The Molecular Determinants of Tumor Cell Modulation Of
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supplementary Data
Supplementary Data for Quantitative Changes in the Mitochondrial Proteome from Subjects with Mild Cognitive Impairment, Early Stage and Late Stage Alzheimer’s disease Table 1 - 112 unique, non-redundant proteins identified and quantified in at least two of the three analytical replicates for all three disease stages. Table 2 - MCI mitochondrial samples, Protein Summary Table 3 - MCI mitochondrial samples, Experiment 1 Table 4 - MCI mitochondrial samples, Experiment 2 Table 5 - MCI mitochondrial samples, Experiment 3 Table 6 - EAD Mitochondrial Study, Protein Summary Table 7 - EAD Mitochondrial Study, Experiment 1 Table 8 - EAD Mitochondrial Study, Experiment 2 Table 9 - EAD Mitochondrial Study, Experiment 3 Table 10 - LAD Mitochondrial Study, Protein Summary Table 11 - LAD Mitochondrial Study, Experiment 1 Table 12 - LAD Mitochondrial Study, Experiment 2 Table 13 - LAD Mitochondrial Study, Experiment 3 Supplemental Table 1. 112 unique, non-redundant proteins identified and quantified in at least two of the three analytical replicates for all three disease stages. Description Data MCI EAD LAD AATM_HUMAN (P00505) Aspartate aminotransferase, mitochondrial precursor (EC Mean 1.43 1.70 1.31 2.6.1.1) (Transaminase A) (Glutamate oxaloacetate transaminase 2) [MASS=47475] SEM 0.07 0.09 0.09 Count 3.00 3.00 3.00 ACON_HUMAN (Q99798) Aconitate hydratase, mitochondrial precursor (EC 4.2.1.3) Mean 1.24 1.61 1.19 (Citrate hydro-lyase) (Aconitase) [MASS=85425] SEM 0.05 0.17 0.18 Count 3.00 2.00 3.00 ACPM_HUMAN (O14561) Acyl carrier protein, mitochondrial -
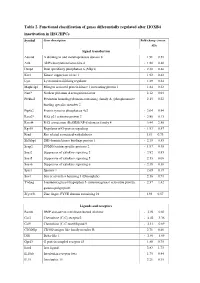
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Table 2. Significant
Table 2. Significant (Q < 0.05 and |d | > 0.5) transcripts from the meta-analysis Gene Chr Mb Gene Name Affy ProbeSet cDNA_IDs d HAP/LAP d HAP/LAP d d IS Average d Ztest P values Q-value Symbol ID (study #5) 1 2 STS B2m 2 122 beta-2 microglobulin 1452428_a_at AI848245 1.75334941 4 3.2 4 3.2316485 1.07398E-09 5.69E-08 Man2b1 8 84.4 mannosidase 2, alpha B1 1416340_a_at H4049B01 3.75722111 3.87309653 2.1 1.6 2.84852656 5.32443E-07 1.58E-05 1110032A03Rik 9 50.9 RIKEN cDNA 1110032A03 gene 1417211_a_at H4035E05 4 1.66015788 4 1.7 2.82772795 2.94266E-05 0.000527 NA 9 48.5 --- 1456111_at 3.43701477 1.85785922 4 2 2.8237185 9.97969E-08 3.48E-06 Scn4b 9 45.3 Sodium channel, type IV, beta 1434008_at AI844796 3.79536664 1.63774235 3.3 2.3 2.75319499 1.48057E-08 6.21E-07 polypeptide Gadd45gip1 8 84.1 RIKEN cDNA 2310040G17 gene 1417619_at 4 3.38875643 1.4 2 2.69163229 8.84279E-06 0.0001904 BC056474 15 12.1 Mus musculus cDNA clone 1424117_at H3030A06 3.95752801 2.42838452 1.9 2.2 2.62132809 1.3344E-08 5.66E-07 MGC:67360 IMAGE:6823629, complete cds NA 4 153 guanine nucleotide binding protein, 1454696_at -3.46081884 -4 -1.3 -1.6 -2.6026947 8.58458E-05 0.0012617 beta 1 Gnb1 4 153 guanine nucleotide binding protein, 1417432_a_at H3094D02 -3.13334396 -4 -1.6 -1.7 -2.5946297 1.04542E-05 0.0002202 beta 1 Gadd45gip1 8 84.1 RAD23a homolog (S. -

Targeted and Novel Therapy in Advanced Gastric Cancer Julie H
Selim et al. Exp Hematol Oncol (2019) 8:25 https://doi.org/10.1186/s40164-019-0149-6 Experimental Hematology & Oncology REVIEW Open Access Targeted and novel therapy in advanced gastric cancer Julie H. Selim1 , Shagufta Shaheen2 , Wei‑Chun Sheu3 and Chung‑Tsen Hsueh4* Abstract The systemic treatment options for advanced gastric cancer (GC) have evolved rapidly in recent years. We have reviewed the recent data of clinical trial incorporating targeted agents, including inhibitors of angiogenesis, human epidermal growth factor receptor 2 (HER2), mesenchymal–epithelial transition, epidermal growth factor receptor, mammalian target of rapamycin, claudin‑18.2, programmed death‑1 and DNA. Addition of trastuzumab to platinum‑ based chemotherapy has become standard of care as front‑line therapy in advanced GC overexpressing HER2. In the second‑line setting, ramucirumab with paclitaxel signifcantly improves overall survival compared to paclitaxel alone. For patients with refractory disease, apatinib, nivolumab, ramucirumab and TAS‑102 have demonstrated single‑agent activity with improved overall survival compared to placebo alone. Pembrolizumab has demonstrated more than 50% response rate in microsatellite instability‑high tumors, 15% response rate in tumors expressing programmed death ligand 1, and non‑inferior outcome in frst‑line treatment compared to chemotherapy. This review summarizes the current state and progress of research on targeted therapy for advanced GC. Keywords: Gastric cancer, Targeted therapy, Human epidermal growth factor receptor 2, Programmed death‑1, Vascular endothelial growth factor receptor 2 Background GC mortality which is consistent with overall decrease in Gastric cancer (GC), including adenocarcinoma of the GC-related deaths [4]. gastroesophageal junction (GEJ) and stomach, is the ffth Tere have been several eforts to perform large-scale most common cancer and the third leading cause of can- molecular profling and classifcation of GC. -
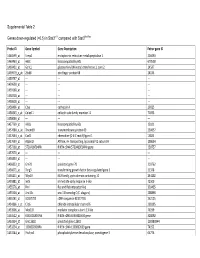
0.5) in Stat3∆/∆ Compared with Stat3flox/Flox
Supplemental Table 2 Genes down-regulated (<0.5) in Stat3∆/∆ compared with Stat3flox/flox Probe ID Gene Symbol Gene Description Entrez gene ID 1460599_at Ermp1 endoplasmic reticulum metallopeptidase 1 226090 1460463_at H60c histocompatibility 60c 670558 1460431_at Gcnt1 glucosaminyl (N-acetyl) transferase 1, core 2 14537 1459979_x_at Zfp68 zinc finger protein 68 24135 1459747_at --- --- --- 1459608_at --- --- --- 1459168_at --- --- --- 1458718_at --- --- --- 1458618_at --- --- --- 1458466_at Ctsa cathepsin A 19025 1458345_s_at Colec11 collectin sub-family member 11 71693 1458046_at --- --- --- 1457769_at H60a histocompatibility 60a 15101 1457680_a_at Tmem69 transmembrane protein 69 230657 1457644_s_at Cxcl1 chemokine (C-X-C motif) ligand 1 14825 1457639_at Atp6v1h ATPase, H+ transporting, lysosomal V1 subunit H 108664 1457260_at 5730409E04Rik RIKEN cDNA 5730409E04Rik gene 230757 1457070_at --- --- --- 1456893_at --- --- --- 1456823_at Gm70 predicted gene 70 210762 1456671_at Tbrg3 transforming growth factor beta regulated gene 3 21378 1456211_at Nlrp10 NLR family, pyrin domain containing 10 244202 1455881_at Ier5l immediate early response 5-like 72500 1455576_at Rinl Ras and Rab interactor-like 320435 1455304_at Unc13c unc-13 homolog C (C. elegans) 208898 1455241_at BC037703 cDNA sequence BC037703 242125 1454866_s_at Clic6 chloride intracellular channel 6 209195 1453906_at Med13l mediator complex subunit 13-like 76199 1453522_at 6530401N04Rik RIKEN cDNA 6530401N04 gene 328092 1453354_at Gm11602 predicted gene 11602 100380944 1453234_at -

4-6 Weeks Old Female C57BL/6 Mice Obtained from Jackson Labs Were Used for Cell Isolation
Methods Mice: 4-6 weeks old female C57BL/6 mice obtained from Jackson labs were used for cell isolation. Female Foxp3-IRES-GFP reporter mice (1), backcrossed to B6/C57 background for 10 generations, were used for the isolation of naïve CD4 and naïve CD8 cells for the RNAseq experiments. The mice were housed in pathogen-free animal facility in the La Jolla Institute for Allergy and Immunology and were used according to protocols approved by the Institutional Animal Care and use Committee. Preparation of cells: Subsets of thymocytes were isolated by cell sorting as previously described (2), after cell surface staining using CD4 (GK1.5), CD8 (53-6.7), CD3ε (145- 2C11), CD24 (M1/69) (all from Biolegend). DP cells: CD4+CD8 int/hi; CD4 SP cells: CD4CD3 hi, CD24 int/lo; CD8 SP cells: CD8 int/hi CD4 CD3 hi, CD24 int/lo (Fig S2). Peripheral subsets were isolated after pooling spleen and lymph nodes. T cells were enriched by negative isolation using Dynabeads (Dynabeads untouched mouse T cells, 11413D, Invitrogen). After surface staining for CD4 (GK1.5), CD8 (53-6.7), CD62L (MEL-14), CD25 (PC61) and CD44 (IM7), naïve CD4+CD62L hiCD25-CD44lo and naïve CD8+CD62L hiCD25-CD44lo were obtained by sorting (BD FACS Aria). Additionally, for the RNAseq experiments, CD4 and CD8 naïve cells were isolated by sorting T cells from the Foxp3- IRES-GFP mice: CD4+CD62LhiCD25–CD44lo GFP(FOXP3)– and CD8+CD62LhiCD25– CD44lo GFP(FOXP3)– (antibodies were from Biolegend). In some cases, naïve CD4 cells were cultured in vitro under Th1 or Th2 polarizing conditions (3, 4). -

Downloaded from USCS Tables (
bioRxiv preprint doi: https://doi.org/10.1101/318329; this version posted February 4, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. The selection arena in early human blastocysts resolves the pluripotent inner cell mass Manvendra Singh1, Thomas J. Widmann2, Vikas Bansal7, Jose L. Cortes2, Gerald G. Schumann3, Stephanie Wunderlich4, Ulrich Martin4, Marta Garcia-Canadas2, Jose L. Garcia- Perez2,5*, Laurence D. Hurst6*#, Zsuzsanna Izsvák1* 1 Max-Delbrück-Center for Molecular Medicine in the Helmholtz Society, Robert-Rössle- Strasse 10, 13125 Berlin, Germany. 2 GENYO. Centre for Genomics and Oncological Research: Pfizer/University of Granada/Andalusian Regional Government, PTS Granada, 18016 Granada, Spain. 3 Paul-Ehrlich-Institute, Division of Medical Biotechnology, Paul-Ehrlich-Strasse 51-59, 63225 Langen, Germany. 4Center for Regenerative Medicine Hannover Medical School (MHH) Carl-Neuberg-Str.1, Building J11, D-30625 Hannover, Germany 5Institute of Genetics and Molecular Medicine (IGMM), University of Edinburgh, Crewe Road, Edinburgh EH4 2XU, United Kingdom 6 The Milner Centre for Evolution, Department of Biology and Biochemistry, University of Bath, Bath, Somerset, UK, BA2 7AY. 7 Institute of Medical Systems Biology, Center for Molecular Neurobiology (ZMNH), University Medical Center Hamburg-Eppendorf (UKE), 20246, Hamburg, Germany. *Corresponding authors *Zsuzsanna Izsvák Max Delbrück Center for Molecular Medicine Robert Rössle Strasse 10, 13092 Berlin, Germany Telefon: +49 030-9406-3510 Fax: +49 030-9406-2547 email: [email protected] http://www.mdcberlin.de/en/research/research_teams/mobile_dna/index.html and *Laurence D. -

UNIVERSITY of CALIFORNIA RIVERSIDE Investigations Into The
UNIVERSITY OF CALIFORNIA RIVERSIDE Investigations into the Role of TAF1-mediated Phosphorylation in Gene Regulation A Dissertation submitted in partial satisfaction of the requirements for the degree of Doctor of Philosophy in Cell, Molecular and Developmental Biology by Brian James Gadd December 2012 Dissertation Committee: Dr. Xuan Liu, Chairperson Dr. Frank Sauer Dr. Frances M. Sladek Copyright by Brian James Gadd 2012 The Dissertation of Brian James Gadd is approved Committee Chairperson University of California, Riverside Acknowledgments I am thankful to Dr. Liu for her patience and support over the last eight years. I am deeply indebted to my committee members, Dr. Frank Sauer and Dr. Frances Sladek for the insightful comments on my research and this dissertation. Thanks goes out to CMDB, especially Dr. Bachant, Dr. Springer and Kathy Redd for their support. Thanks to all the members of the Liu lab both past and present. A very special thanks to the members of the Sauer lab, including Silvia, Stephane, David, Matt, Stephen, Ninuo, Toby, Josh, Alice, Alex and Flora. You have made all the years here fly by and made them so enjoyable. From the Sladek lab I want to thank Eugene, John, Linh and Karthi. Special thanks go out to all the friends I’ve made over the years here. Chris, Amber, Stephane and David, thank you so much for feeding me, encouraging me and keeping me sane. Thanks to the brothers for all your encouragement and prayers. To any I haven’t mentioned by name, I promise I haven’t forgotten all you’ve done for me during my graduate years. -

Peroxisomal Functions in the Lung and Their Role in the Pathogenesis of Lung Diseases
Aus dem Institut für Anatomie und Zellbiologie der Justus-Liebig-Universität Gießen Leiter: Prof. Dr. Eveline Baumgart-Vogt Peroxisomal functions in the lung and their role in the pathogenesis of lung diseases Habilitationsschrift zur Erlangung der Venia legendi des Fachbereichs Medizin der Justus-Liebig-Universität Gießen vorgelegt von Srikanth Karnati Gießen 2018 Die nachfolgende Arbeit nimmt Bezug auf folgende Originalarbeiten: 1. Karnati S*, Graulich T, Oruqaj G, Pfreimer S, Seimetz M, Stamme C, Mariani TJ, Weissmann N, Mühlfeld C, Baumgart-Vogt E (2016). Postnatal development of the secretory cells of the distal airways, the bronchiolar club cells in the mouse lung: stereological and molecular biological studies. Cell and Tissue Research. Jun;364(3):543- 57. 2. Karnati S, Baumgart-Vogt E (2009) PeroXisomes in airway epithelia and future prospects of these organelles for pulmonary cell biology. Histochem Cell Biol. Apr: 131(4):447-54. 3. Karnati S, Lüers G, Pfreimer S and Baumgart-Vogt E (2013) Manganese SuperoXide dismutase 2 (MnSOD) is localized to mitochondria but not in peroXisomes. Histochemistry and Cell Biology, Aug:140(2):105-17 4. Karnati S, Palaniswamy S, Alam MR, Oruqaj G, Stamme C, Baumgart-Vogt E (2015) C22- bronchial and T7-alveolar epithelial cell lines of the immortomouse are eXcellent murine cell culture model systems to study pulmonary peroXisome biology and metabolism. Histochemistry and Cell Biology Mar;145(3):287-304. 5. Oruqaj G§, Karnati S§, Vijayan V, Kotarkonda LK, Boateng E, Zhang W, Ruppert C, Günther A, Shi W, Baumgart-Vogt E (2015) Compromised peroXisomes in idiopathic pulmonary fibrosis, a vicious cycle inducing a higher fibrotic response via TGF-β signaling. -

Modifications to the Harmonized Tariff Schedule of the United States To
U.S. International Trade Commission COMMISSIONERS Shara L. Aranoff, Chairman Daniel R. Pearson, Vice Chairman Deanna Tanner Okun Charlotte R. Lane Irving A. Williamson Dean A. Pinkert Address all communications to Secretary to the Commission United States International Trade Commission Washington, DC 20436 U.S. International Trade Commission Washington, DC 20436 www.usitc.gov Modifications to the Harmonized Tariff Schedule of the United States to Implement the Dominican Republic- Central America-United States Free Trade Agreement With Respect to Costa Rica Publication 4038 December 2008 (This page is intentionally blank) Pursuant to the letter of request from the United States Trade Representative of December 18, 2008, set forth in the Appendix hereto, and pursuant to section 1207(a) of the Omnibus Trade and Competitiveness Act, the Commission is publishing the following modifications to the Harmonized Tariff Schedule of the United States (HTS) to implement the Dominican Republic- Central America-United States Free Trade Agreement, as approved in the Dominican Republic-Central America- United States Free Trade Agreement Implementation Act, with respect to Costa Rica. (This page is intentionally blank) Annex I Effective with respect to goods that are entered, or withdrawn from warehouse for consumption, on or after January 1, 2009, the Harmonized Tariff Schedule of the United States (HTS) is modified as provided herein, with bracketed matter included to assist in the understanding of proclaimed modifications. The following supersedes matter now in the HTS. (1). General note 4 is modified as follows: (a). by deleting from subdivision (a) the following country from the enumeration of independent beneficiary developing countries: Costa Rica (b). -

Supplement 1A Steffensen Et
Liver Wild-type Knockout C T C T 1 2 4 7 8 9 1 2 4 5 7 9 1 1 1 1 1 1 C C C T T T C C C T T T W W W W W W K K K K K K IMAGE:793166 RIKEN cDNA 6720463E02 gene IMAGE:1447421 ESTs, Weakly similar to ZF37 MOUSE ZINC FINGER PROTEIN 37 [M.musculus] IMAGE:934291 RIKEN cDNA 2810418N01 gene IMAGE:1247525 small EDRK-rich f2actor IMAGE:1449402 expressed sequence AW321064 IMAGE:1279847 ESTs IMAGE:518737 expressed sequence AW049941 IMAGE:860231 a disintegrin and metalloproteinase domain 17 IMAGE:642836 CD86 antigen IMAGE:1003885 phosphoribosyl pyrophosphate sy1nthetase IMAGE:524862 RIKEN cDNA 5730469D23 gene IMAGE:1264473 protein inhibitor of activat1ed STAT IMAGE:847035 RIKEN cDNA 4833422F06 gene IMAGE:374550 requiem IMAGE:976520 nuclear receptor coact4ivator IMAGE:1264311 Unknown IMAGE:976735 expressed sequence AI987692 IMAGE:976659 cathepsLin IMAGE:1477580 RIKEN cDNA 1600010J02 gene IMAGE:1277168 ribosomal protein, large, P1 IMAGE:524842 RIKEN cDNA 0710008D09 gene IMAGE:373019 split hand/foot delete1d gene IMAGE:404428 expressed sequence AI413851 IMAGE:619810 RIKEN cDNA 1700003F10 gene IMAGE:1749558 caspase 3, apoptosis related cysteine protease IMAGE:718718 RIKEN cDNA 2810003F23 gene IMAGE:819789 Unknown IMAGE:524474 ATP-binding cassette, sub-family A ABC1, member IMAGE:804950 Mus musculus, Similar to ribosomal protein S20, clone MGC:6876 IMAGE:2651405, mRNA, complete cds IMAGE:806143 gap junction membrane channel prot2ein beta IMAGE:1745887 expressed sequence AI836376 IMAGE:779426 RIKEN cDNA 5230400G24 gene IMAGE:1125615 Unknown IMAGE:535025 DNA -

One Target, Different Effects: a Comparison of Distinct Therapeutic Antibodies Against the Same Targets
EXPERIMENTAL and MOLECULAR MEDICINE, Vol. 43, No. 10, 539-549, October 2011 One target, different effects: a comparison of distinct therapeutic antibodies against the same targets Hyunbo Shim ple, four antibodies against TNF-α have been approved by the FDA -- infliximab, adalimumab, golimumab, and Department of Life Science certolizumab pegol -- with many more in clinical and Division of Life and Pharmaceutical Sciences preclinical development. The situation is similar for Ewha Womans University HER2, CD20, EGFR, and VEGF, each having one or Seoul 120-750, Korea more approved antibodies and many more under Correspondence: Tel, 82-2-3277-4240; development. This review discusses the different bind- Fax, 82-2-3277-3760; E-mail, [email protected] ing characteristics, mechanisms of action, and bio- http://dx.doi.org/10.3858/emm.2011.43.10.063 logical and clinical activities of multiple monoclonal antibodies against TNF-α, HER-2, CD20, and EGFR and Accepted 2 August 2011 provides insights into the development of therapeutic Available Online 3 August 2011 antibodies. Abbreviations: ADC, antibody-drug conjugate; ADCC, antibody- dependent cellular cytotoxicity; CD20, cluster of differentiation Keywords: antibodies, monoclonal; antigens, CD20; 20; CDC, complement dependent cytotoxicity; CLL, chronic pharmacology; receptor, epidermal growth factor; re- lymphocytic leukemia; ECD, extracellular domain; EGFR, epi- ceptor, erbB-2; tumor necrosis factor-α dermal growth factor receptor; EpCAM, epithelial cell adhe- sion molecule; FcγR, Fc gamma receptor;