-analysis" OR "network meta-analysis" OR "network meta-analysis" OR "network meta-regression" OR "network meta-regression" OR "network meta-regression" OR "multiple treatment meta- "multiple treatment meta- "multiple treatment meta- analysis” OR "multiple analysis OR "multiple analysis OR "multiple treatments meta-analysis" OR treatments meta-analysis" OR treatments meta-analysis" OR "mixed treatment comparison" "mixed treatment comparison" "mixed treatment comparison" OR "mixed treatment OR "mixed treatment OR "mixed treatment comparisons” comparisons comparisons 2 MTC AND meta-analysis MTC AND meta-analysis (mtc AND 'meta analysis'/exp) 3 #1 or #2 #1 or #2 #1 or #2 4 meta-analysis meta-analysis[sb] 'meta analysis'/exp 5 “systematic review” systematic [sb] 'Systematic review '/exp 6 #4or #5 #4or #5 #4or #5 7 "mixed treatment" OR "mixed treatment" OR "mixed treatment" OR "multiple treatment" OR "multiple treatment" OR "multiple treatment" OR "multiple treatments" OR "multiple treatments" OR "multiple treatments" OR "treatment networks" OR "treatment networks" OR "treatment networks" OR "multiple comparison" "multiple comparison" "multiple comparison" 8 #6 and #7 #6 and #7 #6 and #7 9 8 or 3 8 or 3 8 or 3 OVERVIEW 10 "overview of reviews" OR ( overview AND reviews) OR "overview of reviews" OR OF REVIEWS "umbrella review" OR (umbrella AND review) OR "umbrella review" OR "overview of systematic -

That Are Not Lielu Uutuullittu
THAT ARE NOT LIELUUS009987356B2 UUTUULLITTU (12 ) United States Patent ( 10 ) Patent No. : US 9 ,987 , 356 B2 Reimann et al. ( 45) Date of Patent : Jun . 5 , 2018 (54 ) ANTI -CD40 ANTIBODIES AND METHODS 762 A 12 / 1997 Queen et al. 5 , 801, 227 A 9 / 1998 Fanslow , III et al. OF ADMINISTERING THEREOF 5 , 874 ,082 A 2 / 1999 de Boer 6 ,004 , 552 A 12 / 1999 de Boer et al. ( 75 ) Inventors: Keith A . Reimann , Marblehead , MA 6 ,051 ,228 A 4 / 2000 Aruffo et al. (US ) ; Rijian Wang , Saugus, MA (US ) ; 6 ,054 ,297 A 4 / 2000 Carter et al . Christian P . Larsen , Atlanta , GA (US ) 6 ,056 , 959 A 5 /2000 de Boer et al. 6 , 132 , 978 A 10 /2000 Gelfand et al. 6 , 280 , 957 B18 / 2001 Sayegh et al. ( 73) Assignees : Beth Israel Deaconess Medical 6 , 312 ,693 B1 11/ 2001 Aruffo et al. Center , Inc ., Boston , MA (US ) ; Emory 6 , 315 , 998 B111 / 2001 de Boer et al. University , Atlanta , GA (US ) 6 , 413 ,514 B1 7 / 2002 Aruffo et al. 6 , 632, 927 B2 10 /2003 Adair et al. ( * ) Notice : Subject to any disclaimer , the term of this 7 ,063 , 845 B2 6 / 2006 Mikayama et al. patent is extended or adjusted under 35 7 , 193 , 064 B2 3 / 2007 Mikayama et al. 7 , 288 , 251 B2 10 / 2007 Bedian et al . U . S . C . 154 ( b ) by 464 days . 7 , 361 , 345 B2 4 /2008 de Boer et al. 7 , 445 , 780 B2 11/ 2008 Chu et al. (21 ) Appl . No. -

WHO Drug Information Vol. 20, No. 3, 2006
WHO DRUG INFORMATION VOLUME 20• NUMBER 3 • 2006 RECOMMENDED INN LIST 56 INTERNATIONAL NONPROPRIETARY NAMES FOR PHARMACEUTICAL SUBSTANCES WORLD HEALTH ORGANIZATION • GENEVA WHO Drug Information Vol 20, No. 3, 2006 World Health Organization WHO Drug Information Contents Biomedicines and Vaccines Counterfeit taskforce launched 192 Fake artesunate warning sheet 193 Monoclonal antibodies: a special Resolution on counterfeiting 193 regulatory challenge? 165 Improving world health through regulation of biologicals 173 Regulatory Action and News Influenza virus vaccines for 2006–2007 Safety and Efficacy Issues northern hemisphere 196 Trastuzumab approved for primary Global progress in monitoring immuniza- breast cancer 196 tion adverse events 180 Emergency contraception over-the- Intracranial haemorrhage in patients counter 197 receiving tipranavir 180 Ocular Fusarium infections: ReNu Infliximab: hepatosplenic T cell MoistureLoc® voluntary withdrawal 197 lymphoma 181 Saquinavir: withdrawal of soft gel Lamotrigine: increased risk of non- capsule Fortovase® 197 syndromic oral clefts 181 Latest list of prequalified products and Biphosphonates: osteonecrosis of manufacturers 198 the jaw 181 Clopidogrel: new medical use 199 Enoxaparin dosage in chronic kidney Dronedarone: withdrawal of marketing disease 182 authorization 199 Terbinafine and life-threatening blood dyscrasias 183 Adverse reactions in children: why Recent Publications, report? 183 Hepatitis B reactivation and anti-TNF- Information and Events alpha agents 185 MSF issues ninth antiretroviral