Natural Organic Matter in Drinking Water Sources: Its Characterisation and Treatability
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Total Organic Carbon (TOC) Guidance Manual
September 2002 RG-379 (Revised) Total Organic Carbon (TOC) Guidance Manual Water Supply Division printed on recycled paper TEXAS COMMISSION ON ENVIRONMENTAL QUALITY Total Organic Carbon (TOC) Guidance Manual Prepared by Water Supply Division RG-379 (Revised) September 2002 Robert J. Huston, Chairman R. B. “Ralph” Marquez, Commissioner Kathleen Hartnett White, Commissioner Jeffrey A. Saitas, Executive Director Authorization to use or reproduce any original material contained in this publication—that is, not obtained from other sources—is freely granted. The commission would appreciate acknowledgment. Copies of this publication are available for public use through the Texas State Library, other state depository libraries, and the TCEQ Library, in compli- ance with state depository law. For more information on TCEQ publications call 512/239-0028 or visit our Web site at: www.tceq.state.tx.us/publications Published and distributed by the Texas Commission on Environmental Quality PO Box 13087 Austin TX 78711-3087 The Texas Commission on Environmental Quality was formerly called the Texas Natural Resource Conservation Commission. The TCEQ is an equal opportunity/affirmative action employer. The agency does not allow discrimination on the basis of race, color, religion, national origin, sex, disability, age, sexual orientation or veteran status. In compliance with the Americans with Disabilities Act, this document may be requested in alternate formats by contacting the TCEQ at 512/239-0028, Fax 239-4488, or 1-800-RELAY-TX (TDD), or by writing -

Recommended Standards for Water Works 2007 Edition
Recommended Standards for Water Works 2007 Edition Policies for the Review and Approval of Plans and Specifications for Public Water Supplies A Report of the Water Supply Committee of the Great Lakes--Upper Mississippi River Board of State and Provincial Public Health and Environmental Managers MEMBER STATES AND PROVINCE Illinois Indiana Iowa Michigan Minnesota Missouri New York Ohio Ontario Pennsylvania Wisconsin Published by: Health Research Inc., Health Education Services Division, P.O. Box 7126, Albany, NY 12224 (518)439-7286 www.hes.org Copyright © 2007 by the Great Lakes - Upper Mississippi River Board of State and Provincial Public Health and Environmental Managers This book, or portions thereof, may be reproduced without permission from the author if proper credit is given. TABLE OF CONTENTS FOREWORD POLICY STATEMENT ON PRE-ENGINEERED WATER TREATMENT PLANTS POLICY STATEMENT ON AUTOMATED/UNATTENDED OPERATION OF SURFACE WATER TREATMENT PLANTS POLICY STATEMENT ON BAG AND CARTRIDGE FILTERS FOR PUBLIC WATER SUPPLIES POLICY STATEMENT ON ULTRA VIOLET LIGHT FOR TREATMENT OF PUBLIC WATER SUPPLIES POLICY STATEMENT ON INFRASTRUCTURE SECURITY FOR PUBLIC WATER SUPPLIES POLICY STATEMENT ON ARSENIC REMOVAL INTERIM STANDARD - NITRATE REMOVAL USING SULFATE SELECTIVE ANION EXCHANGE RESIN INTERIM STANDARD - USE OF CHLORAMINE DISINFECTANT FOR PUBLIC WATER SUPPLIES INTERIM STANDARD ON MEMBRANE TECHNOLOGIES FOR PUBLIC WATER SUPPLIES PART 1 - SUBMISSION OF PLANS 1.0 GENERAL 1.1 ENGINEER’S REPORT 1.1.1 General Information 1.1.2 Extent of water works -

Dissolved Organic Carbon (DOC) (For Private Water and Health Regulated Public Water Supplies)
Dissolved Organic Carbon (DOC) (For Private Water and Health Regulated Public Water Supplies) What Is Dissolved Organic Carbon? Dissolved organic carbon (DOC) is a general description of the organic material dissolved in water. Organic carbon occurs as the result of decomposition of plant or animal material. Organic carbon present in soil or water bodies may then dissolve when contacted by water. This dissolved organic carbon moves with both surface water and ground water. Acknowledgement: How Does Dissolved Organic Carbon Get Into Water? Organic material (including carbon) results from decomposition of plants or animals. This Fact Sheet is one of a Once this decomposed organic material contacts water it may partially dissolve. series developed by an Interagency Committee with representatives from How Does Dissolved Organic Carbon Affect My Health? Saskatchewan Ministry of DOC does not pose health risk itself but may become potentially harmful when in Health, Regional Health combination with other aspects of your water. When water with high DOC is Authorities, Saskatchewan chlorinated, harmful byproducts called trihalomethanes may be produced (see Watershed Authority, SaskH2O factsheet on trihalomethanes). Trihalomethanes may have long-term Saskatchewan Ministry of effects on health and they should be considered when chlorinating drinking water Environment, Saskatchewan Ministry of Agriculture, high in DOC. According to Health Canada, the benefits of chlorinating drinking Agriculture and Agri-Food water are much greater than the health risks associated with chlorination by- Canada – PFRA and Health products such as trihalomethanes Canada. DOC can interfere with the effectiveness of disinfection processes such as Responsibility for chlorination, ultraviolet and ozone sterilization. DOC can also promote the growth of interpretation of the content of microorganisms by providing a food source. -

Turbidity Analysis for Oregon Public Water Systems Water Quality in Coast Range Drinking Water Source Areas June 2010
Report Turbidity Analysis for Oregon Public Water Systems Water Quality in Coast Range Drinking Water Source Areas June 2010 Last Updated: 06/29/10 By: Joshua Seeds DEQ 09-WQ-024 This report prepared by: Oregon Department of Environmental Quality 811 SW 6th Avenue Portland, OR 97204 1-800-452-4011 Contact: Joshua Seeds (503) 229-5081 [email protected] Turbidity Analysis for Oregon Public Water Systems Table of Contents Table of Contents .......................................................................................... i List of Figures .............................................................................................. iii List of Tables ............................................................................................... iiv List of Maps ................................................................................................. iiv Executive Summary ..................................................................................... 1 Introduction .................................................................................................. 2 Applicable Turbidity Standards for Water Quality ............................................... 2 Public Water System Evaluations....................................................................... 3 Methods .......................................................................................................................... 5 Definitions of terms........................................................................................................ -

Publication No. 17: Ozone Treatment of Private Drinking Water Systems
PRIVATE DRINKING WATER IN CONNECTICUT Publication Date: April 2009 Publication No. 17: Ozone Treatment of Private Drinking Water Systems Effective Against: Pathogenic (disease-causing) organisms including bacteria and viruses, phenols (aromatic organic compounds), some color, taste and odor problems, iron, manganese, and turbidity. Not Effective Against: Large cysts and some other large organisms resulting from possible or probable sewage contamination, inorganic chemicals, and heavy metals. How Ozone (O3) Treatment Works Ozone is a chemical form of pure oxygen. Like chlorine, ozone is a strong oxidizing agent and is used in much the same way to kill disease-causing bacteria and viruses. It is effective against most amoebic cysts, and destroys bacteria and some aromatic organic compounds (such as phenols). Ozone may not kill large cysts and some other large organisms, so these should be eliminated by filtration or other procedures prior to ozone treatment. Ozone is effective in eliminating or controlling color, taste, and odor problems. It also oxidizes iron and manganese. Ozone treatment units are installed as point-of-use treatment systems. Raw water enters one opening and treated water emerges from another. Inside the treatment unit, ozone is produced by an electrical corona discharge or ultraviolet irradiation of dry air or oxygen. The ozone is mixed with the water whenever the water pump is running. Ozone generation units require a system to clean and remove the humidity from the air. For proper disinfection the water to be treated must have negligible color and turbidity levels. The system requires routine maintenance and an ozone treatment system can be very energy consumptive. -

Raw Water Irrigation System Master Plan
TOWN OF FIRESTONE RAW WATER IRRIGATION SYSTEM MASTER PLAN MARCH 2010 COLORADO CIVIL GROUP, INC. Raw Water Irrigation System Master Plan March 2010 Table of Contents 1 Introduction ............................................................................................................................................................................................. 1 1.1 Project Goals ................................................................................................................................................................................... 1 1.2 Scope of Work ................................................................................................................................................................................ 1 1.2.1 Existing Master Plan Evaluation ............................................................................................................... 1 1.2.2 Identifying Potential Areas for Raw Water Irrigation ............................................................................... 1 1.2.3 Irrigation Ditch Evaluation ........................................................................................................................ 1 1.2.4 Hydraulic Analysis and Calculations ......................................................................................................... 2 1.2.5 Raw Water System Modeling ................................................................................................................... 2 1.2.6 Raw Water Irrigation System Master -

21 Reverse Osmosis Treatment of PDWS 03-09
PRIVATE DRINKING WATER IN CONNECTICUT Publication Date: March 2009 Publication No. 21: Reverse Osmosis Treatment of Private Drinking Water Systems Effective Against: inorganic contaminants such as: dissolved salts of sodium, dissolved (ferrous) iron, nitrate, lead, fluoride, sulfate, potassium, manganese, aluminum, silica, chloride, total dissolved solids, chromium, and orthophosphate. Also effective in removing some detergents, some taste, color and odor-producing chemicals, certain organic contaminants, uranium, and some pesticides. Not Effective Against: dissolved gases, most volatile and semi-volatile organic contaminants including some pesticides and solvents. Alone, reverse osmosis (RO) units are not recommended for treatment of bacteria and other microscopic organisms. How Reverse Osmosis Works A complete reverse osmosis system consists of a RO module, a storage tank, and a separate faucet. The module contains a semi-permeable membrane that allows water to selectively pass through and collect in the storage tank. The contaminants being treated by the RO unit are rejected and then washed off the membrane into a waste stream. It is not practical to treat all water entering a home with an RO system because about 75 percent of the water introduced is wasted. Thus, four gallons of raw water into the system produce about one gallon of treated water. This treated water comes out much slower than water from a regular tap, so a tank is used to store the treated water. The treated water is often used only for drinking and cooking. Each manufacturer’s RO units differ, but the time needed to produce one gallon of water ranges from 2-78 hours. The volume of wastewater produced by RO systems varies by make and model. -

REVIEW of TURBIDITY: Information for Regulators and Water Suppliers
WHO/FWC/WSH/17.01 WATER QUALITY AND HEALTH - TECHNICAL BRIEF TECHNICAL REVIEW OF TURBIDITY: Information for regulators and water suppliers 1. Summary This technical brief provides information on the uses and significance of turbidity in drinking-water and is intended for regulators and operators of drinking-water supplies. Turbidity is an extremely useful indicator that can yield valuable information quickly, relatively cheaply and on an ongoing basis. Measurement of turbidity is applicable in a variety of settings, from low-resource small systems all the way through to large and sophisticated water treatment plants. Turbidity, which is caused by suspended chemical and biological particles, can have both water safety and aesthetic implications for drinking-water supplies. Turbidity itself does not always represent a direct risk to public health; however, it can indicate the presence of pathogenic microorganisms and be an effective indicator of hazardous events throughout the water supply system, from catchment to point of use. For example, high turbidity in source waters can harbour microbial pathogens, which can be attached to particles and impair disinfection; high turbidity in filtered water can indicate poor removal of pathogens; and an increase in turbidity in distribution systems can indicate sloughing of biofilms and oxide scales or ingress of contaminants through faults such as mains breaks. Turbidity can be easily, accurately and rapidly measured, and is commonly used for operational monitoring of control measures included in water safety plans (WSPs), the recommended approach to managing drinking-water quality in the WHO Guidelines for Drinking-water Quality (WHO, 2017). It can be used as a basis for choosing between alternative source waters and for assessing the performance of a number of control measures, including coagulation and clarification, filtration, disinfection and management of distribution systems. -

Guidelines for Drinking-Water Quality FIRST ADDENDUM to THIRD EDITION Volume 1 Recommendations WHO Library Cataloguing-In-Publication Data World Health Organization
Guidelines for Drinking-water Quality FIRST ADDENDUM TO THIRD EDITION Volume 1 Recommendations WHO Library Cataloguing-in-Publication Data World Health Organization. Guidelines for drinking-water quality [electronic resource] : incorporating first addendum. Vol. 1, Recommendations. – 3rd ed. Electronic version for the Web. 1.Potable water – standards. 2.Water – standards. 3.Water quality – standards. 4.Guidelines. I. Title. ISBN 92 4 154696 4 (NLM classification: WA 675) © World Health Organization 2006 All rights reserved. Publications of the World Health Organization can be obtained from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel: +41 22 791 3264; fax: +41 22 791 4857; email: [email protected]). Requests for permission to reproduce or translate WHO publications – whether for sale or for noncommercial distribution – should be addressed to WHO Press, at the above address (fax: +41 22 791 4806; email: [email protected]). The designations employed and the presentation of the material in this publication do not imply the expres- sion of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. -

2020 Water Quality Report the City of New Philadelphia Water Department
2020 WATER QUALITY REPORT THE CITY OF NEW PHILADELPHIA WATER DEPARTMENT Joel B. Day, Mayor Ron McAbier, Service Director Prepared by: 05/20/2021 Scott A. DeVault, Water Department Superintendent Scott A. DeVault Commitment to Quality The City of New Philadelphia is pleased to provide you, the water consumer, with our 2020 Water Quality Report. Results outlined in this report shows the City’s water does not contain any substances at levels that may be harmful to your health. We are proud to report that the drinking water we are supplying you with meets or exceeds all state and federal drinking water standards. As your water provider, we pride ourselves with providing our consumers safe drinking water of the highest quality available. The City of New Philadelphia has been providing water to the community for over 100 years. We are committed to furnishing the citizens of New Philadelphia quality potable water at a reasonable cost. For more information on your drinking water and/or this report, please contact Scott A. DeVault, Water Department Superintendent for the City of New Philadelphia at (330) 339‐2332. Report Information This report contains information on issues pertaining to the quality and supply of our drinking water including: • Water Source • Drinking Water Contents • Water Treatment Process • Water Quality Test Results • Special Health Concerns • Resources Available for Additional Information The Source of New Philadelphia’s Drinking Water The City of New Philadelphia obtains its drinking water from four wells screened in unconsolidated sand and gravel. The sources of drinking water both tap water and bottled water includes rivers, lakes, streams, ponds, reservoirs, springs, and wells. -

Process Water Production from River Water by Ultrafiltration and Reverse Osmosis
Process Water Production from River Water by Ultrafiltration and Reverse Osmosis M. Clever1, F.Jordt1,R.Knauf1,N.Räbiger2, M. Rüdebusch2, R. Hilker-Scheibel3 1 Axiva GmbH, Geb. G811, Industriepark Höchst, D 65926 Frankfurt Tel.: 069 / 305 16935, Fax: 069 / 30 90 14, E-Mail: [email protected] 2 Universität Bremen, 3 Stahlwerke Bremen GmbH 1. Abstract The new process for process water production from river water is divided into three stages: prefiltration, ultrafiltration and reverse osmosis. The latter technique has been state-of-the-art in the preparation of drinking water, boiler feed water and ultrapure water from conventionally pretreated raw water for many years. On the other hand, ultrafiltration is new to this sector and has only recently been used on the industrial scale. It is used as a single-stage process to purify drinking and process water as well as surface water as an alternative to conventional treatment processes (e.g. ozonization-precipitation-flocculation-coagulation-chlorination-gravel filtration). Multistage, fairly complex processes are employed in the conventional pretreatment of river water. The use of different chemicals necessitates special safety measures and careful harmonization and control of water chemistry in view of the requirements of downstream reverse osmosis. By contrast, processes based on membrane technology enable a simply designed plant to be used with several advantages. Axiva in cooperation with the water supply company for the Höchst Industrial Park have developed and successfully tested an efficient and cost-effective ultrafiltration process for river water in pilot-scale operation over several years. During this trial period low-energy systems based on different filtration concepts (cross-flow, dead-end), efficient hydrophilic membranes and a specific operating and backflushing technique specially designed for the application were employed. -
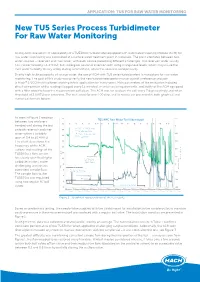
New TU5 Series Process Turbidimeter for Raw Water Monitoring
APPLICATION: TU5 FOR RAW WATER MONITORING New TU5 Series Process Turbidimeter For Raw Water Monitoring A long-term evaluation of applicability of a TU5300sc turbidimeter equipped with Automated Cleaning Module (ACM) for raw water monitoring was conducted at a surface water treatment plant in Colorado. The plant alternates between two water sources – reservoir and river water, with each source presenting different challenges. The reservoir water usually has a lower turbidity (~1-2 NTU), but undergoes seasonal inversion with rising manganese levels, which may cause the river water turbidity to vary wildly during summertime, when this source is used primarily. Due to high fouling capacity of source water, the use of ACM with TU5 series turbidimeters is mandatory for raw water monitoring. The goal of this study was to verify the new turbidimeter performance against a reference analyser (a Hach® 1720D) that had been working in this application for many years. Main parameters of the evaluation included direct com parison of the readings (logged every 15 minutes), maintenance requirements, and ability of the ACM equipped with a fiber wiper to keep the measurement cell clean. The ACM was set to clean the cell every 7 days routinely and when threshold of 3.5 NTU was exceeded. The test lasted for over 100 days and its results are presented in both graphical and numerical formats below. As seen in Figure 1 readings TU5 AMC Raw Water Test (fiber wiper) between two analysers trended well during the test on both reservoir and river TU5300-ACM, NTU 1720D RW, NTU water within a turbidity Auto-cleaning event span of 0.4 to 30.4 NTU.