TITLE 77 PUBLIC HEALTH Chapter: I Department of Public Health Sub Chapter: a General Rules Part #: 100 Rules of Practice and Procedure in Administrative Hearings
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Table 2. 2012 AGS Beers Criteria for Potentially
Table 2. 2012 AGS Beers Criteria for Potentially Inappropriate Medication Use in Older Adults Strength of Organ System/ Recommendat Quality of Recomm Therapeutic Category/Drug(s) Rationale ion Evidence endation References Anticholinergics (excludes TCAs) First-generation antihistamines Highly anticholinergic; Avoid Hydroxyzin Strong Agostini 2001 (as single agent or as part of clearance reduced with e and Boustani 2007 combination products) advanced age, and promethazi Guaiana 2010 Brompheniramine tolerance develops ne: high; Han 2001 Carbinoxamine when used as hypnotic; All others: Rudolph 2008 Chlorpheniramine increased risk of moderate Clemastine confusion, dry mouth, Cyproheptadine constipation, and other Dexbrompheniramine anticholinergic Dexchlorpheniramine effects/toxicity. Diphenhydramine (oral) Doxylamine Use of diphenhydramine in Hydroxyzine special situations such Promethazine as acute treatment of Triprolidine severe allergic reaction may be appropriate. Antiparkinson agents Not recommended for Avoid Moderate Strong Rudolph 2008 Benztropine (oral) prevention of Trihexyphenidyl extrapyramidal symptoms with antipsychotics; more effective agents available for treatment of Parkinson disease. Antispasmodics Highly anticholinergic, Avoid Moderate Strong Lechevallier- Belladonna alkaloids uncertain except in Michel 2005 Clidinium-chlordiazepoxide effectiveness. short-term Rudolph 2008 Dicyclomine palliative Hyoscyamine care to Propantheline decrease Scopolamine oral secretions. Antithrombotics Dipyridamole, oral short-acting* May -

MSM Cross Reference Antihistamine Decongestant 20100701 Final Posted
MISSISSIPPI DIVISION OF MEDICAID Antihistamine/Decongestant Product and Active Ingredient Cross-Reference List The agents listed below are the antihistamine/decongestant drug products listed in the Mississippi Medicaid Preferred Drug List (PDL). This is a cross-reference between the drug product name and its active ingredients to reference the antihistamine/decongestant portion of the PDL. For more information concerning the PDL, including non- preferred agents, the OTC formulary, and other specifics, please visit our website at www.medicaid.ms.gov. List Effective 07/16/10 Therapeutic Class Active Ingredients Preferred Non-Preferred ANTIHISTAMINES - 1ST GENERATION BROMPHENIRAMINE MALEATE BPM BROMAX BROMPHENIRAMINE MALEATE J-TAN PD BROMSPIRO LODRANE 24 LOHIST 12HR VAZOL BROMPHENIRAMINE TANNATE BROMPHENIRAMINE TANNATE J-TAN P-TEX BROMPHENIRAMINE/DIPHENHYDRAM ALA-HIST CARBINOXAMINE MALEATE CARBINOXAMINE MALEATE PALGIC CHLORPHENIRAMINE MALEATE CHLORPHENIRAMINE MALEATE CPM 12 CHLORPHENIRAMINE TANNATE ED CHLORPED ED-CHLOR-TAN MYCI CHLOR-TAN MYCI CHLORPED PEDIAPHYL TANAHIST-PD CLEMASTINE FUMARATE CLEMASTINE FUMARATE CYPROHEPTADINE HCL CYPROHEPTADINE HCL DEXCHLORPHENIRAMINE MALEATE DEXCHLORPHENIRAMINE MALEATE DIPHENHYDRAMINE HCL ALLERGY MEDICINE ALLERGY RELIEF BANOPHEN BENADRYL BENADRYL ALLERGY CHILDREN'S ALLERGY CHILDREN'S COLD & ALLERGY COMPLETE ALLERGY DIPHEDRYL DIPHENDRYL DIPHENHIST DIPHENHYDRAMINE HCL DYTUSS GENAHIST HYDRAMINE MEDI-PHEDRYL PHARBEDRYL Q-DRYL QUENALIN SILADRYL SILPHEN DIPHENHYDRAMINE TANNATE DIPHENMAX DOXYLAMINE SUCCINATE -

2-Bromopyridine Safety Data Sheet Jubilant Ingrevia Limited
2-Bromopyridine Safety Data Sheet According to the federal final rule of hazard communication revised on 2012 (HazCom 2012) Date of Compilation : July 03 ’ 2019 Date of Revision : February 09 ’ 2021 Revision due date : January 2024 Revision Number : 01 Version Name : 0034Gj Ghs01 Div.3 sds 2-Bromopyridine Supersedes date : July 03 ’ 2019 Supersedes version : 0034Gj Ghs00 Div.3 sds 2-Bromopyridine Jubilant Ingrevia Limited Page 1 of 9 2-Bromopyridine Safety Data Sheet According to the federal final rule of hazard communication revised on 2012 (HazCom 2012) SECTION 1: IDENTIFICATION OF THE SUBSTANCE/MIXTURE AND OF THE COMPANY/UNDERTAKING 1.1. Product identifier PRODUCT NAME : 2-Bromopyridine CAS RN : 109-04-6 EC# : 203-641-6 SYNONYMS : 2-Pyridyl bromide, Pyridine, 2-bromo-, beta-Bromopyridine, o-Bromopyridine SYSTEMATIC NAME : 2-Bromopyridine, -Pyridine, 2-bromo- MOLECULAR FORMULA : C5H4BrN STRUCTURAL FORMULA N Br 1.2. Relevant identified uses of the substance or mixture and uses advised against 1.2.1. Relevant identified uses 2-Bromopyridine is used as an intermediate in the pharmaceutical industry for the manufacture of Atazanavir (an antiretroviral drug), Carbinoxamine, Chloropyramine, triprolidine (antihistamine drugs), Disopyramide Phosphate (an antiarrythmic drug), Mefloquine (antimalarial drug), Pipradrol (mild CNS stimulant) etc. Uses advised against: None 1.3. Details of the supplier of the safety data sheet Jubilant Ingrevia Limited REGISTERED & FACTORY OFFICE: Jubilant Ingrevia Limited Bhartiagram, Gajraula , District: Amroha, Uttar Pradesh-244223, India PHONE NO: +91-5924-252353 to 252360 Contact Department-Safety: Ext. 7424 , FAX NO : +91-5924-252352 HEAD OFFICE: Jubilant Ingrevia Limited, Plot 1-A, Sector 16-A,Institutional Area, Noida, Uttar Pradesh, 201301 - India T +91-120-4361000 - F +91-120-4234881 / 84 / 85 / 87 / 95 / 96 [email protected] -www.jubilantingrevia.com 1.4. -

Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017
Q UO N T FA R U T A F E BERMUDA PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 BR 111 / 2017 The Minister responsible for health, in exercise of the power conferred by section 48A(1) of the Pharmacy and Poisons Act 1979, makes the following Order: Citation 1 This Order may be cited as the Pharmacy and Poisons (Third and Fourth Schedule Amendment) Order 2017. Repeals and replaces the Third and Fourth Schedule of the Pharmacy and Poisons Act 1979 2 The Third and Fourth Schedules to the Pharmacy and Poisons Act 1979 are repealed and replaced with— “THIRD SCHEDULE (Sections 25(6); 27(1))) DRUGS OBTAINABLE ONLY ON PRESCRIPTION EXCEPT WHERE SPECIFIED IN THE FOURTH SCHEDULE (PART I AND PART II) Note: The following annotations used in this Schedule have the following meanings: md (maximum dose) i.e. the maximum quantity of the substance contained in the amount of a medicinal product which is recommended to be taken or administered at any one time. 1 PHARMACY AND POISONS (THIRD AND FOURTH SCHEDULE AMENDMENT) ORDER 2017 mdd (maximum daily dose) i.e. the maximum quantity of the substance that is contained in the amount of a medicinal product which is recommended to be taken or administered in any period of 24 hours. mg milligram ms (maximum strength) i.e. either or, if so specified, both of the following: (a) the maximum quantity of the substance by weight or volume that is contained in the dosage unit of a medicinal product; or (b) the maximum percentage of the substance contained in a medicinal product calculated in terms of w/w, w/v, v/w, or v/v, as appropriate. -

Letter Bill 1..143
Public Act 097-0334 HB2917 Enrolled LRB097 06471 RLC 50343 b AN ACT concerning controlled substances. Be it enacted by the People of the State of Illinois, represented in the General Assembly: Section 5. The Illinois Controlled Substances Act is amended by changing Sections 100, 102, 201, 202, 203, 204, 205, 206, 207, 208, 209, 210, 211, 212, 301, 302, 303, 303.05, 303.1, 304, 305, 306, 309, 312, 313, 316, 317, 318, 319, 320, 405, 405.1, 406, 408, 410, 411.2, 413, 501, 501.1, 503, 504, 505, 507, and 510 and by adding Sections 311.5, 314.5, and 507.2 as follows: (720 ILCS 570/100) (from Ch. 56 1/2, par. 1100) Sec. 100. Legislative intent. It is the intent of the General Assembly, recognizing the rising incidence in the abuse of drugs and other dangerous substances and its resultant damage to the peace, health, and welfare of the citizens of Illinois, to provide a system of control over the distribution and use of controlled substances which will more effectively: (1) limit access of such substances only to those persons who have demonstrated an appropriate sense of responsibility and have a lawful and legitimate reason to possess them; (2) deter the unlawful and destructive abuse of controlled substances; (3) penalize most heavily the illicit traffickers or profiteers of controlled substances, who propagate and perpetuate the Public Act 097-0334 HB2917 Enrolled LRB097 06471 RLC 50343 b abuse of such substances with reckless disregard for its consumptive consequences upon every element of society; (4) acknowledge the functional and consequential differences between the various types of controlled substances and provide for correspondingly different degrees of control over each of the various types; (5) unify where feasible and codify the efforts of this State to conform with the regulatory systems of the Federal government and other states to establish national coordination of efforts to control the abuse of controlled substances; and (6) provide law enforcement authorities with the necessary resources to make this system efficacious. -

Medicines Classification Committee
Medicines Classification Committee Meeting date 1 May 2017 58th Meeting Title Reclassification of Sedating Antihistamines Medsafe Pharmacovigilance Submitted by Paper type For decision Team Proposal for The Medicines Adverse Reactions Committee (MARC) recommended that the reclassification to committee consider reclassifying sedating antihistamines to prescription prescription medicines when used in children under 6 years of age for the treatment of medicine for some nausea and vomiting and travel sickness [exact wording to be determined by indications the committee]. Reason for The purpose of this document is to provide the committee with an overview submission of the information provided to the MARC about safety concerns associated with sedating antihistamines and reasons for recommendations. Associated March 2013 Children and Sedating Antihistamines Prescriber Update articles February 2010 Cough and cold medicines clarification – antihistamines Medsafe website Safety information: Use of cough and cold medicines in children – new advice Medicines for Alimemazine Diphenhydramine consideration Brompheniramine Doxylamine Chlorpheniramine Meclozine Cyclizine Promethazine Dexchlorpheniramine New Zealand Some oral sedating antihistamines available without exposure to a prescription (pharmacist-only and pharmacy only), sedating therefore usage data is not easily available. antihistamines Table of Contents 1.0 PURPOSE ...................................................................................................................................... -

Letter Bill 0..70
HB2534 *LRB10008419RLC18533b* 100TH GENERAL ASSEMBLY State of Illinois 2017 and 2018 HB2534 by Rep. Avery Bourne SYNOPSIS AS INTRODUCED: 720 ILCS 570/102 from Ch. 56 1/2, par. 1102 720 ILCS 570/204 from Ch. 56 1/2, par. 1204 720 ILCS 570/401 from Ch. 56 1/2, par. 1401 720 ILCS 570/402 from Ch. 56 1/2, par. 1402 Amends the Illinois Controlled Substances Act. Requires that to be illegal a drug analog must not be approved by the United States Food and Drug Administration or, if approved, it is not dispensed or possessed in accordance with State and federal law. Defines "controlled substance" to include a synthetic drug enumerated as a scheduled drug under the Act. Adds chemical structural classes of synthetic cannabinoids and piperazines to the list of Schedule I controlled substances. Includes certain substances approved by the FDA which are not dispensed or possessed in accordance with State or federal law and certain modified substances. LRB100 08419 RLC 18533 b CORRECTIONAL BUDGET AND IMPACT NOTE ACT MAY APPLY A BILL FOR HB2534 LRB100 08419 RLC 18533 b 1 AN ACT concerning criminal law. 2 Be it enacted by the People of the State of Illinois, 3 represented in the General Assembly: 4 Section 5. The Illinois Controlled Substances Act is 5 amended by changing Sections 102, 204, 401, and 402 as follows: 6 (720 ILCS 570/102) (from Ch. 56 1/2, par. 1102) 7 Sec. 102. Definitions. As used in this Act, unless the 8 context otherwise requires: 9 (a) "Addict" means any person who habitually uses any drug, 10 chemical, substance or dangerous drug other than alcohol so as 11 to endanger the public morals, health, safety or welfare or who 12 is so far addicted to the use of a dangerous drug or controlled 13 substance other than alcohol as to have lost the power of self 14 control with reference to his or her addiction. -
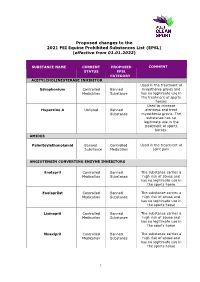
Proposed Changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (Effective from 01.01.2022)
Proposed changes to the 2021 FEI Equine Prohibited Substances List (EPSL) (effective from 01.01.2022) SUBSTANCE NAME CURRENT PROPOSED COMMENT STATUS EPSL CATEGORY ACETYLCHOLINESTERASE INHIBITOR Used in the treatment of Edrophonium Controlled Banned myasthenia gravis and Medication Substance has no legitimate use in the treatment of sports horses Used to increase Huperzine A Unlisted Banned alertness and treat Substance myasthenia gravis. The substance has no legitimate use in the treatment of sports horses. AMIDES Palmitoylethanolamid Banned Controlled Used in the treatment of Substance Medication joint pain ANGIOTENSIN CONVERTING ENZYME INHIBITORS Enalapril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Enalaprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Lisinopril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse Moexipril Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse 1 Perindoprilat Controlled Banned The substance carries a Medication Substance high risk of abuse and has no legitimate use in the sports horse ANTIHISTAMINES Antazoline Controlled Banned The substance has no Medication Substance legitimate use in the sports horse Azatadine Controlled Banned The substance has Medication Substance sedative effects -
![With [3H]Mepyramine (Trieyclic Antidepressants/Antihistamine/Neurotransmitter/Amitriptyline) VINH TAN TRAN, RAYMOND S](https://docslib.b-cdn.net/cover/2862/with-3h-mepyramine-trieyclic-antidepressants-antihistamine-neurotransmitter-amitriptyline-vinh-tan-tran-raymond-s-1512862.webp)
With [3H]Mepyramine (Trieyclic Antidepressants/Antihistamine/Neurotransmitter/Amitriptyline) VINH TAN TRAN, RAYMOND S
Proc. Nati. Acad. Sci. USA Vol. 75, No. 12, pp. 6290-6294,, December 1978 Neurobiology Histamine H1 receptors identified in mammalian brain membranes with [3H]mepyramine (trieyclic antidepressants/antihistamine/neurotransmitter/amitriptyline) VINH TAN TRAN, RAYMOND S. L. CHANG, AND SOLOMON H. SNYDER* Departments of Pharmacology and Experimental Therapeutics, and Psychiatry and Behavioral Sciences, Johns Hopkins University School of Medicine, Baltimore, Maryland 21205 Communicated by Julius Axelrod, August 30,1978 ABSTRACT The antihistamine [3H mepyramine binds to Male Sprague-Dawley rats (150-200 g) were killed by cer- HI histamine receptors in mammalian brain membranes. vical dislocation, their brains were rapidly removed and ho- Potencies of H1 antihistamines at the binding sites correlate mogenized with a Polytron for 30 min (setting 5) in 30 vol of with their pharmacological antihistamine effects in the guinea pig ileum. Specific [3Himepyramine binding is saturable with ice-cold Na/K phosphate buffer (50 mM, pH 7.5), and the a dissociation constant of about 4 nM in both equilibrium and suspension was centrifuged (50,000 X g for 10 min). The pellet kinetic experiments and a density of 10pmolper gram ofwhole was resuspended in the same volume of fresh buffer and cen- brain. Some tricyclic antidepressants are potent inhibitors of trifuged, and the final pellet was resuspended in the original secific [3Hmepamine binding. Regional variations of volume of ice-cold buffer by Polytron homogenization. Calf [3Hjmepyramine ing do not correlate with variations in brains were obtained from a local abattoir within 2 hr after the endogeneous histamine and histidine decarboxylase activity. death of the animals and transferred to the laboratory in ice- Histamine is a neurotransmitter candidate in mammalian brain cold saline. -

LITERATURE REVIEW Drug Review
LITERATURE REVIEW Drug Review Document ID: N04-023 Author: Julie Qidwai Date: December 2004 National Advanced Driving Simulator 2401 Oakdale Blvd. Iowa City, IA 52242-5003 Fax (319) 335-4658 TABLE OF CONTENTS 1 Introduction ............................................................................................................................ 1 2 Alprazolam.............................................................................................................................. 1 3 Amitriptyline........................................................................................................................... 2 4 Biperiden................................................................................................................................. 2 5 Brompheniramine .................................................................................................................. 2 6 Butorphanol ............................................................................................................................ 2 7 Cetirizine................................................................................................................................. 3 8 Chloroquine ............................................................................................................................ 3 9 Chlorpheniramine .................................................................................................................. 4 10 Clemastine.............................................................................................................................. -

A Unique Serotonin Receptor in Choroid Plexus Is Linked To
Proc. Natl. Acad. Sci. USA Vol. 83, pp. 4086-4088, June 1986 Neurobiology A unique serotonin receptor in choroid plexus is linked to phosphatidylinositol turnover (serotonin 5-HTjc site/serotonin 5-HT2 site/serotonin antagonists/cerebrospinal fluid/phosphoinositide hydrolysis) P. JEFFREY CONN*, ELAINE SANDERS-BUSH*t, BETH J. HOFFMANt, AND PAUL R. HARTIGt *Tennessee Neuropsychiatric Institute and Department of Pharmacology, Vanderbilt University School of Medicine, Nashville, TN 37232; and tDepartment of Biology, Johns Hopkins University, Baltimore, MD 21218 Communicated by Saul Roseman, January 23, 1986 ABSTRACT A novel serotonergic binding site, the 5-HTlc (11). These findings have been confirmed and extended in site, has been characterized recently in choroid plexus and brain (12-14) and other tissues (15-17). Serotonin-stimulated several brain regions. The biochemical and physiological roles phosphatidylinositol turnover in cerebral cortex is not sec- ofthis site have not been previously described. In this report we ondary to release of another neurotransmitter or of arachi- show that serotonin (5-hydroxytryptamine, 5-HT) stimulates donic acid (18), suggesting that the 5-HT2 receptor is directly phosphatidylinositol turnover in rat choroid plexus. The phar- coupled to phosphatidylinositol turnover. In the present macology of serotonin-stimulated phosphatidylinositol hydrol- studies, we have examined the effect of serotonin and ysis in choroid plexus was compared to the pharmacology in serotonin antagonists upon phosphatidylinositol turnover in cerebral cortex, where this response is mediated by the rat choroid plexus. The pharmacology of the response in serotonin 5-HT2 receptor. Serotonin increased phosphatidyl- choroid plexus was compared with that in cerebral cortex and inositol turnover in choroid plexus by 6-fold and in cerebral with the pharmacology ofthe 5-HT1c and 5-HT2 binding sites. -

(+)Lysergic Acid Diethylamide, but Not Its Nonhallucinogenic Congeners, Is a Potent Serotonin 5HT� Receptor Agonist1
0022.3565/91/2583-0891$03.OO/O THE JOURNAL OF PHARMACOLOGY AND EXPERIMENTAL ThERAPEUTICS Vol. 258, No. 3 Copyright © 1991 by The American Society for PharmacoIogj and Experimental Therapeutics Printed in U.S.A. (+)Lysergic Acid Diethylamide, but not Its Nonhallucinogenic Congeners, Is a Potent Serotonin 5HT Receptor Agonist1 KEVIN D. BURRIS, MARSHA BREEDING and ELAINE SANDERS-BUSH Department of Pharmacology, Vanderbilt University School of Medicine, Nashville, Tennessee Accepted for publication May 20, 1991 ABSTRACT Activation of central serotonin 5HT2 receptors is believed to be epithelial cells, with ECro values of 9 and 26 nM, respectively. the primary mechanism whereby lysergic acid diethylamide(LSD) The effect of (+)LSD in both systems was blocked by 5HT and other hallucinogens induce psychoactive effects. This hy- receptor antagonists with an order of activity consistent with pothesis is based on extensive radioligand binding and electro- interaction at 5HT1 receptors. Neither (+)-2-bromo-LSD nor physiological and behavioral studies in laboratory animals. How- lisunde, two nonhallucinogenic congeners of LSD, were able to ever, the pharmacological profiles of 5HT2 and 5HT1 receptors stimulate 5HT1 receptors in cultured cells or intact choroid are similar, making it difficult to distinguish between effects due plexus. In contrast, lisunde, like (+)LSD, is a partial agonist at to activation of one or the other receptor. For this reason, it was 5HT2 receptors in cerebral cortex slices and in NIH 3T3 cells of interest to investigate the interaction of LSD with 5HT1 transfected with 5HT2 receptor cDNA. The present finding that receptors. Agonist-stimulated phosphoinositide hydrolysis in rat (+)LSD, but not its nonhallucinogenic congeners, is a 5HT1 choroid plexus was used as a direct measure of 5HT1 receptor receptor agonist suggests a possible role for these receptors in activation.