Metagenome-Assembled Genomes of Phytoplankton Communities Across
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
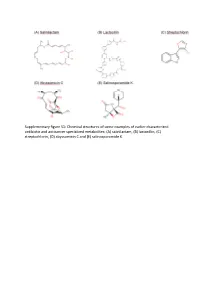
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Genomics 98 (2011) 370–375
Genomics 98 (2011) 370–375 Contents lists available at ScienceDirect Genomics journal homepage: www.elsevier.com/locate/ygeno Whole-genome comparison clarifies close phylogenetic relationships between the phyla Dictyoglomi and Thermotogae Hiromi Nishida a,⁎, Teruhiko Beppu b, Kenji Ueda b a Agricultural Bioinformatics Research Unit, Graduate School of Agricultural and Life Sciences, University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-8657, Japan b Life Science Research Center, College of Bioresource Sciences, Nihon University, Fujisawa, Japan article info abstract Article history: The anaerobic thermophilic bacterial genus Dictyoglomus is characterized by the ability to produce useful Received 2 June 2011 enzymes such as amylase, mannanase, and xylanase. Despite the significance, the phylogenetic position of Accepted 1 August 2011 Dictyoglomus has not yet been clarified, since it exhibits ambiguous phylogenetic positions in a single gene Available online 7 August 2011 sequence comparison-based analysis. The number of substitutions at the diverging point of Dictyoglomus is insufficient to show the relationships in a single gene comparison-based analysis. Hence, we studied its Keywords: evolutionary trait based on whole-genome comparison. Both gene content and orthologous protein sequence Whole-genome comparison Dictyoglomus comparisons indicated that Dictyoglomus is most closely related to the phylum Thermotogae and it forms a Bacterial systematics monophyletic group with Coprothermobacter proteolyticus (a constituent of the phylum Firmicutes) and Coprothermobacter proteolyticus Thermotogae. Our findings indicate that C. proteolyticus does not belong to the phylum Firmicutes and that the Thermotogae phylum Dictyoglomi is not closely related to either the phylum Firmicutes or Synergistetes but to the phylum Thermotogae. © 2011 Elsevier Inc. -

Diversity of Understudied Archaeal and Bacterial Populations of Yellowstone National Park: from Genes to Genomes Daniel Colman
University of New Mexico UNM Digital Repository Biology ETDs Electronic Theses and Dissertations 7-1-2015 Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes Daniel Colman Follow this and additional works at: https://digitalrepository.unm.edu/biol_etds Recommended Citation Colman, Daniel. "Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes." (2015). https://digitalrepository.unm.edu/biol_etds/18 This Dissertation is brought to you for free and open access by the Electronic Theses and Dissertations at UNM Digital Repository. It has been accepted for inclusion in Biology ETDs by an authorized administrator of UNM Digital Repository. For more information, please contact [email protected]. Daniel Robert Colman Candidate Biology Department This dissertation is approved, and it is acceptable in quality and form for publication: Approved by the Dissertation Committee: Cristina Takacs-Vesbach , Chairperson Robert Sinsabaugh Laura Crossey Diana Northup i Diversity of understudied archaeal and bacterial populations from Yellowstone National Park: from genes to genomes by Daniel Robert Colman B.S. Biology, University of New Mexico, 2009 DISSERTATION Submitted in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy Biology The University of New Mexico Albuquerque, New Mexico July 2015 ii DEDICATION I would like to dedicate this dissertation to my late grandfather, Kenneth Leo Colman, associate professor of Animal Science in the Wool laboratory at Montana State University, who even very near the end of his earthly tenure, thought it pertinent to quiz my knowledge of oxidized nitrogen compounds. He was a man of great curiosity about the natural world, and to whom I owe an acknowledgement for his legacy of intellectual (and actual) wanderlust. -

Biology and Systematics of Heterokont and Haptophyte Algae1
American Journal of Botany 91(10): 1508±1522. 2004. BIOLOGY AND SYSTEMATICS OF HETEROKONT AND HAPTOPHYTE ALGAE1 ROBERT A. ANDERSEN Bigelow Laboratory for Ocean Sciences, P.O. Box 475, West Boothbay Harbor, Maine 04575 USA In this paper, I review what is currently known of phylogenetic relationships of heterokont and haptophyte algae. Heterokont algae are a monophyletic group that is classi®ed into 17 classes and represents a diverse group of marine, freshwater, and terrestrial algae. Classes are distinguished by morphology, chloroplast pigments, ultrastructural features, and gene sequence data. Electron microscopy and molecular biology have contributed signi®cantly to our understanding of their evolutionary relationships, but even today class relationships are poorly understood. Haptophyte algae are a second monophyletic group that consists of two classes of predominately marine phytoplankton. The closest relatives of the haptophytes are currently unknown, but recent evidence indicates they may be part of a large assemblage (chromalveolates) that includes heterokont algae and other stramenopiles, alveolates, and cryptophytes. Heter- okont and haptophyte algae are important primary producers in aquatic habitats, and they are probably the primary carbon source for petroleum products (crude oil, natural gas). Key words: chromalveolate; chromist; chromophyte; ¯agella; phylogeny; stramenopile; tree of life. Heterokont algae are a monophyletic group that includes all (Phaeophyceae) by Linnaeus (1753), and shortly thereafter, photosynthetic organisms with tripartite tubular hairs on the microscopic chrysophytes (currently 5 Oikomonas, Anthophy- mature ¯agellum (discussed later; also see Wetherbee et al., sa) were described by MuÈller (1773, 1786). The history of 1988, for de®nitions of mature and immature ¯agella), as well heterokont algae was recently discussed in detail (Andersen, as some nonphotosynthetic relatives and some that have sec- 2004), and four distinct periods were identi®ed. -

Thermaerobacter Marianensis Type Strain (7P75a)
Lawrence Berkeley National Laboratory Recent Work Title Complete genome sequence of Thermaerobacter marianensis type strain (7p75a). Permalink https://escholarship.org/uc/item/2km2g1rj Journal Standards in genomic sciences, 3(3) ISSN 1944-3277 Authors Han, Cliff Gu, Wei Zhang, Xiaojing et al. Publication Date 2010-12-15 DOI 10.4056/sigs.1373474 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Standards in Genomic Sciences (2010) 3:337-345 DOI:10.4056/sigs.1373474 Complete genome sequence of Thermaerobacter T marianensis type strain (7p75a ) Cliff Han1,2, Wei Gu1,2, Xiaojing Zhang1,2, Alla Lapidus1, Matt Nolan1, Alex Copeland1, Susan Lucas1, Tijana Glavina Del Rio1, Hope Tice1, Jan-Fang Cheng1, Roxane Tapia1,2, Lynne Goodwin1,2, Sam Pitluck1, Ioanna Pagani1, Natalia Ivanova1, Konstantinos Mavromatis1, Natalia Mikhailova1, Amrita Pati1, Amy Chen3, Krishna Palaniappan3, Miriam Land1,4, Loren Hauser1,4, Yun-Juan Chang1,4, Cynthia D. Jeffries1,4, Susanne Schneider5, Manfred Rohde6, Markus Göker5, Rüdiger Pukall5, Tanja Woyke1, James Bristow1, Jonathan A. Eisen1,7, Victor Markowitz3, Philip Hugenholtz1, Nikos C. Kyrpides1, Hans-Peter Klenk5*, and John C. Detter1,2 1 DOE Joint Genome Institute, Walnut Creek, California, USA 2 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 3 Biological Data Management and Technology Center, Lawrence Berkeley National Laboratory, Berkeley, California, USA 4 Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA 5 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 6 HZI – Helmholtz Centre for Infection Research, Braunschweig, Germany 7 University of California Davis Genome Center, Davis, California, USA *Corresponding author: Hans-Peter Klenk Keywords: strictly aerobic, none-motile, Gram-variable, thermophilic, chemoheterotrophic, deep-sea, family Incertae Sedis XVII, Clostridiales, GEBA Thermaerobacter marianensis Takai et al. -

Article-Associated Bac- Teria and Colony Isolation in Soft Agar Medium for Bacteria Unable to Grow at the Air-Water Interface
Biogeosciences, 8, 1955–1970, 2011 www.biogeosciences.net/8/1955/2011/ Biogeosciences doi:10.5194/bg-8-1955-2011 © Author(s) 2011. CC Attribution 3.0 License. Diversity of cultivated and metabolically active aerobic anoxygenic phototrophic bacteria along an oligotrophic gradient in the Mediterranean Sea C. Jeanthon1,2, D. Boeuf1,2, O. Dahan1,2, F. Le Gall1,2, L. Garczarek1,2, E. M. Bendif1,2, and A.-C. Lehours3 1Observatoire Oceanologique´ de Roscoff, UMR7144, INSU-CNRS – Groupe Plancton Oceanique,´ 29680 Roscoff, France 2UPMC Univ Paris 06, UMR7144, Adaptation et Diversite´ en Milieu Marin, Station Biologique de Roscoff, 29680 Roscoff, France 3CNRS, UMR6023, Microorganismes: Genome´ et Environnement, Universite´ Blaise Pascal, 63177 Aubiere` Cedex, France Received: 21 April 2011 – Published in Biogeosciences Discuss.: 5 May 2011 Revised: 7 July 2011 – Accepted: 8 July 2011 – Published: 20 July 2011 Abstract. Aerobic anoxygenic phototrophic (AAP) bac- detected in the eastern basin, reflecting the highest diver- teria play significant roles in the bacterioplankton produc- sity of pufM transcripts observed in this ultra-oligotrophic tivity and biogeochemical cycles of the surface ocean. In region. To our knowledge, this is the first study to document this study, we applied both cultivation and mRNA-based extensively the diversity of AAP isolates and to unveil the ac- molecular methods to explore the diversity of AAP bacte- tive AAP community in an oligotrophic marine environment. ria along an oligotrophic gradient in the Mediterranean Sea By pointing out the discrepancies between culture-based and in early summer 2008. Colony-forming units obtained on molecular methods, this study highlights the existing gaps in three different agar media were screened for the production the understanding of the AAP bacteria ecology, especially in of bacteriochlorophyll-a (BChl-a), the light-harvesting pig- the Mediterranean Sea and likely globally. -

Sporulation Evolution and Specialization in Bacillus
bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Research article From root to tips: sporulation evolution and specialization in Bacillus subtilis and the intestinal pathogen Clostridioides difficile Paula Ramos-Silva1*, Mónica Serrano2, Adriano O. Henriques2 1Instituto Gulbenkian de Ciência, Oeiras, Portugal 2Instituto de Tecnologia Química e Biológica, Universidade Nova de Lisboa, Oeiras, Portugal *Corresponding author: Present address: Naturalis Biodiversity Center, Marine Biodiversity, Leiden, The Netherlands Phone: 0031 717519283 Email: [email protected] (Paula Ramos-Silva) Running title: Sporulation from root to tips Keywords: sporulation, bacterial genome evolution, horizontal gene transfer, taxon- specific genes, Bacillus subtilis, Clostridioides difficile 1 bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Abstract Bacteria of the Firmicutes phylum are able to enter a developmental pathway that culminates with the formation of a highly resistant, dormant spore. Spores allow environmental persistence, dissemination and for pathogens, are infection vehicles. In both the model Bacillus subtilis, an aerobic species, and in the intestinal pathogen Clostridioides difficile, an obligate anaerobe, sporulation mobilizes hundreds of genes. -

The Mitochondrial Genome of the Stramenopile Proteromonas Lacertae
GBE A Linear Molecule with Two Large Inverted Repeats: The Mitochondrial Genome of the Stramenopile Proteromonas lacertae Vicente Pe´ rez-Brocalà, Revital Shahar-Golan, and C. Graham Clark* Department of Infectious and Tropical Diseases, London School of Hygiene and Tropical Medicine, London, United Kingdom *Corresponding author: E-mail: [email protected]. àPresent address: Centro Superior de Investigacio´ n en Salud Pu´ blica, A´ rea de Geno´ mica y Salud, Avda. de Catalun˜ a 21, Valencia, Spain Accepted: 12 April 2010 Genome sequence deposited with accession number GU563431. Abstract Downloaded from Mitochondrial evolution has given rise to a complex array of organelles, ranging from classical aerobic mitochondria to mitochondrial remnants known as hydrogenosomes and mitosomes. The latter are found in anaerobic eukaryotes, and these highly derived organelles often retain only scant evidence of their mitochondrial origins. Intermediate evolutionary stages have also been reported as facultatively or even strictly anaerobic mitochondria, and hydrogenosomes that still retain some http://gbe.oxfordjournals.org/ mitochondrial features. However, the diversity among these organelles with transitional features remains rather unclear and barely studied. Here, we report the sequence, structure, and gene content of the mitochondrial DNA of the anaerobic stramenopile Proteromonas lacertae. It has a linear genome with a unique central region flanked by two identical large inverted repeats containing numerous genes and ‘‘telomeres’’ with short inverted repeats. Comparison with the organelle genome of the strictly anaerobic human parasite Blastocystis reveals that, despite the close similarity of the sequences, features such as the genome structure display striking differences. It remains unclear whether the virtually identical gene repertoires are the result of convergence or descent. -

Which Organisms Are Used for Anti-Biofouling Studies
Table S1. Semi-systematic review raw data answering: Which organisms are used for anti-biofouling studies? Antifoulant Method Organism(s) Model Bacteria Type of Biofilm Source (Y if mentioned) Detection Method composite membranes E. coli ATCC25922 Y LIVE/DEAD baclight [1] stain S. aureus ATCC255923 composite membranes E. coli ATCC25922 Y colony counting [2] S. aureus RSKK 1009 graphene oxide Saccharomycetes colony counting [3] methyl p-hydroxybenzoate L. monocytogenes [4] potassium sorbate P. putida Y. enterocolitica A. hydrophila composite membranes E. coli Y FESEM [5] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) K. pneumonia ATCC13883 P. aeruginosa BAA-1744 composite membranes E. coli Y SEM [6] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) graphene oxide E. coli ATCC25922 Y colony counting [7] S. aureus ATCC9144 P. aeruginosa ATCCPAO1 composite membranes E. coli Y measuring flux [8] (unspecified/unique sample type) graphene oxide E. coli Y colony counting [9] (unspecified/unique SEM sample type) LIVE/DEAD baclight S. aureus stain (unspecified/unique sample type) modified membrane P. aeruginosa P60 Y DAPI [10] Bacillus sp. G-84 LIVE/DEAD baclight stain bacteriophages E. coli (K12) Y measuring flux [11] ATCC11303-B4 quorum quenching P. aeruginosa KCTC LIVE/DEAD baclight [12] 2513 stain modified membrane E. coli colony counting [13] (unspecified/unique colony counting sample type) measuring flux S. aureus (unspecified/unique sample type) modified membrane E. coli BW26437 Y measuring flux [14] graphene oxide Klebsiella colony counting [15] (unspecified/unique sample type) P. aeruginosa (unspecified/unique sample type) graphene oxide P. aeruginosa measuring flux [16] (unspecified/unique sample type) composite membranes E. -

How to Cite Complete Issue More Information About This
Revista Internacional de Contaminación Ambiental ISSN: 0188-4999 [email protected] Universidad Nacional Autónoma de México México Luis-Villaseñor, Irasema E.; Zamudio-Armenta, Olga O.; Voltolina, Domenico; Rochin- Arenas, Jesús A.; Gómez-Gil, Bruno; Audelo-Naranjo, Juan M.; Flores-Higuera, Francisco A. BACTERIAL COMMUNITIES OF THE OYSTERS Crassostrea corteziensis AND C. sikamea OF COSPITA BAY, SINALOA, MEXICO Revista Internacional de Contaminación Ambiental, vol. 34, no. 2, 2018, May-July, pp. 203-213 Universidad Nacional Autónoma de México México DOI: https://doi.org/10.20937/RICA.2018.34.02.02 Available in: https://www.redalyc.org/articulo.oa?id=37056657002 How to cite Complete issue Scientific Information System Redalyc More information about this article Network of Scientific Journals from Latin America and the Caribbean, Spain and Journal's webpage in redalyc.org Portugal Project academic non-profit, developed under the open access initiative Rev. Int. Contam. Ambie. 34 (2) 203-213, 2018 DOI: 10.20937/RICA.2018.34.02.02 BACTERIAL COMMUNITIES OF THE OYSTERS Crassostrea corteziensis AND C. sikamea OF COSPITA BAY, SINALOA, MEXICO Irasema E. LUIS-VILLASEÑOR1*, Olga O. ZAMUDIO-ARMENTA1, Domenico VOLTOLINA2, Jesús A. ROCHIN-ARENAS1, Bruno GÓMEZ-GIL3, Juan M. AUDELO-NARANJO1 y Francisco A. FLORES-HIGUERA1 1 Universidad Autónoma de Sinaloa, Facultad de Ciencias del Mar, Paseo Claussen s/n, Mazatlán, Sinaloa, México 2 Centro de Investigaciones Biológicas del Noroeste, Apartado 1132, Mazatlán, Sinaloa, México 3 Centro de Investigación en Alimentación y Desarrollo, Unidad Mazatlán, Apartado 711, Mazatlán, Sinaloa, México * Author for correspondence; [email protected] (Received December 2016; accepted September 2017) Key words: bacteria, cultured oysters, wild oysters ABSTRACT This work aimed to quantify the bacterial loads and determine the taxonomic composi- tion of the microbial communities of oysters Crassostrea corteziensis and C. -

Phylogenetic Position of Karotomorpha and Paraphyly of Proteromonadidae
Molecular Phylogenetics and Evolution 43 (2007) 1167–1170 www.elsevier.com/locate/ympev Short communication Phylogenetic position of Karotomorpha and paraphyly of Proteromonadidae Martin Kostka a,¤, Ivan Cepicka b, Vladimir Hampl a, Jaroslav Flegr a a Department of Parasitology, Faculty of Science, Charles University, Vinicna 7, 128 44 Prague, Czech Republic b Department of Zoology, Faculty of Science, Charles University, Vinicna 7, 128 44 Prague, Czech Republic Received 9 May 2006; revised 17 October 2006; accepted 2 November 2006 Available online 17 November 2006 1. Introduction tional region is alike that of proteromonadids as well, double transitional helix is present. These similarities led Patterson The taxon Slopalinida (Patterson, 1985) comprises two (1985) to unite the two families in the order Slopalinida and families of anaerobic protists living as commensals in the to postulate the paraphyly of the family Proteromonadidae intestine of vertebrates. The proteromonadids are small (Karotomorpha being closer to the opalinids). The ultrastruc- Xagellates (ca. 15 m) with one nucleus, a single large mito- ture of Xagellar transition region and proposed homology chondrion with tubular cristae, Golgi apparatus and a Wbril- between the somatonemes of Proteromonas and mastigo- lar rhizoplast connecting the basal bodies and nucleus nemes of heterokont Xagellates led him further to conclude (Brugerolle and Mignot, 1989). The number of Xagella diVers that the slopalinids are relatives of the heterokont algae, in between the two genera belonging to the family: Protero- other words that they belong among stramenopiles. Phyloge- monas, the commensal of urodelans, lizards, and rodents, has netic analysis of Silberman et al. (1996) not only conWrmed two Xagella, whereas Karotomorpha, the commensal of frogs that Proteromonas is a stramenopile, but also showed that its and other amphibians, has four Xagella. -

Evidence for Endosymbiotic Gene Transfer and the Early Evolution of Photosynthesis
Evolution of Glutamine Synthetase in Heterokonts: Evidence for Endosymbiotic Gene Transfer and the Early Evolution of Photosynthesis Deborah L. Robertson and Aure´lien Tartar Biology Department, Clark University Although the endosymbiotic evolution of chloroplasts through primary and secondary associations is well established, the evolutionary timing and stability of the secondary endosymbiotic events is less well resolved. Heterokonts include both photosynthetic and nonphotosynthetic members and the nonphotosynthetic lineages branch basally in phylogenetic reconstructions. Molecular and morphological data indicate that heterokont chloroplasts evolved via a secondary endo- symbiosis, involving a heterotrophic host cell and a photosynthetic ancestor of the red algae and this endosymbiotic event may have preceded the divergence of heterokonts and alveolates. If photosynthesis evolved early in this lineage, nuclear genomes of the nonphotosynthetic groups may contain genes that are not essential to photosynthesis but were derived from the endosymbiont genome through gene transfer. These genes offer the potential to trace the evolutionary history of chloroplast gains and losses within these lineages. Glutamine synthetase (GS) is essential for ammonium assimilation and glutamine biosynthesis in all organisms. Three paralogous gene families (GSI, GSII, and GSIII) have been identified and are broadly distributed among prokaryotic and eukaryotic lineages. In diatoms (Heterokonta), the nuclear-encoded chloroplast and cytosolic-localized GS isoforms are encoded by members of the GSII and GSIII family, respectively. Here, we explore the evolutionary history of GSII in both photosynthetic and nonphotosynthetic heterokonts, red algae, and other eukaryotes. GSII cDNA sequences were obtained from two species of oomycetes by polymerase chain reaction amplification. Additional GSII sequences from eukaryotes and bacteria were obtained from publicly available databases and genome projects.